��Ŀ����
ʵ������Ҫ�ⶨ���ֱ��ʵ��������ƵĴ��ȣ�������ͼ1װ��ʵ�֣�
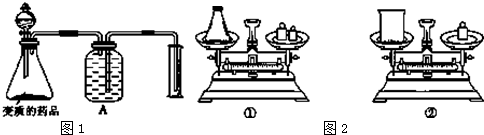
��1����Һ©���з� �������ƣ���Aװ�õ����� ���ŵ��Լ�Ϊ �������ƣ�����ʵ��ʱ�����ַ�Һ©���е�Һ����ѵ��£�ԭ����Ϊ ��
��2����ͼ2��������ƽ��ȡ��Ʒ��Ӧѡ����ͼ2�е� ����١��ڡ�������ѡ����һװ�õ�ԭ���� ��
��3����ȡ����Ʒ������Ϊ2.5g���ռ�����������Ϊ280mL����״����������Ʒ��NaOH�Ĵ���Ϊ ��
��4�����в����ᵼ��NaOH���ȵIJ���ֵƫ�͵��� ��
A����ҩƷ����������
B��ĩ��ȴ�����¾Ͷ���
C����ʼʱ��������A����Ͳ�ĵ���������ˮ����Ӧ��������ˮ
D������Ͳ������ʱ���ӣ�

��1����Һ©���з�
��2����ͼ2��������ƽ��ȡ��Ʒ��Ӧѡ����ͼ2�е�
��3����ȡ����Ʒ������Ϊ2.5g���ռ�����������Ϊ280mL����״����������Ʒ��NaOH�Ĵ���Ϊ
��4�����в����ᵼ��NaOH���ȵIJ���ֵƫ�͵���
A����ҩƷ����������
B��ĩ��ȴ�����¾Ͷ���
C����ʼʱ��������A����Ͳ�ĵ���������ˮ����Ӧ��������ˮ
D������Ͳ������ʱ���ӣ�
���㣺̽�����ʵ���ɻ�������ʵĺ���
ר�⣺ʵ��̽�������ݴ�����
��������1�����ֱ��ʵ����������к���̼���ƣ����װ��ͼ��������ƿ���DZ��ʵ���Ʒ����Һ©������ϡ���ᣬϡ�����̼������Һ��Ӧ���ɶ�����̼��Ϊ���ٶ�����̼���ܽ�ȿ����ű���̼��������Һ�ķ����ⶨ������̼���������Һ©����ʹ����Ҫ��Һ©�����ϵIJ����������ϵİ�����©����С������Һ�����£�
��2�����������������տ����е�H2O��CO2�����˱�¶�ڿ����г������ձ�����������������Ӵ������
��3���������ɶ�����̼����������ʵ����õ�̼�������ʵ�������������ȥ̼���������õ����������������������������ƵĴ��ȣ�
��4��A����ҩƷ���������⣬���������������ɶ�����̼����������ⶨ�������ƴ��ȼ�С��
B��ĩ��ȴ�����¾Ͷ����������������ⶨ�������ƫ��
C����ʼʱ��������A����Ͳ�ĵ���������ˮ����Ӧ��������ˮ���ⶨ���������С��
D������Ͳ������ʱ���Ӷ�ȡ������̼�����С���ⶨ�������ƴ�������
��2�����������������տ����е�H2O��CO2�����˱�¶�ڿ����г������ձ�����������������Ӵ������
��3���������ɶ�����̼����������ʵ����õ�̼�������ʵ�������������ȥ̼���������õ����������������������������ƵĴ��ȣ�
��4��A����ҩƷ���������⣬���������������ɶ�����̼����������ⶨ�������ƴ��ȼ�С��
B��ĩ��ȴ�����¾Ͷ����������������ⶨ�������ƫ��
C����ʼʱ��������A����Ͳ�ĵ���������ˮ����Ӧ��������ˮ���ⶨ���������С��
D������Ͳ������ʱ���Ӷ�ȡ������̼�����С���ⶨ�������ƴ�������
���
�⣺��1�����ֱ��ʵ����������к���̼���ƣ����װ��ͼ��������ƿ���DZ��ʵ���Ʒ����Һ©������ϡ���ᣬϡ�����̼������Һ��Ӧ���ɶ�����̼��Ϊ���ٶ�����̼���ܽ�ȿ����ű���̼��������Һ�ķ����ⶨ������̼�������Aװ��Ϊ���ƿ������Һ�巽���ⶨ���������Ϊ����ƿ����Һ©����ʹ����Ҫ��Һ©�����ϵIJ����������ϵİ�����©����С������Һ�����£�
