��Ŀ����
14��̼�����׳ƴ����¶�ڿ����п�������еĶ�����̼��ˮ������Ӧ��Na2CO3+CO2+H2O�T2NaHCO3����һƿ���õ�̼���ƣ����к���̼�����ƣ���Ϊ�˲ⶨ����̼���Ƶ��������������������ʵ�鷽����
[��������]̼������ϡ���ᷴӦ�����������е�һ����ӦΪ��Na2CO3+HCl�TNaHCO3+NaCl
[ʵ��]ȡһ�������ľ���̼���ƹ�����Ʒ���ձ��У���ˮ���Ƴ���Һ��Ȼ����������μ���1mol/L��ϡ���ᣬʵ���õ�������ͼ��
����������Ϣ���й�ʵ�����ݷ������㣺
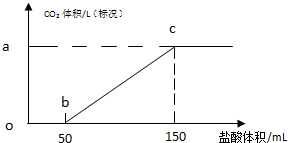
��1��ͼ��ob�μ���ϡ����û������ų���ԭ����̼���������ᷴӦ����̼�����ƺ��Ȼ��ƣ�
��2������ͼ��a�����ֵ��д��������̣���
��3�����õ�̼������̼���Ƶ�����������55.8%��������С�����һλ����
���� ��1����һ��̼���ƺ�HCl��Ӧ����NaHCO3��NaCl��û���������ɣ�
��2��50-100mLHClʱ������ӦNaHCO3+HCl=NaCl+CO2��+H2O�����������CO2֮���ϵʽ����CO2���ʵ������ٸ���V=nVm���������̼�����
��3������Na2CO3+HCl�TNaHCO3+NaCl��̼���ƺ�ϡ����֮���ϵʽ����Na2CO3����������Cԭ���غ�õ�̼����ת��Ϊ̼��������������ϣ�2���õ�ԭ����Һ��̼���������������Ӷ��õ�̼��������������
��� �⣺��1����һ��̼���ƺ�HCl��Ӧ����NaHCO3��NaCl��û���������ɣ��������������0-50mLʱû���������ɣ��ʴ�Ϊ��̼���������ᷴӦ����̼�����ƺ��Ȼ��ƣ�
��2��50-100mLHClʱ������ӦNaHCO3+HCl=NaCl+CO2��+H2O��
������CO2�����ʵ���Ϊn��CO2��
NaHCO3+HCl=NaCl+CO2��+H2O
1 1
1mol/L��0.10L n��CO2��
��ã�n��CO2��=0.10 mol
V��CO2��=0.10 mol��22.4 L/mol=2.24L
��a=2.24��
��3������Na2CO3+HCl�TNaHCO3+NaCl��̼���ƺ�ϡ����֮���ϵʽ��n��Na2CO3��=n��HCl��=1mol/L��0.05L=0.05mol����m��Na2CO3��=0.05mol��106g/mol=5.3g��
����Cԭ���غ�õ�̼����ת��Ϊ̼���������ʵ�����n��NaHCO3��=n��Na2CO3��=0.05mol��m��NaHCO3��=0.05mol��84g/mol=4.2g��
����Cԭ���غ��ԭ��m��NaHCO3��=n��CO2����M��NaHCO3��=0.1mol��84g/mol-4.2g=4.2g��
̼������������=$\frac{5.3g}{5.3g+4.2g}$��100%=55.8%��
�ʴ�Ϊ��55.8%��
���� ���⿼��������йؼ��㣬Ϊ��Ƶ���㣬���ؿ���ѧ������������������ȷ����������֮���ϵ�ǽⱾ��ؼ���ע��ԭ���غ��������ã���Ŀ�ѶȲ���

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | ʳ��-C2H5OH | B�� | ����-KAl��SO4��2 | C�� | �մ�-NaHCO3 | D�� | ��ʯ��-CaO |
| A�� | 0.1 mol•L-1 Na2CO3��Һ�У�c��HCO3-��=2c��H2CO3��+c��H+��-c��OH-�� | |
| B�� | ���ʵ���Ũ����ȵ�Na2SO3��NaHSO3��Һ�У�3c��Na+��=2[c��HSO3-��+c��H2SO3��+c��SO32-��] | |
| C�� | ͬŨ�ȵ�������Һ�У���NH4HSO4 ��NH4Cl ��NH3•H2O��c��NH4+���ɴ�С��˳���ǣ��ڣ��٣��� | |
| D�� | �����£�Ũ�Ⱦ�Ϊ0.1mol/L NaA��HA��Һ��pH=8��c��HA����c��Na+����c��A-����c��OH-����c��H+�� |
| A�� | �ø����pH��ֽ�ⶨNaClO��Һ��pH | |
| B�� | �����µ�ʯӢ�����н����ۻ��������ƹ����ʵ�� | |
| C�� | �÷�Һ©�������屽��ˮ�Ļ����ʱ���屽���¿ڷų���ˮ���Ͽڵ��� | |
| D�� | ��������������Ϊ10%������ͭ��Һ����ȷ��ȡ10 g����ͭ��������90 gˮ�� |
| A�� | ���ڵ���ƽ�⡢ˮ��ƽ��ͳ����ܽ�ƽ�� | |
| B�� | c��Na+��=c��Cl-����c��HCO3-����c��CO32-��=c��Ca2+�� | |
| C�� | c��Na+��+2c��Ca2+��=c��HCO3-��+2c��CO32-��+c��Cl-�� | |
| D�� | ��Ӧ�����ӷ���ʽ��2HCO3-+Ca2+�TCaCO3��+H2CO3 |
| A�� | AlCl3��Һ�Ͱ�ˮ | B�� | NaHCO3��Һ��Ca��OH��2��Һ | ||
| C�� | Na HCO3��Һ������ | D�� | ������Һ���ռ���Һ |
 ���벹��������
���벹��������