��Ŀ����
������;�㷺��ij��ѧ��ѧ��ȤС��������Ʊ�����������һϵ�����ʵ���װ�ã��г��豸���ԣ���
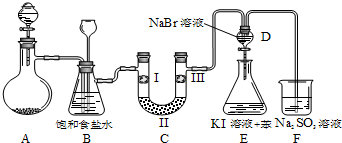
��1���Ʊ�����ѡ��ҩƷΪ��Ư�۹����Ũ���ᣬ��صĻ�ѧ��Ӧ����ʽΪ�� ��
��2����ϸ�۲�װ��B����װ��B������Ϊ ��
��3��װ��C��ʵ��Ŀ������֤�����Ƿ����Ư���ԣ�Ϊ��C�Т����η��룺 ����ѡ����ĸ��ţ�
��4�����װ��D��E��Ŀ���DZȽ��ȡ��塢��ķǽ����ԣ�����D�л���ͨ����������ʱ�����Կ�����ɫ��Һ��Ϊ����ɫ��˵���ȵķǽ����Դ����壮��������װ��D��������Һ����װ��E�У����۲쵽�������� ������������˵����ķǽ�����ǿ�ڵ⣬ԭ���� ��
��5����������������ˮ�������������������������ClO2���ȶ�������NaOH��Һ��H2O2��Ӧ��ת��Ϊ�Ƚ��ȶ����������ƣ��÷�Ӧ�Ļ�ѧ����ʽΪ ��
��ClO2����������ˮ�л����ClO2������FeCl2���仹ԭ������V L���ѻ���Ϊ��״����ClO2����һ��������ˮ���ټ���a mol FeCl2�ɽ�������ClO2ǡ�ó�ȥ���������ˮ�в���ClO2�����ʵ�����Ϊ g��
��6��ijͬѧ�Լ��Ʊ���Ư�ۣ������Ʊ���������ˮ���ټ����̪���Ժ�ɫ��һ���ֱ����ɫ����ͬѧ�ݴ��жϣ��Լ��Ʊ���Ư�۳ɷ�ΪCa��OH��2��Ca��ClO��2����ȤС����Ϊ��ͬѧ���жϲ���ȷ��ԭ���� ��

��1���Ʊ�����ѡ��ҩƷΪ��Ư�۹����Ũ���ᣬ��صĻ�ѧ��Ӧ����ʽΪ��
��2����ϸ�۲�װ��B����װ��B������