��Ŀ����
A��B��C��D��E��F��G��H��Ϊ�л������

������ͼ�ش����⣺
��1��D�Ļ�ѧ������ ��
��2��D��һ����������O2��Ӧ����H��H�ɷ���������Ӧ��H����������Ӧ�Ļ�ѧ����ʽ ��
��3��B�ķ���ʽ�� ��A�Ľṹ��ʽ�� ����Ӧ�ٵķ�Ӧ������ ��
��4����������3��������B��ͬ���칹�����Ŀ�� ����
�ٺ����ڶ�ȡ�������ṹ ����B����ͬ������ �۲���FeCl3��Һ������ɫ��Ӧд������һ��ͬ���칹��Ľṹ��ʽ ��
��5��G����Ҫ�Ĺ�ҵԭ�ϣ��û�ѧ����ʽ��ʾG��һ����Ҫ�Ĺ�ҵ��; ��

������ͼ�ش����⣺
��1��D�Ļ�ѧ������
��2��D��һ����������O2��Ӧ����H��H�ɷ���������Ӧ��H����������Ӧ�Ļ�ѧ����ʽ
��3��B�ķ���ʽ��
��4����������3��������B��ͬ���칹�����Ŀ��
�ٺ����ڶ�ȡ�������ṹ ����B����ͬ������ �۲���FeCl3��Һ������ɫ��Ӧд������һ��ͬ���칹��Ľṹ��ʽ
��5��G����Ҫ�Ĺ�ҵԭ�ϣ��û�ѧ����ʽ��ʾG��һ����Ҫ�Ĺ�ҵ��;
���㣺�л�����ƶ�
ר�⣺
������B��Ũ�����������������E����E�Ľṹ��֪��B����������Ӧ����E����BΪ ��D�ķ���ʽΪC2H6O��һ�������¿�������C2H4����DΪCH3CH2OH��GΪCH2=CH2��C��CH3CH2OH��Ũ���ᡢ��������������F�����F�ķ���ʽC4H8O2��֪������F�ķ�ӦΪ������Ӧ����CΪCH3COOH��FΪCH3COOCH2CH3��A����������ˮ��Һ�����������·���ˮ�ⷴӦ���ữ�õ�B��C��D����AΪ
��D�ķ���ʽΪC2H6O��һ�������¿�������C2H4����DΪCH3CH2OH��GΪCH2=CH2��C��CH3CH2OH��Ũ���ᡢ��������������F�����F�ķ���ʽC4H8O2��֪������F�ķ�ӦΪ������Ӧ����CΪCH3COOH��FΪCH3COOCH2CH3��A����������ˮ��Һ�����������·���ˮ�ⷴӦ���ữ�õ�B��C��D����AΪ ���ݴ˽��
���ݴ˽��
 ��D�ķ���ʽΪC2H6O��һ�������¿�������C2H4����DΪCH3CH2OH��GΪCH2=CH2��C��CH3CH2OH��Ũ���ᡢ��������������F�����F�ķ���ʽC4H8O2��֪������F�ķ�ӦΪ������Ӧ����CΪCH3COOH��FΪCH3COOCH2CH3��A����������ˮ��Һ�����������·���ˮ�ⷴӦ���ữ�õ�B��C��D����AΪ
��D�ķ���ʽΪC2H6O��һ�������¿�������C2H4����DΪCH3CH2OH��GΪCH2=CH2��C��CH3CH2OH��Ũ���ᡢ��������������F�����F�ķ���ʽC4H8O2��֪������F�ķ�ӦΪ������Ӧ����CΪCH3COOH��FΪCH3COOCH2CH3��A����������ˮ��Һ�����������·���ˮ�ⷴӦ���ữ�õ�B��C��D����AΪ ���ݴ˽��
���ݴ˽�����
�⣺B��Ũ�����������������E����E�Ľṹ��֪��B����������Ӧ����E����BΪ ��D�ķ���ʽΪC2H6O��һ�������¿�������C2H4����DΪCH3CH2OH��GΪCH2=CH2��C��CH3CH2OH��Ũ���ᡢ��������������F�����F�ķ���ʽC4H8O2��֪������F�ķ�ӦΪ������Ӧ����CΪCH3COOH��FΪCH3COOCH2CH3��A����������ˮ��Һ�����������·���ˮ�ⷴӦ���ữ�õ�B��C��D����AΪ
��D�ķ���ʽΪC2H6O��һ�������¿�������C2H4����DΪCH3CH2OH��GΪCH2=CH2��C��CH3CH2OH��Ũ���ᡢ��������������F�����F�ķ���ʽC4H8O2��֪������F�ķ�ӦΪ������Ӧ����CΪCH3COOH��FΪCH3COOCH2CH3��A����������ˮ��Һ�����������·���ˮ�ⷴӦ���ữ�õ�B��C��D����AΪ ��
