��Ŀ����
�ڷ�Ӧ3Cu + 8HNO3 = 3Cu(NO3)2 + 2NO�� + 4H2O�У�
��1��_________Ԫ�ػ��ϼ����ߣ�_________Ԫ�ػ��ϼ۽��͡�����Ԫ��������
��2��_________����������Cu������_________��Ӧ��_________���������
��3�������·�Ӧ�������ת�Ƶķ������Ŀ�����õ����ŷ���ʾ��
3Cu + 8HNO3 = 3 Cu(NO3)2 + 2NO�� + 4H2O
����1.5molCu�����˷�Ӧ����ת�Ƶĵ��ӵ�������__________________��
��1��ͭ���� ����1�֣� ![]() ��2��HNO3 ��������Cu(NO3)2����1�֣�
��2��HNO3 ��������Cu(NO3)2����1�֣�
![]() ��3��
��3��
3 Cu + 8 HNO3 = 3 Cu(NO3)2 + 2 NO �� + 4 H2O�� 2�֣���1.806��1024��2�֣�

��ϰ��ϵ�д�
 ��һ����ͬ���ɽ�����ϵ�д�
��һ����ͬ���ɽ�����ϵ�д� ������Ӧ���ϵ�д�
������Ӧ���ϵ�д� ��ʦ�㾦�ִʾ��ƪϵ�д�
��ʦ�㾦�ִʾ��ƪϵ�д�
�����Ŀ
|
�ڷ�Ӧ3Cu��8HNO3(ϡ) | |
| [����] | |
A�� |
8 mol |
B�� |
|
C�� |
2 mol |
D�� |
|
�ڷ�Ӧ3Cu��8HNO3(ϡ)===3Cu(NO3)2��2NO����4H2O�У�����1 molͭ������ʱ������ԭ������Ϊ(����)
| A��8 mol | B��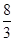 mol mol | C��2 mol | D��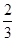 mol mol |
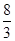 mol C��2 mol D��
mol C��2 mol D��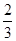 mol
mol