��Ŀ����
11����NaOH��Һ���������е�SO2�������õ�Na2SO3��Һ���е�⣬��ѭ������NaOH��ͬʱ�õ�H2SO4����ԭ����ͼ��ʾ�����缫����Ϊʯī��
��1��д���ù�����NaOH��Һ���������е�SO2�ķ���ʽ2NaOH+SO2=Na2SO3+H2O
��2����Ƚ�Na2SO3��Һ�и�����Ũ�ȵĴ�СC��Na+����C��SO32-����C��OH-����C��HSO3-����C��H+��
��3��ͼ��a��Ҫ���ӵ�Դ�ģ������������������C�����������������ᣮ
��4��SO32-�ŵ�ĵ缫��ӦʽΪSO32--2e-+H2O=SO42-+2H+��
��5��������������������������ǿ����ƽ���ƶ�ԭ������ԭ��H2O?H++OH-��������H+�ŵ�����H2��c��H+����С��ˮ�ĵ���ƽ�������ƶ���������ǿ��
���� ��1��������NaOH��Һ���������е�SO2���������������ƺ�ˮ��
��2��SO32-ˮ������ˮ�⣬�����Ӳ�ˮ�⣬ˮ�ĵ���������������ˮ�ⶼ����OH-���ݴ˵ó���
��3�����ݵ������Һ���������ӵ��ƶ������жϵ缫���������������������ж�ͼ��a��Ҫ���ӵ�Դ�ĸ�����SO32-������ʧȥ���ӱ��SO42-������C��������������H2SO4��
��4�������������������ʧȥ���ӷ���������Ӧ������������ӣ�
��5�������������ŵ�����Ϊ�����ӽ��ˮ�ĵ���ƽ��H2O?H++OH-���
��� �⣺��1��������NaOH��Һ���������е�SO2���������������ƺ�ˮ����ѧ����ʽΪ��2NaOH+SO2=Na2SO3+H2O
�ʴ�Ϊ��2NaOH+SO2=Na2SO3+H2O��
��2��Na2SO3ˮ�����ӷ���ʽΪ��SO32-+H2O?HSO3-+OH-�������Ӳ�ˮ�⣬����c��Na+�����ˮ�ĵ���������������ˮ�ⶼ����OH-������c��OH-����c��HSO3-����c��H+�� ��С�����Ը���Һ������Ũ�ȴ�С˳����c��Na+����c��SO32-����c��OH-����c��HSO3-����c��H+����
�ʴ�Ϊ��c��Na+����c��SO32-����c��OH-����c��HSO3-����c��H+����
��3��ͼ�п�֪������������a�������������������b��������b��Ϊ������Ӧ�ýӵ�Դ��������SO32-������ʧȥ���ӱ��SO42-������C��������������H2SO4��
�ʴ�Ϊ���������
��4�������������������ʧȥ���ӷ���������Ӧ������������ӣ��缫��ӦʽΪ��SO32--2e-+H2O=SO42-+2H+��
�ʴ�Ϊ��SO32--2e-+H2O=SO42-+2H+��
��5��������H+�ŵ�����H2��c��H+����С��ˮ�ĵ���ƽ��H2O?H++OH-�����ƶ������Լ�����ǿ��
�ʴ�Ϊ��ˮΪ������ʴ���H2O?H++OH-��������H+�ŵ�����H2��C��H+����С��ˮ�ĵ���ƽ���������ƶ����������ӣ�
���� ������Ҫ�����˵���ԭ������ȷ���ع���ԭ���ǽ���ؼ���ע�����ʵ缫�жϵķ����͵缫��Ӧʽ��дע������Ϊ�״��㣮

| A�� | ԭ�Ӱ뾶��С˳��D��C��B��A | |
| B�� | y��p �о�ֻ�������Ӽ� | |
| C�� | B��D��Ԫ������������Ӧˮ����ֱ�Ϊǿ�ᡢǿ�� | |
| D�� | p������ˮ�ĵ����q�ܴٽ�ˮ�ĵ��� |
| A�� | ��=��=��=�� | B�� | �ۣ��ڣ��٣��� | C�� | �ܣ��ڣ��٣��� | D�� | �ۣ��٣��ڣ��� |
| A�� | Ԫ��Y��Z��W������ͬ���Ӳ�ṹ�����ӣ���뾶�������� | |
| B�� | Ԫ��Y����Ԫ��Z�γɻ�����Z2Y2���û������ڲ������ֻ�ѧ�� | |
| C�� | Ԫ��Y��R�ֱ���Ԫ��X�γɵĻ���������ȶ��ԣ�XmY��XmR���е�XmY��XmR | |
| D�� | Ԫ��W��R������������ˮ���ﶼ��ǿ�� |
| A�� | ������̼�Ľṹʽ��O-C-O | B�� | �ǻ��ĵ���ʽΪ��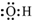 | ||
| C�� | ������ӵ����ģ�ͣ� | D�� | �����ӵĽṹʾ��ͼ��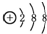 |
| ѡ�� | ������ | ������ |
| A | SO2��Ư���� | SO2��ʹ��ˮ��ɫ |
| B | NH3���м��� | NH3��������ˮ |
| C | Ũ��������ˮ�� | ��Ũ�������SO2 |
| D | HNO3�������� | ��������ϡHNO3��ȡ���� |
| A�� | A | B�� | B | C�� | C | D�� | D |
 ̽����������������������ǿ��������ͭ��Ӧ�Ļ�ԭ��������ʣ�
̽����������������������ǿ��������ͭ��Ӧ�Ļ�ԭ��������ʣ�
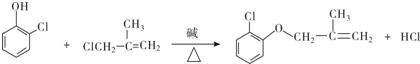 ���л����ýṹ��ʽ��ʾ����ͬ����
���л����ýṹ��ʽ��ʾ����ͬ����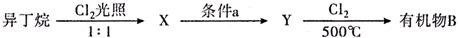
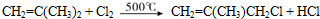 ��
��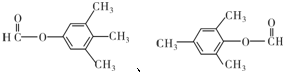
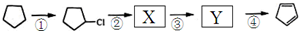

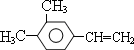 ����дһ�֣����������������ۻ����ϩ�Ĺ�ϵ��B��������ĸ��
����дһ�֣����������������ۻ����ϩ�Ĺ�ϵ��B��������ĸ��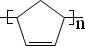 ����
���� ����д�����з�Ӧ���л�����Ľṹ��ʽ��
����д�����з�Ӧ���л�����Ľṹ��ʽ�� OHC-CHO��OHC-CH2-CHO��
OHC-CHO��OHC-CH2-CHO��