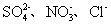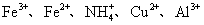��Ŀ����
ij100mL��Һ�н����±������е�5�����ӣ�������ˮ�ĵ��뼰���ӵ�ˮ�⣩���Ҹ����ӵ����ʵ�����Ϊ0.1mol��
|
������ |
|
|
������ |
|
������ԭ��Һ�м���KSCN��Һ�������Ա仯��
������ԭ��Һ�м�����������ᣬ���������ɣ���Һ������������䡣
������ԭ��Һ�м���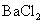 ��Һ���а�ɫ�������ɡ�
��Һ���а�ɫ�������ɡ�
�Իش��������⣺
��1��ԭ��Һ�������������ǣ�д���ӷ��ţ���ͬ�� ���������� ��
��2������ԭ��Һ���ȼ����������ᣬ�ٵ���KSCN��Һ��ʵ������������Ӧ�����ӷ���ʽ�� �� ��
��3������ԭ��Һ�м�������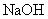 ��Һ����ַ�Ӧ����һ��ʱ�䣬���ˡ�ϴ�ӡ����գ��������ù����������
��
��Һ����ַ�Ӧ����һ��ʱ�䣬���ˡ�ϴ�ӡ����գ��������ù����������
��
��4����ԭ��Һ�м����������������ˮ���ռ������������岢ʹ����ǡ�ó����������Խ�����������ˮ���У�����������ͨ�� mL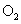 �����������ָ��״��������ʹ��Һ������������
�����������ָ��״��������ʹ��Һ������������
���𰸡�
��1��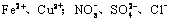 [��Դ:ѧ*��*��Z*X*X*K]
[��Դ:ѧ*��*��Z*X*X*K]
��2��
��3��16g
��4��560

��ϰ��ϵ�д�
 �ľ�ͼ���ʱ�ȷ�ϵ�д�
�ľ�ͼ���ʱ�ȷ�ϵ�д�
�����Ŀ
ij100mL��Һ�н����±������е�5�֣�������ˮ�ĵ��뼰���ӵ�ˮ�⣩���Ҹ����ӵ����ʵ�����Ϊ0.1mol��
������ԭ��Һ�м���KSCN��Һ�������Ա仯��
������ԭ��Һ�м�����������ᣬ���������ɣ���Һ������������䣮
������ԭ��Һ�м���BaCl2��Һ���а�ɫ�������ɣ�
�Իش��������⣺
��1��ԭ��Һ�������������ǣ�д���ӷ��ţ���ͬ��______����������______��
��2������ԭ��Һ���ȼ����������ᣬ�ٵ���KSCN��Һ��ʵ������������Ӧ�����ӷ���ʽ��______��______��
��3������ԭ��Һ�м�������NaOH��Һ����ַ�Ӧ����һ��ʱ�䣬���ˡ�ϴ�ӡ����գ��������ù����������______��
��4����ԭ��Һ�м����������������ˮ���ռ������������岢ʹ����ǡ�ó����������Խ�����������ˮ���У�����������ͨ��______ mL O2�����������ָ��״��������ʹ��Һ������������
| ������ | SO42-��NO3-��Cl- |
| ������ | Fe3+��Fe2+��NH4+��Cu2+��Al3+ |
������ԭ��Һ�м�����������ᣬ���������ɣ���Һ������������䣮
������ԭ��Һ�м���BaCl2��Һ���а�ɫ�������ɣ�
�Իش��������⣺
��1��ԭ��Һ�������������ǣ�д���ӷ��ţ���ͬ��______����������______��
��2������ԭ��Һ���ȼ����������ᣬ�ٵ���KSCN��Һ��ʵ������������Ӧ�����ӷ���ʽ��______��______��
��3������ԭ��Һ�м�������NaOH��Һ����ַ�Ӧ����һ��ʱ�䣬���ˡ�ϴ�ӡ����գ��������ù����������______��
��4����ԭ��Һ�м����������������ˮ���ռ������������岢ʹ����ǡ�ó����������Խ�����������ˮ���У�����������ͨ��______ mL O2�����������ָ��״��������ʹ��Һ������������
