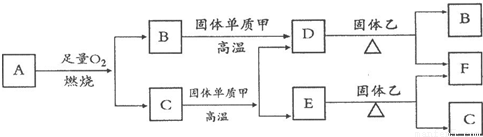
��Ŀ����
ij��ɫ��Һ��ֻ��������8�������е�ij���֣�Mg2+��H+��Ag+��Na+��Cl����HCO3��OH����NO3������֪������Һ�ܸ���������Ӧ���ҷų�������ֻ���������Իش�
��1������Һ������Ӧ��ֻ��AlO2�����ɣ���ԭ��Һһ�����еĴ��������� ��
������Ӧ�Ļ�ѧ����ʽ�� ��
�����ܺ��д��������� ��д��ѧʽ����
��2������Һ������Ӧ����Al3+���ɣ���ԭ��Һ��һ�������еĴ��������� ��
��1��NaOH��2Al+2NaOH+2H2O=2NaAlO2+3H2����NaCl��NaNO3
��2��HCO3����OH����Ag+��NO3��
����:
����Һ������Ӧ��ֻ��AlO2�����ɣ���ԭ��Һһ������OH������Mg2+��H+��Ag+��HCO3����������OH�����棬�ʲ�����Щ���ӣ�����Һ�ĵ����Կ�֪��Һ�б��������ӣ���������ֻ��Na+�����һ�����еĴ���������NaOH�����ܺ��д���������NaNO3��NaCl������Һ������Ӧ����Al3+���ɣ���ԭ��Һ��һ������H+����HCO3��OH����H+���ܹ��棻����NO3������ʱ����Һ�������ò��ų�����������Һ����HCO3����OH����NO3������Һ�е�������ֻ����Cl������Cl����Ag+���ܹ��棬��Ag+Ҳ�����ڡ�

 Fe2++Ag+����ش��������⣺
Fe2++Ag+����ش��������⣺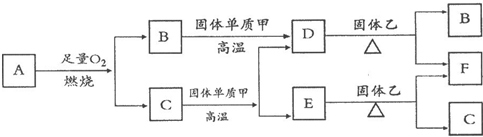

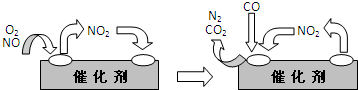
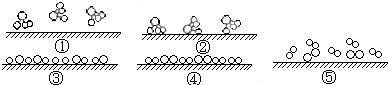
 Fe2++Ag+����ش��������⣺
Fe2++Ag+����ش��������⣺
 Fe2++Ag+����ش��������⣺
Fe2++Ag+����ش��������⣺