��Ŀ����
11��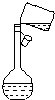 ��1��ʵ������NaNO3��������500mL 0.40mol/L NaNO3��Һ��
��1��ʵ������NaNO3��������500mL 0.40mol/L NaNO3��Һ����������Һʱ���������²�����
a�����ݣ�b�����㣻c���ܽ⣻d��ҡ�ȣ�e��ת�ƣ�f��ϴ�ӣ�j��������
��ȡNaNO3�����������17.0 g�����ղ���˳��4����e������ţ���
��ijͬѧת����Һ�IJ�����ͼ��ʾ��ͼ���������������ձ���500mL����ƿ����ͬѧ�����еĴ�����δ�ò�����������
��2��������������Ϊ98%��Ũ�������Ƴ�1000mL 0��lmol/L��������Һ����������ʱ������������ȷ��ֻ��������ijһ������ᵼ����ҺŨ��ƫ�ߵIJ����Ǣۢݢޣ�
������ƿ������������ˮ
���ձ��Ͳ�����û��ϴ��
��ϡ��Ũ����ʱ��û����ȴ������ת�Ƶ�����ƿ��
�����Ƶ���Һװ��ྻ�ĵ�����������ˮ���Լ�ƿ��
��������ʱ���ӣ�������Һ�����ʵ���Ũ��
������Ͳ��ȡŨ����ʱ�����ӿ̶��ߣ�
���� ��1��������m=CVM������Ҫ���ʵ�������
��������һ�����ʵ���Ũ����Һһ�㲽�裺���㡢�������ܽ⡢��Һ��ϴ�ӡ����ݡ�ҡ�Ƚ��
������ͼ��������״�ж��������ƣ���Һ����Ӧ�ò�����������
��2���������������ʵ����ʵ���n����Һ���V��Ӱ�죬����C=$\frac{n}{V}$���з���������ʹnƫ����ʹVƫС�IJ���������ʹ��ҺŨ��ƫ�ߣ���֮����ҺŨ��ƫ�ͣ�
��� �⣺��1��������500mL 0.40mol/L NaNO3��Һ����Ҫ�����Ƶ�����m=0.40mol/L��0.5L��85g/mol=17.0g��
����һ�����ʵ���Ũ����Һһ�㲽�裺���㡢�������ܽ⡢��Һ��ϴ�ӡ����ݡ�ҡ�ȣ����Ե�4����ת�ƣ�
�ʴ�Ϊ��17.0��e��
��ͼ������Ϊ500mL����ƿ����Һ����Ӧ�ò�����������
�ʴ�Ϊ��500mL����ƿ��δ�ò�����������
��2��������ƿ������������ˮ�������ʵ����ʵ�������Һ����������Ӱ�죬��ҺŨ�Ȳ��䣬�ʲ�ѡ��
���ձ��Ͳ�����û��ϴ�ӣ��������ʲ�����ģ����ʵ����ʵ���ƫС����ҺŨ��ƫ�ͣ��ʲ�ѡ��
��ϡ��Ũ����ʱ��û����ȴ������ת�Ƶ�����ƿ�У���ȴ����Һ���ƫС����ҺŨ��ƫ�ߣ���ѡ��
�����Ƶ���Һװ��ྻ�ĵ�����������ˮ���Լ�ƿ�У���Һ��ϡ�ͣ�Ũ��ƫ�ͣ��ʲ�ѡ��
��������ʱ���ӣ�������Һ�����ʵ���Ũ�ȣ�������Һ���ƫС����ҺŨ��ƫ�ߣ���ѡ��
������Ͳ��ȡŨ����ʱ�����ӿ̶��ߣ�������Һ���ƫ����ҺŨ��ƫ�ߣ���ѡ��
��ѡ���ۢݢޣ�
���� ���⿼����һ�����ʵ���Ũ����Һ�����ƣ���ȷ����ԭ�������Ʋ��������ǽ���ؼ���ע������ƿ��ʹ�÷�����ע���������ķ����ͼ��ɣ���Ŀ�ѶȲ���

| A�� | �ܵ�һ�ֺ��� | B�� | ������Ϊ27 | C�� | ���������Ϊ27 | D�� | ������Ϊ60 |
 | ��A | ��A | ��A | ��A | ��A | ��A | ����A | 0 |
| 2 | C | N | F | Ne | ||||
| 3 | Na | Mg | Al | Si | S | Cl |
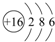
��2��S ԭ�ӽṹʾ��ͼΪ
 ��
����3��Mg��Al�У�ԭ�Ӱ뾶��С����Al��
��4��N���⻯��ķ���ʽNH3����������NԪ����HԪ�ص�������Ϊ14��3�������ԭ��������N-14 H-1������ˮ��Һ�ʼ����ԣ�
��5��Ԫ�ع�������ﳣ����������ά����һ�ָ����ܵ��ִ�ͨѶ���ϵ����ƣ���
��6��Ԫ������������Ӧ��ˮ�����У�������ǿ����NaOH���ѧʽ���������Ե���Al��OH��3���ѧʽ�������߷�Ӧ�Ļ�ѧ����ʽNaOH+Al��OH��3=NaAlO2+2H2O��
| A�� | Fe2O3��Al2O3�������� | B�� | Cl2��HCl����NaOH��Һ | ||
| C�� | Mg��Al���ô�����Һ | D�� | SO2��HCl���ñ��͵�NaHSO3��Һ |
| ʵ����� | �� | �� | �� |
| �Ͻ�����/mg | 255 | 385 | 459 |
| �����������/mL | 280 | 336 | 336 |
��2��Ҫ�������������ʵ���Ũ�ȣ����п���Ϊ�������ݵ��������һ���������������ʵ���Ũ��Ϊ1mol/L��
��3����Ͻ���þ���������ʵ���֮�ȣ����п���Ϊ�������ݵ������Ǽף���õ�þ���������ʵ���֮����1��1��
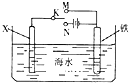
| A�� | ��XΪ̼��������K����N�������Լӿ����ĸ�ʴ | |
| B�� | ��XΪ̼��������K����N������X��������Һ���ɫ | |
| C�� | ��XΪп������K����M�����������缫��Ϊ�������������������� | |
| D�� | ��XΪп������K����M������X�缫�ķ�ӦʽΪ��O2+4e+2H2O��4OH- |
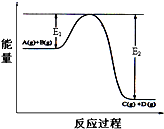 ��ӦA��g��+B��g��?C��g��+D��g�������е������仯��ͼ��ʾ���ش��������⣮
��ӦA��g��+B��g��?C��g��+D��g�������е������仯��ͼ��ʾ���ش��������⣮ ��
��