��
��1��������������֪��DΪCH3CH2OH���������Ҵ����ʴ�Ϊ���Ҵ���
��2��D��һ����������O2��Ӧ����HΪCH3CHO��CH3CHO����������Ӧ�Ļ�ѧ����ʽΪ ��
��
�ʴ�Ϊ�� ��
��
��3����������ķ�����֪��BΪ ��B�ķ���ʽ��C9H10O3��A�Ľṹ��ʽ��
��B�ķ���ʽ��C9H10O3��A�Ľṹ��ʽ�� ����Ӧ�ٵķ�Ӧ������ˮ�⣨ȡ������Ӧ��
����Ӧ�ٵķ�Ӧ������ˮ�⣨ȡ������Ӧ��
�ʴ�Ϊ��C9H10O3�� ��ˮ�⣨ȡ������Ӧ��
��ˮ�⣨ȡ������Ӧ��
��4��BΪ �����������ٺ����ڶ�ȡ�������ṹ ����B����ͬ�����ţ��������Ȼ����ǻ����۲���FeCl3��Һ������ɫ��Ӧ��˵���������ǻ��������������B��ͬ���칹��Ϊ
�����������ٺ����ڶ�ȡ�������ṹ ����B����ͬ�����ţ��������Ȼ����ǻ����۲���FeCl3��Һ������ɫ��Ӧ��˵���������ǻ��������������B��ͬ���칹��Ϊ �������֣�
�������֣�
�ʴ�Ϊ��3�� ������һ�֣���
������һ�֣���
��5��GΪCH2=CH2��G����Ҫ�Ĺ�ҵԭ�ϣ����Է����Ӿ۷�Ӧ�ƾ���ϩ����ˮ�ӳ����Ҵ�����Ӧ����ʽΪ ��
��
�ʴ�Ϊ�� ��
��
 ��D�ķ���ʽΪC2H6O��һ�������¿�������C2H4����DΪCH3CH2OH��GΪCH2=CH2��C��CH3CH2OH��Ũ���ᡢ��������������F�����F�ķ���ʽC4H8O2��֪������F�ķ�ӦΪ������Ӧ����CΪCH3COOH��FΪCH3COOCH2CH3��A����������ˮ��Һ�����������·���ˮ�ⷴӦ���ữ�õ�B��C��D����AΪ
��D�ķ���ʽΪC2H6O��һ�������¿�������C2H4����DΪCH3CH2OH��GΪCH2=CH2��C��CH3CH2OH��Ũ���ᡢ��������������F�����F�ķ���ʽC4H8O2��֪������F�ķ�ӦΪ������Ӧ����CΪCH3COOH��FΪCH3COOCH2CH3��A����������ˮ��Һ�����������·���ˮ�ⷴӦ���ữ�õ�B��C��D����AΪ ��
����1��������������֪��DΪCH3CH2OH���������Ҵ����ʴ�Ϊ���Ҵ���
��2��D��һ����������O2��Ӧ����HΪCH3CHO��CH3CHO����������Ӧ�Ļ�ѧ����ʽΪ
 ��
���ʴ�Ϊ��
 ��
����3����������ķ�����֪��BΪ
 ��B�ķ���ʽ��C9H10O3��A�Ľṹ��ʽ��
��B�ķ���ʽ��C9H10O3��A�Ľṹ��ʽ�� ����Ӧ�ٵķ�Ӧ������ˮ�⣨ȡ������Ӧ��
����Ӧ�ٵķ�Ӧ������ˮ�⣨ȡ������Ӧ���ʴ�Ϊ��C9H10O3��
 ��ˮ�⣨ȡ������Ӧ��
��ˮ�⣨ȡ������Ӧ����4��BΪ
 �����������ٺ����ڶ�ȡ�������ṹ ����B����ͬ�����ţ��������Ȼ����ǻ����۲���FeCl3��Һ������ɫ��Ӧ��˵���������ǻ��������������B��ͬ���칹��Ϊ
�����������ٺ����ڶ�ȡ�������ṹ ����B����ͬ�����ţ��������Ȼ����ǻ����۲���FeCl3��Һ������ɫ��Ӧ��˵���������ǻ��������������B��ͬ���칹��Ϊ �������֣�
�������֣��ʴ�Ϊ��3��
 ������һ�֣���
������һ�֣�����5��GΪCH2=CH2��G����Ҫ�Ĺ�ҵԭ�ϣ����Է����Ӿ۷�Ӧ�ƾ���ϩ����ˮ�ӳ����Ҵ�����Ӧ����ʽΪ
 ��
���ʴ�Ϊ��
 ��
��
���������⿼���л�����ƶϣ�ע�����ת����ϵ��E�Ľṹ�Լ�D��G�ķ���ʽ�����ƶϣ����չ����ŵ�������ת���ǹؼ����Ѷ��еȣ�

