��Ŀ����
20������˵����ȷ���ǣ�������| A�� | ��pH��Ϊa������������Һ�Ͱ�ˮ�ֱ��ˮϡ��100����pH��Ϊb��c����a��b��c�Ĵ�С��ϵ�ǣ�b��c��a | |
| B�� | ������5.0��10-3mol•L-1KHA��Һ��pH=3.75������Һ��c��A2-����c��H2A�� | |
| C�� | a mol/L��HCN��Һ��b mol/L��NaOH��Һ�������ϣ�������Һ��c��Na+����c��CN-������aһ��С��b | |
| D�� | Na2C2O4��Һ��c��OH-��=c��H+��+c��HC2O4-��+2c��H2C2O4�� |
���� A������������Һ�Ͱ�ˮ������ͬ��pH����������������ǿ����ʣ�����ϡ��ʱǿ��ı仯�̶ȴ�ˮϡ�ʹٽ�һˮ�ϰ����룻
B��������5.0��10-3mol/LKHA��Һ��pH=3.75��˵��HA-���ֵ���������ӣ���HA-�ĵ���̶ȴ�����ˮ��̶ȣ�
C��c��Na+����c��CN-�������ݵ���غ��֪��Һ�ʼ��ԣ�a���ܵ���b��
D�����ݲ�������Һ�е������غ��жϣ�
��� �⣺A����ˮϡ������������Һ�Ͱ�ˮ��c��OH-������С����ˮϡ�ʹٽ�һˮ�ϰ����룬��ϡ�ͺ�ˮ��pH�����������ƣ���a��b��c�Ĵ�С��ϵ�ǣ�a��c��b����A����
B��������5.0��10-3mol/LKHA��Һ��pH=3.75��˵��HA-����Һ�в��ֵ���������ӣ���HA-�ĵ���̶ȴ�����ˮ��̶ȣ���c��A2-����c��H2A������B����
C����a=bʱ����Ӧ����ǿ��������NaCN����Һ�ʼ��ԣ���������Ũ��С�����������ӣ����ݵ���غ��֪c��Na+����c��CN-��������a���ܵ���b����C����
D��Na2C2O4��Һ�У����������غ�ɵã�c��OH-��=c��H+��+c��HC2O4-��+2c��H2C2O4������D��ȷ��
��ѡD��
���� ���⿼��������Ũ�ȴ�С�Ƚϡ��������ܶȻ��ļ��㣬��Ŀ�Ѷ��еȣ�ע�������������ܶȻ��ĸ�����㷽������ȷ��Һ���������Һ�ļ��㷽�����ܹ����ݵ���غ㡢�����غ㡢�����غ㡢�ε�ˮ��ԭ���ж���Һ�и�����Ũ�ȴ�С��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| �� �� |  | 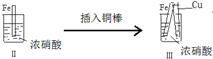 | |
| �� �� | ���������������ɫ���ݣ�Һ���Ϸ���Ϊ����ɫ | �����������������ɫ���ݺ�Ѹ��ֹͣ | ����ͭ�Ӵ�����������������ɫ���� |
| A�� | ������������ɫ�����ɫ�Ļ�ѧ����ʽΪ��2NO+O2��2NO2 | |
| B�� | ���е�����˵��Fe�����γ����ܵ������㣬��ֹFe��һ����Ӧ | |
| C�� | �ԱȢ�������˵��ϡHNO3��������ǿ��ŨHNO3 | |
| D�� | ���III��������Fe��Cu֮�����ӵ����ƣ����ж�Fe�Ƿ���������� |
| A�� | 6gʯī�еĹ��ۼ���Ŀ | |
| B�� | ���³�ѹ�£�17g ND3������������Ŀ | |
| C�� | 0.1mol�ǻ���-OH��������������Ŀ | |
| D�� | ��0.2mol NH4Al��SO4��2��Һ�еμ�NaOH��Һ��������ȫ�ܽ⣬���ĵ�OH-��Ŀ |
| A�� | ����Һ��pH=3��CH3COOH��pH=11��NaOH��Һ�������϶��� | |
| B�� | ����Һ�ɵ����ʵ���Ũ�ȡ��������NaOH��Һ��CH3COOH��Һ��϶��� | |
| C�� | ����������NaOH����Һ������Ũ��Ϊc��CH3COO-����c��Na+����c��OH-����c��H+�� | |
| D�� | ����������ˮ��c��CH3COO-�������c��Na+����c��NH4+��֮�� |
| A�� | NH4++H2O?NH3•H2O+H+ | |
| B�� | Al3++3H2O?Al��OH��3+3H+ | |
| C�� | CH3COO-+NH4++H2O?CH3COOH+NH3•H2O | |
| D�� | HCO3-+H2O?H3O++CO32- |
| A�� | ��x=4����W��ת�������� | B�� | ��x=3����W��ת���ʲ��� | ||
| C�� | ��W��ת����������xһ������1 | D�� | ȱ���������ж� |
��1�����Dz�����������Һ��pH������С��7�������ӷ���ʽ������ԭ����Fe2++2H2O?Fe��OH��2+2H+
��2����ͬѧ����������������Һ��Ϊ��ֹˮ�⣬����һ��������������������pH=1��ϡ��
���У���Һ������ɫ��Һ���Ϸ��к���ɫ���壬����һ��ʱ�䣬��Һ���ճʻ�ɫ��
����֪��Fe2+����NO����γ�����ɫ��[Fe��NO��]2+��Fe2++NO?��Fe��NO��]2+��
��Һ���Ϸ�����ɫ������NO2
�ھ������ɫ��Һ����Fe3+������Fe3+�IJ�����ȡ������ɫ��Һ���Թ��У�����KSCN��Һ����Һ�Ժ�ɫ��֤������Fe3+
�������Һ������[Fe��NO��]2+ʹ��Һ������ɫ��������Һ���ɫ��ԭ������ϡ������Fe2+��NO3-����ΪFe3+��ʹc��Fe2+�����ͣ�Fe2++NO?��Fe��NO��]2+��ƽ�������ƶ�����Fe2+����ȫ����ΪFe3+ʱ����Һ������ɫ��Ϊ��ɫ
��3����ͬѧ������������Һ����ʵ�飮
| ���� | ���輰���� |
 | i������Һ�л���ͨ��SO2��Һ���Ϸ��к���ɫ���壬��Һ��ɫ�����Ա仯 ii������ͨ��������SO2����Һ��Ϊ����ɫ��һ��ʱ�����Һ��Ϊdz��ɫ |
�ڼ���Ϊ�ɲ���i������ó�SO2��NO3-�����˷�Ӧ��û����Fe3+������Ӧ�����жϼĽ����Ƿ���ȷ��˵��ԭ����ȷ����Һ�Ϸ���������ɫ�����壬˵��NO3-����ԭ����Һû������ɫ���֣�˵��Fe2+û�б�����ΪFe3+
�۲���ii��Һ��Ϊdz��ɫ����Ӧ�����ӷ���ʽ��2Fe3++SO2+2H2O=2Fe2++SO42-+4H+
�ܸ�ʵ��ó��Ľ�����SO2�Ļ�ԭ�Ա�Fe2+��ǿ��

 2Na2CO3•3H2O2��һ�����͵���ϵƯ����ijʵ����ȤС�����������ʵ�飮
2Na2CO3•3H2O2��һ�����͵���ϵƯ����ijʵ����ȤС�����������ʵ�飮