�ʴ�Ϊ��ϡ�������ƿ������̼�����ƣ�û�д�Һ©�����ϵIJ����������ϵİ�����©����С��û�ж��룻
��2�����������������տ����е�H2O��CO2�����˱�¶�ڿ����г������ձ�����������������Ӵ����������ѡ������ƿ��ȡ��
�ʴ�Ϊ���٣����������������տ����е�H2O��CO2�����˱�¶�ڿ����г�����
��4��A����ҩƷ���������⣬���������������ɶ�����̼����������Ʒ��̼��������������ⶨ�������ƴ��ȼ�С����A��ȷ��
B��ĩ��ȴ�����¾Ͷ����������������ⶨ������̼�������ƫ�����ɶ�����̼����������Ʒ��̼��������������ⶨ�������ƴ��ȼ�С����B��ȷ��
C����ʼʱ��������A����Ͳ�ĵ���������ˮ����Ӧ��������ˮ���ⶨ���������С��̼����������С���������ƴ�������C����
D������Ͳ������ʱ���Ӷ�ȡ������̼�����С���ⶨ�������ƴ�������D����
��ѡAB��
�ʴ�Ϊ��ϡ�������ƿ������̼�����ƣ�û�д�Һ©�����ϵIJ����������ϵİ�����©����С��û�ж��룻
��2�����������������տ����е�H2O��CO2�����˱�¶�ڿ����г������ձ�����������������Ӵ����������ѡ������ƿ��ȡ��
�ʴ�Ϊ���٣����������������տ����е�H2O��CO2�����˱�¶�ڿ����г�����
��4��A����ҩƷ���������⣬���������������ɶ�����̼����������Ʒ��̼��������������ⶨ�������ƴ��ȼ�С����A��ȷ��
B��ĩ��ȴ�����¾Ͷ����������������ⶨ������̼�������ƫ�����ɶ�����̼����������Ʒ��̼��������������ⶨ�������ƴ��ȼ�С����B��ȷ��
C����ʼʱ��������A����Ͳ�ĵ���������ˮ����Ӧ��������ˮ���ⶨ���������С��̼����������С���������ƴ�������C����
D������Ͳ������ʱ���Ӷ�ȡ������̼�����С���ⶨ�������ƴ�������D����
��ѡAB��
���������⿼���˻�ѧʵ��̽��������ɵķ����жϣ���Ҫ�ǹ��̷���������жϣ����������Ӧ�ã����ջ����ǹؼ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
����ӦZn+Br2=ZnBr2��Ƴ����أ��������й�˵������ȷ���ǣ�������
| A�����ʱ�������ĵ缫��ӦʽΪ��2Br--2e-=Br2 |
| B���ŵ�ʱ�������ĵ缫��ӦʽΪ��Zn-2e-=Zn2+ |
| C���ŵ�ʱ��Zn2+ ���� |
| D���������Һ��������ǿ�Ҳ��������ǿ�� |
��½����̬ϵͳ�о��У�2H��13C��15N��18O��34S�ȳ�������������ָʾ�����˵����ȷ���ǣ�������
| A��34Sԭ�Ӻ���������Ϊ16 |
| B������ͬ������1H216O��1H218O�������� |
| C��13C��15Nԭ�Ӻ��ڵ����������2 |
| D��2H+�����Ա�1H+�����Ը�ǿ |
����ʱ��������ʵ����֤��������������ǣ�������
| A����ô�������Һ��pH��7 |
| B�����0.1mol/L������Һ��pH=3.3 |
| C����pH=1�Ĵ�����Һϡ��1000�������pH��4 |
| D������Ũ�ȡ�������Ĵ�����Һ������������Һ��Ϻ�ǡ���к� |
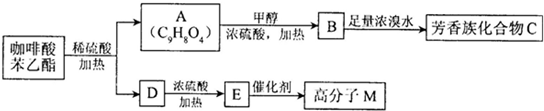
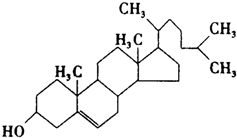
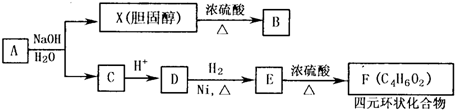
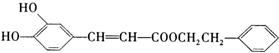 ����һ����Ȼ����ҩ���һ���������ܷ�������ת����
����һ����Ȼ����ҩ���һ���������ܷ�������ת����