��Ŀ����
13���ⶨij�������ClO2�ĺ������٣�����ƿ�м��������ĵ⻯�أ���50mLˮ�ܽ���ټ���һ�����ϡ���
�ڣ���һ�����Ļ������ͨ����ƿ�����գ�
�ۣ���0.1000mol/L��������Ʊ���Һ�ζ���ƿ�е���Һ��I2+2S2O32-=2I-+S4O62-����ָʾ����ʾ�յ�ʱ����ȥ20.00mL�����������Һ��
��3����ƿ�м����ָʾ��ͨ���ǵ�����Һ���ζ�ǰ���������ݣ��ζ������Ӷ�����ʹ�ò������ƫ�ߣ�ƫ�ߡ�ƫ�͡���Ӱ�죩
��4����ƿ��ClO2��⻯�ط�Ӧ�����ӷ���ʽ��2ClO2+10I-+8H+�T2Cl-+5I2+4H2O��
���� ��3��ClO2ͨ����ƿ�����Ե⻯����Һ��Ӧ������I-ΪI2����0.1000mol/L��������Ʊ���Һ�ζ���ƿ�е���Һ��I2+2S2O32-=2I-+S4O62-����ָʾ����ʾ�յ�ʱ����ȥ20.00mL�����������Һ��ָʾ��Ӧ�õ�����Һ������c�����⣩=$\frac{c������V������}{V�����⣩}$���������б���Һ����Ǿ������أ�
��4��ClO2ͨ����ƿ�����Ե⻯����Һ��Ӧ������I-ΪI2����������ԭΪCl-��ͬʱ����ˮ��
��� �⣺��3��ClO2ͨ����ƿ�����Ե⻯����Һ��Ӧ������I-ΪI2����0.1000mol/L��������Ʊ���Һ�ζ���ƿ�е���Һ��I2+2S2O32-=2I-+S4O62-����ָʾ����ʾ�յ�ʱ����ȥ20.00mL�����������Һ��ָʾ��Ӧ�õ�����Һ���ζ�����ɫ��ȥ�Ұ���Ӳ��䣬��Ӧ�ﵽ�յ㣬
���ζ�ǰ���������ݣ��ζ�����ʱ������ʧ�����V������ƫ����c�����⣩=$\frac{c������V������}{V�����⣩}$��������֪c������ƫ�ߣ��ζ������Ӷ�������ȡ����Һ���������ʹ�ò������ƫ�ߣ�
�ʴ�Ϊ��������Һ��ƫ�ߣ�
��4������Ŀ��Ϣ��֪��ClO2ͨ����ƿ�����Ե⻯����Һ��Ӧ������I-ΪI2����������ԭΪCl-��ͬʱ����ˮ����Ӧ���ӷ���ʽΪ2ClO2+10I-+8H+�T2Cl-+5I2+4H2O��
�ʴ�Ϊ��2ClO2+10I-+8H+�T2Cl-+5I2+4H2O��
���� ���⿼���˵ζ�ʵ��Ĺ��̷���������жϺ����ӷ���ʽ��д�����ջ����ǽ���ؼ�����Ŀ�ϼ�

 ��У����ϵ�д�
��У����ϵ�д�| A�� | Cl2��Fe3+��Cu2+��MnO2 | B�� | MnO2��Cl2��Fe3+��Cu2+ | ||
| C�� | Fe3+��Cu2+��MnO2��Cl2 | D�� | Cu2+��Fe3+��Cl2��MnO2 |
��֪��Fe3+��Al3+��Cu2+��ʼ������������ȫ��pH��Χ�ֱ�Ϊ��2.2��3.2��4.1��5.0��5.3��6.6��
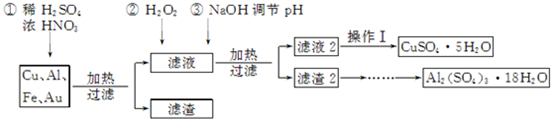
��1���õ�����1����Ҫ�ɷ�ΪAu��
��2�������������ʵ�鲽���У�����Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ�������˲������õ��IJ����������ձ�����������©����
��3���������г�����ˮ�Ҵ��Ծ������ϴ�ӣ�ѡ����ˮ�Ҵ���ԭ���Ǽ��پ�����ܽ⣬���ڸ��
��4���ڢڲ���H2O2������Ӧ�����ӷ���ʽΪ2Fe2++H2O2+2H+�T2Fe3++2H2O��
��5��ȡ��������������������ط��������ȷֽ���Ҫ��Ϊ�����Σ�323K��523K��553K��687K�����¶���1 043K���ϲ���ʧ�أ��±��г��˲�ͬ�¶��µ�ʧ���ʣ�
[���������廯ѧʽΪAl2��SO4��3•18H2O����Է�������Ϊ666]
��֪��ʧ��%=$\frac{���ȼ��ٵ�����}{ԭ������Ʒ��������}$��100%��
| �¶ȣ�K�� | ʧ�أ�%�� | |
| ��һ�� | 323��523 | 40.54 |
| �ڶ��� | 553��687 | 48.65 |
| ������ | 1 043���� | 84.68 |
��ʧ�ص�һ�ηֽ����Ļ�ѧʽ��
��ʧ�صڶ��η�Ӧ�ķ�Ӧ��ѧ����ʽ��
| A�� | ��Ӧ����������������ͬ | |
| B�� | ��Ӧ��ת�Ƶĵ�������ͬ | |
| C�� | ��Ӧ�����ĵ�HCl��NaOH���ʵ�����ͬ | |
| D�� | ��Һ���ӵ�������ͬ |
X����CH4+10OH--8e-=CO32-+7H2O
Y����O2+4e-+2H2O=4OH-
���й��ڴ�ȼ�ϵ�ص��й�˵���д�����ǣ�������
| A�� | ͨ������ĵ缫��X��Ϊ��صĸ�����ͨ�������ĵ缫��Y��Ϊ������� | |
| B�� | �ŵ�һ��ʱ��������Һ��KOH�����ʵ������ı� | |
| C�� | �ڱ�״���£�ͨ��5.6L��������ȫ��Ӧ����1.0mol�ĵ��ӷ���ת�� | |
| D�� | �ŵ�ʱ��ͨ�������һ��������pH���� |
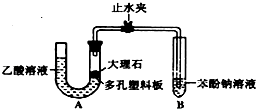 ijУѧ��С��Ϊ̽�����ᡢ̼��ͱ��ӵ�����ǿ����֤������Ϊ���ᣬ��������ʵ�飮
ijУѧ��С��Ϊ̽�����ᡢ̼��ͱ��ӵ�����ǿ����֤������Ϊ���ᣬ��������ʵ�飮 ��Һ
��Һ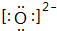 ��
�� ��
��