ij100mL��Һ�н����±������е�5�֣�������ˮ�ĵ��뼰���ӵ�ˮ�⣩���Ҹ����ӵ����ʵ�����Ϊ0.1mol��
������ԭ��Һ�м���KSCN��Һ�������Ա仯��
������ԭ��Һ�м�����������ᣬ���������ɣ���Һ������������䣮
������ԭ��Һ�м���BaCl2��Һ���а�ɫ�������ɣ�
�Իش��������⣺
��1��ԭ��Һ�������������ǣ�д���ӷ��ţ���ͬ��______����������______��
��2������ԭ��Һ���ȼ����������ᣬ�ٵ���KSCN��Һ��ʵ������������Ӧ�����ӷ���ʽ��______��______��
��3������ԭ��Һ�м�������NaOH��Һ����ַ�Ӧ����һ��ʱ�䣬���ˡ�ϴ�ӡ����գ��������ù����������______��
��4����ԭ��Һ�м����������������ˮ���ռ������������岢ʹ����ǡ�ó����������Խ�����������ˮ���У�����������ͨ��______ mL O2�����������ָ��״��������ʹ��Һ������������
| ������ | SO42-��NO3-��Cl- |
| ������ | Fe3+��Fe2+��NH4+��Cu2+��Al3+ |
������ԭ��Һ�м�����������ᣬ���������ɣ���Һ������������䣮
������ԭ��Һ�м���BaCl2��Һ���а�ɫ�������ɣ�
�Իش��������⣺
��1��ԭ��Һ�������������ǣ�д���ӷ��ţ���ͬ��______����������______��
��2������ԭ��Һ���ȼ����������ᣬ�ٵ���KSCN��Һ��ʵ������������Ӧ�����ӷ���ʽ��______��______��
��3������ԭ��Һ�м�������NaOH��Һ����ַ�Ӧ����һ��ʱ�䣬���ˡ�ϴ�ӡ����գ��������ù����������______��
��4����ԭ��Һ�м����������������ˮ���ռ������������岢ʹ����ǡ�ó����������Խ�����������ˮ���У�����������ͨ��______ mL O2�����������ָ��״��������ʹ��Һ������������
ij100mL��Һ�н����±��е�5�����ӣ�������ˮ�ĵ��뼰���ӵ�ˮ�⣩���Ҹ����ӵ����ʵ�����Ϊ0.1mol��
��������Һ�м���KSCN��Һ�������Ա仯
��������Һ�м�����������ᣬ���������ɣ���Һ�������������
��������Һ�м���BaCl2��Һ���а�ɫ�������ɣ�
�Իش��������⣺
��1��ԭ��Һ�������������ǣ�д���ӷ��ţ���ͬ��______����������______��
��2������ԭ��Һ�м�������NaOH��Һ����ַ�Ӧ����һ��ʱ�䣬���ˡ�ϴ�ӡ����գ��������ù����������______��
��3����ԭ��Һ�м����������������Ӧ�����ӷ���ʽ��______��
��4������ˮ���ռ������������岢ʹ����ǡ�ó����������Խ�����������ˮ���У�����������ͨ��______mLO2�����������ָ��״��������ʹ��Һ��������������ʱ��������Һ�����ʵ���Ũ��Ϊ______mol/L��
| ������ | SO42-��NO3-��Cl- |
| ������ | Fe3+��Fe2+��NH4+��Cu2+��Al3+ |
��������Һ�м�����������ᣬ���������ɣ���Һ�������������
��������Һ�м���BaCl2��Һ���а�ɫ�������ɣ�
�Իش��������⣺
��1��ԭ��Һ�������������ǣ�д���ӷ��ţ���ͬ��______����������______��
��2������ԭ��Һ�м�������NaOH��Һ����ַ�Ӧ����һ��ʱ�䣬���ˡ�ϴ�ӡ����գ��������ù����������______��
��3����ԭ��Һ�м����������������Ӧ�����ӷ���ʽ��______��
��4������ˮ���ռ������������岢ʹ����ǡ�ó����������Խ�����������ˮ���У�����������ͨ��______mLO2�����������ָ��״��������ʹ��Һ��������������ʱ��������Һ�����ʵ���Ũ��Ϊ______mol/L��