��Ŀ����
3�������£���10mL 0.1mol•L-1��H2C2O4��Һ����μ���0.1mol•L-1KOH��Һ�����õζ�������ͼ��ʾ������˵����ȷ���ǣ�������
| A�� | KHC2O4��Һ�������� | |
| B�� | B��ʱ��c��K+����c��HC2O4-����c��C2O42-����c��H+����c��OH-�� | |
| C�� | C��ʱ��c��K+����c��HC2O4- ��+2c��C2O42-��+c��H2C2O4�� | |
| D�� | D��ʱ��c��H+��+c��HC2O4-��+c��H2C2O4��=c��OH-�� |
���� ����ͼ֪��B�����ǡ����ȫ��Ӧ����KHC2O4����Һ��pH��7��˵������������ӵ���̶ȴ���ˮ��̶ȵ�����Һ�����ԣ���������ˮ��̶ȶ���С��
C����Һ�����ԣ�����Ϊ������غͲ���صĻ����
D�����ǡ����ȫ��Ӧ����K2C2O4��
�κε������Һ�ж����ڵ���غ�������غ㣬���ݵ���غ�������غ�������
��� �⣺����ͼ֪��B�����ǡ����ȫ��Ӧ����KHC2O4����Һ��pH��7��˵������������ӵ���̶ȴ���ˮ��̶ȵ�����Һ�����ԣ���������ˮ��̶ȶ���С��
C����Һ�����ԣ�����Ϊ������غͲ���صĻ����
D�����ǡ����ȫ��Ӧ����K2C2O4��
A���������Ϸ���֪��KHC2O4��Һ�������ԣ���A����
B��B�����ǡ����ȫ��Ӧ����KHC2O4����Һ��pH��7��˵������������ӵ���̶ȴ���ˮ��̶ȵ�����Һ�����ԣ���������ˮ��̶ȶ���С������Һ��ˮҲ�������������ӣ�����c��K+����c��HC2O4-����c��H+����c��C2O42-����c��OH-������B����
C��C����Һ�����ԣ�����Ϊ������غͲ���صĻ�������ݵ���غ��c��K+��=c��HC2O4- ��+2c��C2O42-��������c��K+����c��HC2O4- ��+2c��C2O42-��+c��H2C2O4������C��ȷ��
D�����������غ��c��H+��+c��HC2O4-��+2c��H2C2O4��=c��OH-������D����
��ѡC��
���� ���⿼������Ũ�ȴ�С�Ƚϣ�Ϊ��Ƶ���㣬���ؿ���ѧ������������������ȷ�жϸ����յ���Һ�����ʳɷ��ǽⱾ��ؼ���ע���ϵ���غ㡢�����غ㼰��ҺpH���������Ŀ�Ѷ��еȣ�

��֪�����ж��ּ�̬������+5�����ȶ���������Һ����Ҫ��VO2+��VO3-����ʽ���ڣ��Ҵ���ƽ��VO2++H2O?VO3-+2H+������˵����ȷ���ǣ�������
| A�� | ��ҵ�����У���������ѡ�ý�Ϊ���۵İ�ˮ | |
| B�� | �����Ʋ�VO2Cl�ܽ�ȴ���NH4VO3 | |
| C�� | ���շǴ��Բ�Ʒ���õĹ����������ܽ�ʱ��Ӧ�������������ʹ���ܽ���ȫ | |
| D�� | �����Բ�Ʒ����ϡ�����ܽ⣬ȡ�ϲ���Һ�ټ���KSCN��Һ��δ��Ѫ��ɫ������Բ�Ʒ��һ��������Ԫ�� |
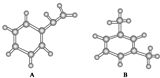
| A�� | CSO�ĵ���ʽ�� | |
| B�� | ������ӵ����ģ�ͣ� | |
| C�� | 2-��ϩ�Ľṹ��ʽCH3CH2CHCHCH3 | |
| D�� | HCO3-��ˮ�ⷽ��ʽΪ��HCO3-+H2O?CO32-+H3O+ |
| A�� | ���߶���ʹ����KMnO4��Һ��ɫ | B�� | ���߶�������ˮ��Ӧ��ʹ��ˮ��ɫ | ||
| C�� | ���߷���������ԭ�Ӿ����ܹ�ƽ�� | D�� | ���ߵ�һ�ȴ������Ŀ��� |
| A�� | һ������Na��O2��ȫ��Ӧ����1mol����ʱ��ת�Ƶ�����Ϊ2NA | |
| B�� | 23g�Ҵ���������������ȫ��Ӧʱ��������5.6L���� | |
| C�� | 1molHCl������1molNH3��ȫ��Ӧʱ��������NA��NH4Cl���� | |
| D�� | ��1molCH4��1molCl2�����³�ַ�Ӧ�������ɵ�һ�ȼ�����ӵ���ĿΪNA |
| ������ | Cu2+��Fe3+��Al3+��K+ |
| ������ | Cl-��CO32-��NO3-��SO42-�� |
I��ȡ������ˮ��Ʒ������KSCN��Һ�����Ա仯��
����ȡ������ˮ��Ʒ����μ���NaOH��Һ�����������������μӹ�����NaOH��Һ�����������ܽ⣮
��ش��������⣺
��1����ˮ��һ�����еĽ�����������Al3+Cu2+�������ӷ��ţ���
��2�����г��������ܽ�����з�����Ӧ�����ӷ���ʽ��Al��OH��3+OH-=[Al��OH��4]-��
��3����ͬѧͨ����������ȷ����ˮ��һ�����ܴ��������ϱ����������������е�CO32-�������ӷ��ţ������ж�������Al3+��CO32-��ˮ��Һ��ͬʱ����ˮ��������ˮ�������ٽ����ʷ�ˮ�в��ܴ�������CO32-����Cu2+��CO32-�������CuCO3�������ʷ�ˮ�в��ܴ�������CO32-����
| A�� | Cu2+��Na+��SO42-��Ba2+ | B�� | K+��Na+��HCO3-��NO3- | ||
| C�� | OH-��HCO3-��Ca2+��Na+ | D�� | Ba2+��Na+��OH-��NO3- |
