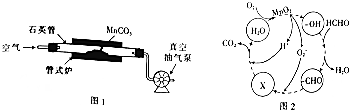
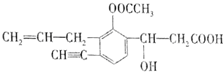
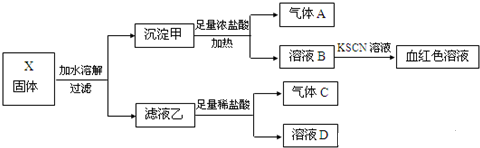
��Ŀ����
20��25��ʱ�������й���Һ���������ʵ���Ũ�ȹ�ϵ��ȷ���ǣ�������| A�� | ���ʵ���Ũ����ȵĢ�NH4Cl�ڣ�NH4��2SO4��NH4Al��SO4��2������Һ�У�c��NH4+���ɴ�С��˳��Ϊ���ۣ��ڣ��� | |
| B�� | 0.1mol•L-1CH3COOH��0.05mol•L-1NaOH��Һ�������ϣ�pH��7���� c��CH3COO-����c��Na+����c��CH3COOH����c��H+�� | |
| C�� | 0.1mol•L-1Na2CO3��0.05mol•L-1NaHCO3��Һ�������ϣ� $\frac{3}{5}$c��Na+��=c��CO32-��+c��HCO3-��+c��H2CO3�� | |
| D�� | ��0.1mol•L-1NaHC2O4��Һ��pH��7���У�c��OH-��+2c��C2O42-��=c��H+��+2c��H2C2O4�� |
���� A����NH4Cl �ڣ�NH4��2SO4 ��NH4Al��SO4��2���Ȳ�����ˮ�⣬��ڣ�NH4��2SO4 ��������NH4+����������NH4+��Ũ�ȴ��������������ʣ��٢���Һ��c��NH4+����������ˮ���Ӱ������жϣ�
B.0.1mol•L-1CH3COOH��0.05mol•L-1NaOH��Һ�������ϵõ������ʹ����ƵĻ����Һ��pH��7��Һ������˵���ǵ�����ڴ�������ӵ�ˮ�⣺
C.0.1 mol•L-1Na2CO3��0.05mol•L-1 NaHCO3��Һ����������Һ�д��������غ㣬0.15n��Na��=0.25n��C������3n��Na��=5n��C����
D����0.1 mol•L-1NaHC2O4��Һ��pH��7������Һ�����ԣ�HC2O4-�������ˮ�⣬��Һ�д��ڵ���غ�������غ���������
��� �⣺A����NH4Cl �ڣ�NH4��2SO4 ��NH4Al��SO4��2���Ȳ�����ˮ�⣬��ڣ�NH4��2SO4 ��������NH4+����������NH4+��Ũ�ȴ��������������ʣ��٢۶��������Т�NH4Cl��NH4+ˮ�⣬��NH4Al��SO4��2�������ӵ�ˮ���笠��������������ã�����Һ��c��NH4+���ڣ��ۣ��٣���A����
B.0.1mol•L-1CH3COOH��0.05mol•L-1NaOH��Һ�������ϵõ������ʹ����ƵĻ����Һ��pH��7��Һ������˵���ǵ�����ڴ�������ӵ�ˮ�⣬��Һ������Ũ�ȴ�СΪ��c��CH3COO-����c��Na+����c��CH3COOH����c��H+������B��ȷ��
C.0.1 mol•L-1Na2CO3��0.05mol•L-1 NaHCO3��Һ����������Һ�д��������غ㣬0.15n��Na��=0.25n��C������3n��Na��=5n��C����$\frac{3}{5}$c��Na+��=c��CO32-��+c��HCO3-��+c��H2CO3������C��ȷ��
