��Ŀ����
ij����С����������װ��̽�������백��֮��ķ�Ӧ������A��F�ֱ�Ϊ�����������ķ���װ�ã�CΪ����������������백����Ӧ��װ�á�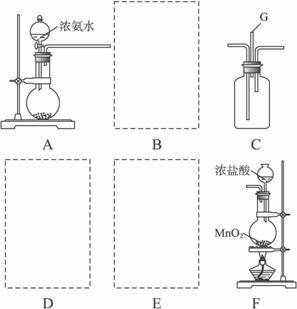
��ش��������⣺
��1��װ��F�з�����Ӧ�Ļ�ѧ����ʽ��__________________________________________��
��2��װ��A�е���ƿ�ڹ����ѡ��_____________��ѡ������ѡ��Ĵ��ţ���
A.��ʯ�� B.Ũ���� C.��ʯ�� D.���������� E.�ռ�
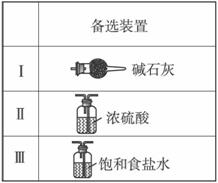
��3�����߿���Ӧ���ӱ�Ҫ�ij���װ�ã������ͼ�ı�ѡװ����ѡ��������������пո�
B______________��D______________��E______________��
��4�������Ͱ����ڳ��������ϾͻᷴӦ�����Ȼ�狀͵������÷�Ӧ�Ļ�ѧ����ʽΪ��__________________��װ��C�ڳ���Ũ��İ��̲��������ڱ����ᣬ�����һ��ʵ�鷽�������ù�������Ȼ�泥�__________________��
��5������װ��C��G���ݳ���β���к���N2������Cl2��Ӧ��δ������ܲ���Ⱦ������__________________________��
��1��MnO2+4HCl(Ũ)![]() MnCl2+Cl2��+2H2O
MnCl2+Cl2��+2H2O
��2��A��C��E
��3���� �� ��
��4��8NH3+3Cl2====N2+6NH4Cl ȡ������ɫ�������Թ��У�����ŨNaOH��Һ�����ȣ���ʪ��ĺ�ɫʯ����ֽ�����ܿڣ���ֽ����֤����![]() ����ȡ������ɫ�����ˮ�ܽ⣬Ȼ����������ữ��AgNO3��Һ��������ɫ������֤����Cl-����ʵ���֤����ɫ����ΪNH4Cl
����ȡ������ɫ�����ˮ�ܽ⣬Ȼ����������ữ��AgNO3��Һ��������ɫ������֤����Cl-����ʵ���֤����ɫ����ΪNH4Cl
��5��������ͨ��ʢ��NaOH��Һ���ձ���ϴ��ƿ����ͨ��ʢ�м�ʯ�ҵĸ���ܣ���Ȼ�����������
������װ��A��ȡ�����Dz��ù�����Һ�巴Ӧ�ķ�������ѡ�ü�ʯ�ң�NaOH��CaO�Ĺ����������ʯ�ң�CaO�����ռNaOH���壩�����Ǿ��Լ�����NH3����Ӧ������ˮ��ų��������ɴ�ʹNH3�Ļӷ�����װ��A������NH3�к�����H2O��g������װ��F������Cl2������HCl(g)��H2O��g����Ϊ�ô����������NH3��Cl2��C�з�Ӧ����NH3Ӧ��װ�м�ʯ�ҵ����θ���ܸ����Cl2��ͨ��װ�б���ʳ��ˮ��װ�â��ȥHCl(g)����ͨ��װ��Ũ�����װ�â��ȥCl2�е�H2O��g��������ij�����Ƿ���NH4Cl����ֱ�����ù�������![]() ��Cl-��
��Cl-��![]() �����ȵ�NaOH��Һ��ʪ��ĺ�ɫʯ����ֽ��������Cl-���������ữ��AgNO3��Һ��������Cl2�ж�����Ⱦ��������β���������ǰӦ��NaOH��Һϴ��������Cl2��
�����ȵ�NaOH��Һ��ʪ��ĺ�ɫʯ����ֽ��������Cl-���������ữ��AgNO3��Һ��������Cl2�ж�����Ⱦ��������β���������ǰӦ��NaOH��Һϴ��������Cl2��

 �������Ӧ���⼯ѵϵ�д�
�������Ӧ���⼯ѵϵ�д� �ۺ��Բ�ϵ�д�
�ۺ��Բ�ϵ�д�

