��Ŀ����
�����л�������ת����ϵ��ͼ��ʾ��
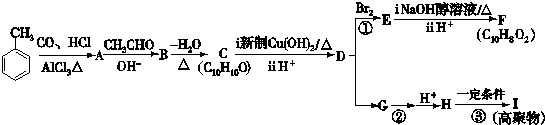
��֪��
��1��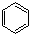
��2��CH3CO+CH3CHO
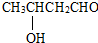
CH3CH=CHCHO���л�����A�����ϵ�һ�ȴ���ֻ�����֣�
��ش��������⣺
��1��д��A�Ľṹ��ʽ ��
��2��B�к��еĹ����ŵ����� ��
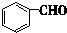
��3���ٵķ�Ӧ����Ϊ ��������F�ĺ˴Ź���������ʾ�� ���壮
��4��д��C������Cu��OH��2��Ӧ�Ļ�ѧ����ʽ ��
��5����֪������H���γ���Ԫ������д����Ӧ�ڵ����� ����Ӧ�۵Ļ�ѧ����ʽ ��
��6��������C�ж���ͬ���칹�壬��д������������������������ͬ���칹��Ľṹ��ʽ ��
�����Ȼ�����Һ����ɫ��
�ڱ�����������ȡ������
�۱����ϵ�һ��ȡ����ֻ�����֣�
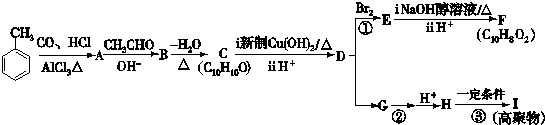
��֪��
��1��

| CO��HCl |
| AlCl3�� |

��2��CH3CO+CH3CHO
| OH- |
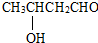
| -H2O |
| �� |
��ش��������⣺
��1��д��A�Ľṹ��ʽ
��2��B�к��еĹ����ŵ�����
��3���ٵķ�Ӧ����Ϊ
��4��д��C������Cu��OH��2��Ӧ�Ļ�ѧ����ʽ
��5����֪������H���γ���Ԫ������д����Ӧ�ڵ�����
��6��������C�ж���ͬ���칹�壬��д������������������������ͬ���칹��Ľṹ��ʽ
�����Ȼ�����Һ����ɫ��
�ڱ�����������ȡ������
�۱����ϵ�һ��ȡ����ֻ�����֣�
���㣺�л�����ƶ�
ר�⣺
������������A�����ϵ�һ�ȴ���ֻ�����֣�������Ϣ��1����֪AΪ ��A������Ϣ��2���еķ�Ӧ��BΪ
��A������Ϣ��2���еķ�Ӧ��BΪ ��CΪ
��CΪ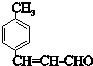 ��C������DΪ
��C������DΪ ��������H���γ���Ԫ������˵��H���Ȼ��Ħ�-̼�������ǻ�������HΪ
��������H���γ���Ԫ������˵��H���Ȼ��Ħ�-̼�������ǻ�������HΪ �����Է�Ӧ�Ƶ�G���Ȼ��Ħ�-̼������±ԭ�ӣ�D��±����ӳɵ�GΪ
�����Է�Ӧ�Ƶ�G���Ȼ��Ħ�-̼������±ԭ�ӣ�D��±����ӳɵ�GΪ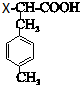 ��D���巢���ӳɷ�Ӧ��EΪ
��D���巢���ӳɷ�Ӧ��EΪ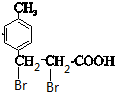 ��E������ȥ��Ӧ��FΪ
��E������ȥ��Ӧ��FΪ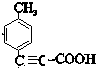 ���ݴ˴��⣮
���ݴ˴��⣮
 ��A������Ϣ��2���еķ�Ӧ��BΪ
��A������Ϣ��2���еķ�Ӧ��BΪ ��CΪ
��CΪ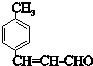 ��C������DΪ
��C������DΪ ��������H���γ���Ԫ������˵��H���Ȼ��Ħ�-̼�������ǻ�������HΪ
��������H���γ���Ԫ������˵��H���Ȼ��Ħ�-̼�������ǻ�������HΪ �����Է�Ӧ�Ƶ�G���Ȼ��Ħ�-̼������±ԭ�ӣ�D��±����ӳɵ�GΪ
�����Է�Ӧ�Ƶ�G���Ȼ��Ħ�-̼������±ԭ�ӣ�D��±����ӳɵ�GΪ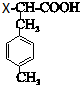 ��D���巢���ӳɷ�Ӧ��EΪ
��D���巢���ӳɷ�Ӧ��EΪ ��E������ȥ��Ӧ��FΪ