D����0.1 mol•L-1NaHC2O4��Һ��pH��7������Һ�����ԣ����������غ��c��Na+��=c��HC2O4-��+c��H2C2O4��+c��C2O42-�������ݵ���غ��c��H+��+c��Na+��=c��HC2O4-��+2c��C2O42-��+c��OH-�������Ե�c��H+��+c��H2C2O4��=2c��C2O42-��+c��OH-������D����
��ѡBC��
���� ���⿼������ˮ���ԭ����Ӧ�á��������Һ�е���غ㡢�����غ������ע������ˮ���ʵ�ʺͰ���������ʵĵ��룬��Ӱ��ƽ���ƶ��ĽǶȷ�������Ũ�ȵĴ�С�Ƚ��ǽ��Ĺؼ�����Ŀ�Ѷ��еȣ�

| ʵ���� | ʵ���¶�/�� | c��Na2S2O3��/��mol•L-1�� | c��H2SO4��/��mol•L-1�� |
| �� | 25 | 0.1 | 0.1 |
| �� | 25 | 0.2 | 0.1 |
| �� | 25 | 0.1 | 0.2 |
| �� | 50 | 0.2 | 0.1 |
| �� | 50 | 0.1 | 0.1 |
��ͬʱѡ��ʵ��١�ʵ��ڡ�ʵ��ۣ��ⶨ���Һ����ǵ�ʱ�䣬��̽���Ƚϸı䲻ͬ��Ӧ���Ũ�ȶԷ�Ӧ���ʵ�Ӱ��Ի�ѧ��Ӧ���ʵ�Ӱ�죮
��2����һ���¶��£�����4a mol H2��2a mol N2����V L���ܱ������У�5min����N2��ת����Ϊ50%����ö�ʱ����H2��ʾ�ķ�Ӧ����Ϊ$\frac{3a}{5V}$ mol/��L•min����
ʵ�������֪2KMnO4+5H2C2O4+3H2SO4�TK2SO4+2MnSO4+8H2O+10CO2�����ڿ�ʼһ��ʱ���ڣ���Ӧ���ʽ�С����Һ��ɫ�����ԣ�������ͻȻ��ɫ����Ӧ������������
��3�������������ijͬѧ��Ϊ�÷�Ӧ���ȣ�������Һ�¶���������Ӧ��������Ӱ�컯ѧ��Ӧ���ʵ����ؿ�������뻹�����Ǵ������������̻�Mn2+�Ĵ����ã���Ӱ�죮
��4������ʵ��֤����IJ��룬�����Ը��������Һ��������Һ�⣬�����ڷ�Ӧһ��ʼʱ����B������ĸ��ţ���
A������� B�������� C���Ȼ��� D��ˮ��
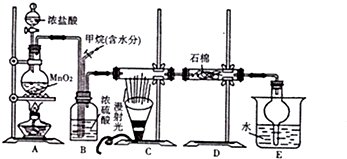

 ��
��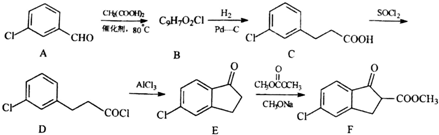
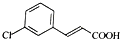 ����D��E�ķ�Ӧ������ȡ����Ӧ��
����D��E�ķ�Ӧ������ȡ����Ӧ�� ��
��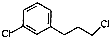 ��Ϊԭ���Ʊ�������E��
��Ϊԭ���Ʊ�������E��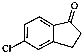 ���ĺϳ�·������ͼ�����Լ����ã����ϳ�·������ͼʾ�����£�
���ĺϳ�·������ͼ�����Լ����ã����ϳ�·������ͼʾ�����£�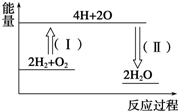 ����ȼ������Һ̬ˮ���Ȼ�ѧ����ʽΪ2H2��g��+O2��g���T2H2O��l����H=-572kJ•mol-1����ش��������⣺
����ȼ������Һ̬ˮ���Ȼ�ѧ����ʽΪ2H2��g��+O2��g���T2H2O��l����H=-572kJ•mol-1����ش��������⣺