��Ŀ����
2��ij�ռ���Ʒ�к����������������õĿ��������ʣ�Ϊ�˲ⶨ�䴿�ȣ����еζ�������ѡ���������£�����Ͳ������ƿ���۵ζ��ܣ��ܲ�������������������ʹ��ǰ�����Ƿ�©ˮ���Ǣۣ�������ţ������������ȱ�IJ���������250mL����ƿ�С���ͷ�ιܣ�
�������裺
A����250mL����ƿ������250mL�ռ���Һ
B���ü�ʽ�ζ�����ȡ25.00mL�ռ���Һ����ƿ�в��Ӽ��μ���ָʾ��
C������ƽ��ȷ��ȡ�ռ���Ʒw g�����ձ��м�����ˮ�ܽ�
D�������ʵ���Ũ��Ϊm mol/L�ı�H2SO4��Һװ����ʽ�ζ��ܣ�����Һ�棬���¿�ʼ�̶�V1 mL
E������ƿ�µ�һ�Ű�ֽ���ζ����յ㣬��¼�յ�̶�ΪV2 mL
�������ָʾ����ɫ��Χ
| ָʾ�� | ��ɫ��Χ | ��ɫ | ��ɫ |
| ʯ�� | 5.0-8.0 | ��ɫ | ��ɫ |
| ��̪ | 8.0-10.0 | ��ɫ | ��ɫ |
| ���� | 3.1-4.4 | ��ɫ | ��ɫ |
��1����ȷ�IJ��������ǣ���д��ĸ��C��A��B��D��E��
��2���ζ��ܶ���Ӧע��ζ��ܴ�ֱ��Һ�治�ٱ仯ʱ�ſɶ�������������Һ��Һ����͵���ƽ��������ȷ��0.01ml��
��3����������ƿ�µ�һ�Ű�ֽ�������DZ���ȷ�ж��յ�ʱ��ɫ�ı仯�����
��4������D��Һ��Ӧ���������ڵ���̶Ȼ������µ�ijһ�̶ȣ�������H2SO4��Һ��
��5���ζ����յ�ʱ��ƿ����Һ��pHԼΪ4���յ�ʱ����ɫ�仯����Һ�ɻ�ɫ��Ϊ��ɫ���Ұ�����ڲ��ָ�Ϊԭ������ɫ��
��6������ʽ�ζ���û�ñ�H2SO4��ϴ����Բⶨ����к�Ӱ��ƫ�ߣ��ƫ�ߡ�����ƫ�͡�����Ӱ�족��������������ȷ����
���� ���л�����ƿ����װ��ʹ��ǰҪ����Ƿ�©ˮ������ʵ������IJ����Լ�ÿ��������Ҫ����ȷ����Ӧ����������
��1��ʵ��ʱӦ�ȳ���һ�������Ĺ��壬�ܽ�����Ƴ���Һ����ȡ����Һ����ƿ�У�Ȼ���ñ�Һ���еζ���
��2���ζ��ܴ�ֱ��Һ�治�ٱ仯ʱ�ſɶ�������������Һ��Һ����͵���ƽ��ע��ζ��ܵľ�ȷֵ��
��3������ƿ�µ�һ�Ű�ֽʹ�ζ��յ���ɫ�仯�����ԣ����ڷֱ棻
��4���ζ���0�̶����ϣ��ζ�ǰӦ���ڵ�0�̶Ȼ�0�̶����µ�ijһ�̶ȣ�Ϊ��С�����첿��Ӧ����Һ�壬�����ݣ�
��5��ָʾ��Ϊ���ȣ���ɫ��ΧΪ3.1-4.4��
��6������c�����⣩=$\frac{V���꣩��c���꣩}{V�����⣩}$��2��������������V�����⣩��Ӱ�죬�Դ��ж�Ũ�ȵ���
��� �⣺�۵ζ��ܴ��л���ʹ��ǰӦ����Ƿ�©ˮ��������250mL�ռ���Һ����Ҫ���������У�250mL����ƿ�С���ͷ�ιܣ�
�ʴ�Ϊ���ۣ�250mL����ƿ�С���ͷ�ιܣ�
��1��ʵ��ʱӦ�ȳ���һ�������Ĺ��壬�ܽ�����Ƴ���Һ����ȡ����Һ����ƿ�У�Ȼ���ñ�Һ���еζ������岽��Ϊ��
C������ƽ��ȷ��ȡ�ռ���Ʒw g�����ձ��м�����ˮ�ܽ�
A����250mL����ƿ�ж��ݳ�250mL�ռ���Һ
B���ü�ʽ�ζ�����ȡ25.00mL�ռ���Һ����ƿ�в��Ӽ��μ���ָʾ��
D�������ʵ���Ũ��Ϊm mol/L�ı�H2SO4��Һװ����ʽ�ζ��ܣ�����Һ�棬���¿�ʼ�̶�V1 mL
E������ƿ�µ�һ�Ű�ֽ���ζ����յ㣬��¼�յ�̶�ΪV2 mL
�ʴ�Ϊ��C��A��B��E��
��2���ζ��ܴ�ֱ��Һ�治�ٱ仯ʱ�ſɶ�������������Һ��Һ����͵���ƽ���ζ��ܶ���ȷ��0.01ml��
�ʴ�Ϊ���ζ��ܴ�ֱ��Һ�治�ٱ仯ʱ�ſɶ�������������Һ��Һ����͵���ƽ��������ȷ��0.01ml��
��3������ƿ�µ�һ�Ű�ֽʹ�ζ��յ���ɫ�仯�����ԣ����ڷֱ棻
�ʴ�Ϊ������ȷ�ж��յ�ʱ��ɫ�ı仯��
��4���ζ���0�̶����ϣ��ζ�ǰӦ���ڵ���̶Ȼ������µ�ijһ�̶ȣ�Ϊ��С�����첿��Ӧ����Һ�壬�����ݣ�
�ʴ�Ϊ�����ڵ���̶Ȼ������µ�ijһ�̶ȣ�������H2SO4��Һ��
��5��ָʾ��Ϊ���ȣ���ɫ��ΧΪ3.1-4.4���յ�ʱpHԼΪ4����Һ�ɻ�ɫ��Ϊ��ɫ���Ұ�����ڲ��ָ�Ϊԭ������ɫ��
�ʴ�Ϊ��4����Һ�ɻ�ɫ��Ϊ��ɫ���Ұ�����ڲ��ָ�Ϊԭ������ɫ��
��6������ʽ�ζ���û�ñ�H2SO4��ϴ����ҺŨ�Ƚ��ͣ����V���꣩ƫ����c�����⣩=$\frac{V���꣩��c���꣩}{V�����⣩}$��2��֪c�����⣩ƫ�ߣ�
�ʴ�Ϊ��ƫ�ߣ�
���� ���⿼������к͵ζ��������ڻ�ѧʵ����������Լ����ʵĺ����IJⶨ�����⣬��Ŀ�Ѷ��еȣ�ѧϰ��ע����ۣ�

 ����ѧ���ʱѧ����ϵ�д�
����ѧ���ʱѧ����ϵ�д� �ʰ�Ӣ��ͬ����ϰ��ϵ�д�
�ʰ�Ӣ��ͬ����ϰ��ϵ�д�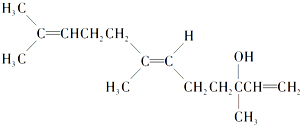
���й��ڳȻ�����������������ǣ�������
| A�� | ���ܷ���ȡ����Ӧ��Ҳ�ܷ����ӳɷ�Ӧ | |
| B�� | ��Ũ������¼�����ˮ���������ɲ�ֹһ����ϩ�� | |
| C�� | 1mol�Ȼ����������г��ȼ�գ�������448 L��������״���� | |
| D�� | 1mol�Ȼ�������������������Ȼ�̼��Һ��Ӧ���������3mol�� |
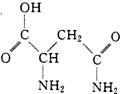 «���е��춬�������ṹ��ͼ������Ԫ�����������̵ȣ�������������������Ĺ�Ч��
«���е��춬�������ṹ��ͼ������Ԫ�����������̵ȣ�������������������Ĺ�Ч����1���춬��������Ԫ���У�������Ԫ�����ƣ�Ԫ�ػ�̬ԭ�Ӻ���δ�ɶԵ�������࣮
��2���춬������̼ԭ�ӵ��ӻ����������sp3��sp2��
��3��H2Se�ľ�������Ϊ���Ӿ��壬���еĹ��ۼ�����Ϊ���Թ��ۼ���
��4����֪�⣨Mo��λ�ڵ������ڡ���B�壬�⡢�����̵IJ��ֵ����������ʾ��
| ��� | I5/kJ•mol-1 | I6/kJ•mol-1 | I7/kJ•mol-1 | I8/kJ•mol-1 |
| A | 6990 | 9220 | 11500 | 18700 |
| B | 6702 | 8745 | 15455 | 17820 |
| C | 5257 | 6641 | 12125 | 13860 |
| A�� | ����̫���ܵ������Դ���滯ʯȼ�ϣ������ڽ�Լ��Դ���������� | |
| B�� | �������Ľո�ת��Ϊ����Ч����Դ | |
| C�� | Ϊ��ֹ����е��ؽ�������Ⱦ������ˮ�壬Ӧ���������ϵ�ص��ۺ����ü��� | |
| D�� | �����¼�������ú��ʯ�ͺ���Ȼ���������㾭�÷�չ����Ҫ |
| A�� | CH4��NH3��H2O��Ne | B�� | OH-��NH4+��H3O+��Ne | ||
| C�� | H3O+��NH4+��Na+��HF | D�� | OH-��F-��Mg2+��Na+ |
| A�� | NaNO2�������� | |
| B�� | ÿ����1 mol N2ʱ��ת�Ƶ�����ĿΪ6NA | |
| C�� | NH4Cl�еĵ�Ԫ�ر���ԭ | |
| D�� | N2���������������ǻ�ԭ�� |
| A�� | �Ҵ����ױ��������� | B�� | �������ӡ���ϩ | ||
| C�� | �����ױ��������� | D�� | �����顢�Ҵ����� |