��Ŀ����
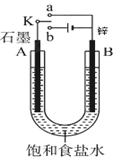
(8��)�����±ߵ�ʵ��װ��ͼ�ش��������⡣
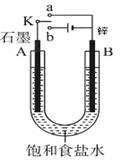
(1)����ʼʱ����K��a���ӣ���B���ĵ缫��ӦʽΪ ��
(2)����ʼʱ����K��b���ӣ���B���ĵ缫��ӦʽΪ ��
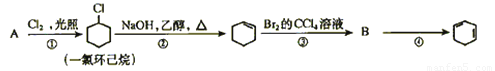
�ܷ�Ӧ�Ļ�ѧ����ʽΪ ��
�й�����ʵ�飬����˵����ȷ����(�����) ��
����Һ��Na+��A���ƶ�
�ڴ�A�����ݳ���������ʹʪ��KI������ֽ����
�۷�Ӧһ��ʱ������������ɻָ������ǰ����ʵ�Ũ��
������״����B������2.24 L���壬���·��ת��0.2 mol����
(1) Zn��2e��=Zn2�� (2)2H����2e����H2��
2NaCl��2H2O Cl2����H2����2NaOH
�ڢ�
Cl2����H2����2NaOH
�ڢ�
��������(1)����ʼʱ����K��a���ӣ���ԭ��أ�п�ǻ��õĽ���������п�Ǹ�����ʯī�����������и�������ʽ��Zn��2e��=Zn2����
(2)����ʼʱ����K��b���ӣ��ɵ��ء�B�缫�͵�Դ�ĸ�������������������Һ�е������ӷŵ磬����ʽ��2H����2e����H2����������ʯī����Һ�е������ӷŵ磬�����ܵķ�Ӧʽ��2NaCl��2H2O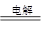 Cl2����H2����2NaOH���������������������ƶ����ٲ���ȷ���������������ԣ��ܰѵ⻯���������ɵ��ʵ⣬������������ɫ������ȷ����������ɵ�������������������Ҫ�ָ������ǰ��״̬��Ӧ����ͨ���Ȼ������壬�۲���ȷ��������2.24L���ڱ�״������0.1mol������ת�Ƶ�����0.2mol������ȷ����ѡ�ڢܡ�
Cl2����H2����2NaOH���������������������ƶ����ٲ���ȷ���������������ԣ��ܰѵ⻯���������ɵ��ʵ⣬������������ɫ������ȷ����������ɵ�������������������Ҫ�ָ������ǰ��״̬��Ӧ����ͨ���Ȼ������壬�۲���ȷ��������2.24L���ڱ�״������0.1mol������ת�Ƶ�����0.2mol������ȷ����ѡ�ڢܡ�

 ÿ��10���ӿ�����������������ϵ�д�
ÿ��10���ӿ�����������������ϵ�д