��E������ȥ��Ӧ��FΪ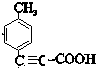 ���ݴ˴��⣮
���ݴ˴��⣮���
�⣺������A�����ϵ�һ�ȴ���ֻ�����֣�������Ϣ��1����֪AΪ ��A������Ϣ��2���еķ�Ӧ��BΪ
��A������Ϣ��2���еķ�Ӧ��BΪ ��CΪ
��CΪ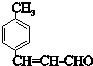 ��C������DΪ
��C������DΪ ��������H���γ���Ԫ������˵��H���Ȼ��Ħ�-̼�������ǻ�������HΪ
��������H���γ���Ԫ������˵��H���Ȼ��Ħ�-̼�������ǻ�������HΪ �����Է�Ӧ�Ƶ�G���Ȼ��Ħ�-̼������±ԭ�ӣ�D��±����ӳɵ�GΪ
�����Է�Ӧ�Ƶ�G���Ȼ��Ħ�-̼������±ԭ�ӣ�D��±����ӳɵ�GΪ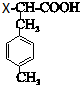 ��D���巢���ӳɷ�Ӧ��EΪ
��D���巢���ӳɷ�Ӧ��EΪ ��E������ȥ��Ӧ��FΪ
��E������ȥ��Ӧ��FΪ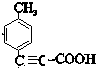 ��
��
��1��������������֪��A�Ľṹ��ʽΪ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��������
��������
��2��BΪ ��B�к��еĹ����ŵ�����Ϊ�ǻ���ȩ�����ʴ�Ϊ���ǻ���ȩ����
��B�к��еĹ����ŵ�����Ϊ�ǻ���ȩ�����ʴ�Ϊ���ǻ���ȩ����
��3����������ķ�����֪���ٵķ�Ӧ����Ϊ �ӳɷ�Ӧ��FΪ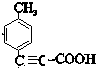 ��F�ĺ˴Ź���������ʾ��4���壬�ʴ�Ϊ���ӳɷ�Ӧ��4��
��F�ĺ˴Ź���������ʾ��4���壬�ʴ�Ϊ���ӳɷ�Ӧ��4��
��4��CΪ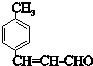 ��C������Cu��OH��2��Ӧ�Ļ�ѧ����ʽΪ
��C������Cu��OH��2��Ӧ�Ļ�ѧ����ʽΪ ��
��
�ʴ�Ϊ�� ��
��
��5����������ķ�����֪����Ӧ�ڵ�����ΪNaOHˮ��Һ���ȣ���Ӧ�۵Ļ�ѧ����ʽ ��
��
�ʴ�Ϊ��NaOHˮ��Һ���ȣ� ��
��
��6��CΪ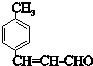 ��C�ж���ͬ���칹���������������������Ȼ�����Һ����ɫ��˵���зӵĽṹ���ڱ�����������ȡ�������۱����ϵ�һ��ȡ����ֻ�����֣�������ͬ���칹��Ľṹ��ʽΪ
��C�ж���ͬ���칹���������������������Ȼ�����Һ����ɫ��˵���зӵĽṹ���ڱ�����������ȡ�������۱����ϵ�һ��ȡ����ֻ�����֣�������ͬ���칹��Ľṹ��ʽΪ ��
��
�ʴ�Ϊ�� ��
��
 ��A������Ϣ��2���еķ�Ӧ��BΪ
��A������Ϣ��2���еķ�Ӧ��BΪ ��CΪ
��CΪ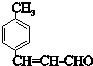 ��C������DΪ
��C������DΪ ��������H���γ���Ԫ������˵��H���Ȼ��Ħ�-̼�������ǻ�������HΪ
��������H���γ���Ԫ������˵��H���Ȼ��Ħ�-̼�������ǻ�������HΪ �����Է�Ӧ�Ƶ�G���Ȼ��Ħ�-̼������±ԭ�ӣ�D��±����ӳɵ�GΪ
�����Է�Ӧ�Ƶ�G���Ȼ��Ħ�-̼������±ԭ�ӣ�D��±����ӳɵ�GΪ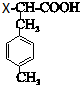 ��D���巢���ӳɷ�Ӧ��EΪ
��D���巢���ӳɷ�Ӧ��EΪ ��E������ȥ��Ӧ��FΪ
��E������ȥ��Ӧ��FΪ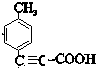 ��
����1��������������֪��A�Ľṹ��ʽΪ
 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��������
����������2��BΪ
 ��B�к��еĹ����ŵ�����Ϊ�ǻ���ȩ�����ʴ�Ϊ���ǻ���ȩ����
��B�к��еĹ����ŵ�����Ϊ�ǻ���ȩ�����ʴ�Ϊ���ǻ���ȩ������3����������ķ�����֪���ٵķ�Ӧ����Ϊ �ӳɷ�Ӧ��FΪ
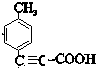 ��F�ĺ˴Ź���������ʾ��4���壬�ʴ�Ϊ���ӳɷ�Ӧ��4��
��F�ĺ˴Ź���������ʾ��4���壬�ʴ�Ϊ���ӳɷ�Ӧ��4����4��CΪ
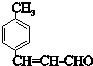 ��C������Cu��OH��2��Ӧ�Ļ�ѧ����ʽΪ
��C������Cu��OH��2��Ӧ�Ļ�ѧ����ʽΪ ��
���ʴ�Ϊ��
 ��
����5����������ķ�����֪����Ӧ�ڵ�����ΪNaOHˮ��Һ���ȣ���Ӧ�۵Ļ�ѧ����ʽ
 ��
���ʴ�Ϊ��NaOHˮ��Һ���ȣ�
 ��
����6��CΪ
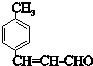 ��C�ж���ͬ���칹���������������������Ȼ�����Һ����ɫ��˵���зӵĽṹ���ڱ�����������ȡ�������۱����ϵ�һ��ȡ����ֻ�����֣�������ͬ���칹��Ľṹ��ʽΪ
��C�ж���ͬ���칹���������������������Ȼ�����Һ����ɫ��˵���зӵĽṹ���ڱ�����������ȡ�������۱����ϵ�һ��ȡ����ֻ�����֣�������ͬ���칹��Ľṹ��ʽΪ ��
���ʴ�Ϊ��
 ��
��
���������⿼���л����ƶϣ�����л���ṹ��ȡ�����������Ӷ��ó����л�������Ľṹ���Ƕ��л���ѧ����֪ʶ���ۺϿ��飬��Ҫѧ���������շ��ӽṹ�еĹ����ţ�ץ���������ʽ��н���Ѷ��еȣ�

��ϰ��ϵ�д�
 ��1����Ԫ�¿�������ĩϵ�д�
��1����Ԫ�¿�������ĩϵ�д�
�����Ŀ
���й���Na2O��Na2O2 ��˵���У���ȷ���ǣ�������
| A��������ˮ��Ӧ |
| B����Ԫ�صĻ��ϼ۾�Ϊ-2 |
| C����Ϊ����ɫ���� |
| D��������CO2��Ӧ�ų����� |
���в����У���ʹ����ƽ�⣺H2O�TH++OH-�������ƶ�����Һ�����Ե��ǣ�������
| A����ˮ�м���Na2CO3��Һ |
| B����ˮ�м���Al2��SO4��3���� |
| C����ˮ�м���NaHSO4��Һ |
| D����ˮ���ȵ�100�棬ʹpH=6 |
1molH2��O2��ȼ������Һ̬ˮ�ų�285.8kJ���������������Ȼ�ѧ����ʽ��ȷ���ǣ�������
| A��2H2��g��+O2��g��=2H2O��l����H=+571.6kJ?mol-1 | ||
| B��2H2��g��+O2��g��=2H2O��l������H=-571.6kJ?mol-1 | ||
C��H2��g��+
| ||
D��H2��g��+
|
���������У���Ҫ�ɷ����ڹ����ε��ǣ�������
| A���ռ� | B��ˮ�� | C��ʯ��ʯ | D��ˮ�� |
��NA��ʾ�����ӵ�������ֵ�������й�˵����ȷ���ǣ�������
| A��ͬ��ͬѹ�£�ԭ������ΪNA�������ͺ���������ͬ����� |
B��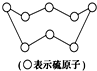 32gS8�����к��е�S-S������ΪNA����ͼ�� |
| C��10L 0.1mol/L Na2S��ˮ��Һ�У�S2-��HS-��������֮��ΪNA�� |
| D����״���£�22.4LNO��11.2LO2��Ϻ�����ķ�������Ϊ1.5NA |
��֪���ᣨHA�������Աȼ��ᣨHB�����������ʵ���Ũ�Ⱦ�Ϊ0.1mol?L-1��NaA��NaB�����Һ�У�����������ȷ���ǣ�������
| A��c��OH-����c��HA����c��HB����c��H+�� |
| B��c��OH-����c��A-����c��B-����c��H+�� |
| C��c��OH-����c��B-����c��A-����c��H+�� |
| D��c��OH-����c��HB����c��HA����c��H+�� |