��ϰ��ϵ�д�
 �̲�ȫ���ִʾ�ƪϵ�д�
�̲�ȫ���ִʾ�ƪϵ�д�
�����Ŀ
����˵����ȷ���ǣ�������
| A����������ͨ�����������γ����Ӽ� |
| B��HF��HCl��HBr��HI�����ȶ��Ժͻ�ԭ�Ծ����μ��� |
| C���������ڷǽ���Ԫ�غ���������Դ�����������ǿ |
| D��Ԫ����������Ԫ��ԭ�Ӻ�������Ų������Ա仯�Ľ�� |
 25��ʱ��c��CH3COOH��+c��CH3COO-��=0.1mol/L�Ĵ��ᡢ�����ƻ����Һ�У�c��CH3COOH����c��CH3COO-����pH�Ĺ�ϵ��ͼ��ʾ�������йظ���Һ����������ȷ���ǣ�������
25��ʱ��c��CH3COOH��+c��CH3COO-��=0.1mol/L�Ĵ��ᡢ�����ƻ����Һ�У�c��CH3COOH����c��CH3COO-����pH�Ĺ�ϵ��ͼ��ʾ�������йظ���Һ����������ȷ���ǣ�������| A��pH=5����Һ�У�c��CH3COOH����c��CH3COO-����c��H+����c��OH-�� |
| B����Һ�У�c��H+��+c��Na+��=c��CH3COO-��+c��OH-�� |
| C����W��������25��ʱCH3COOH�ĵ��볣�� |
| D��pH=4����Һ�У�c��H+��+c��Na+��+c��CH3COOH��-c��OH-��=0.1 mol/L |
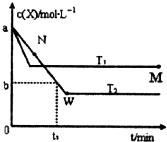 �ں����ܱ�������ͨ��һ������X������Y���岢������Ӧ��2X��g��+Y��g��?2Z��g����������¶�T1��T2��X�����ʵ���Ũ��c��x����ʱ��t�仯��������ͼ��ʾ������������ȷ���ǣ�������
�ں����ܱ�������ͨ��һ������X������Y���岢������Ӧ��2X��g��+Y��g��?2Z��g����������¶�T1��T2��X�����ʵ���Ũ��c��x����ʱ��t�仯��������ͼ��ʾ������������ȷ���ǣ�������| A���÷�Ӧ���е�M��ų����������ڽ��е�W��ų������� | ||
B��T2�£���0?t1ʱ���ڣ��ԣ�Y��=
| ||
| C��M���ƽ�ⳣ����W���ƽ�ⳣ��С | ||
| D��M��ʱ�ټ���һ������X��ƽ���X��ת�������� |
����˵������ȷ���ǣ�������
| A�������ʵ���Ũ�ȵ� NH4HSO4��Һ��NaOH��Һ�������ϣ���Һ�и�����Ũ�ȴ�С��ϵΪ��c��Na+��=c��SO42-����c��NH4+����c��H+����c��OH-�� | ||
B��25��ʱ����a mo1?L-l��ˮ��0.01 moI?L-1����������ϣ���Ӧ��ȫʱ��Һ��c��NH4+��=c��C1-�����ú�a�Ĵ���ʽ��ʾNH3?H2O�ĵ��볣��Kb=
| ||
C�� ij�¶��£���ͬ�������ͬpH������ʹ�����Һ�ֱ��ˮϡ�ͣ�pH����Һ���V�仯��������ͼ��ʾ����Ϊ����ϡ��ʱpH�ı仯���ߣ���a��b����ˮ�ĵ���̶ȣ�a��b ij�¶��£���ͬ�������ͬpH������ʹ�����Һ�ֱ��ˮϡ�ͣ�pH����Һ���V�仯��������ͼ��ʾ����Ϊ����ϡ��ʱpH�ı仯���ߣ���a��b����ˮ�ĵ���̶ȣ�a��b | ||
| D����֪298Kʱ�����ᣨHCN����Ka=4.9��10-10��̼���Ka1=4.4��10-7��Ka2=4.7��10-11���ݴ˿��Ʋ⽫��������뵽̼������Һ���ܹ۲쵽�����ݲ��� |
���ֶ�����Ԫ�������ڱ��е�λ����ͼ������ֻ��MΪ����Ԫ�أ�����˵����ȷ���ǣ�������

| A��ԭ�Ӱ뾶Z��Y |
| B��X�������̬�⻯������ȶ��Ա�Z��ǿ |
| C��M��Z�γɵĻ�������ֻ�������Ӽ� |
| D��M������������Ӧˮ���ﲻ����NaOH��Һ��Ӧ |


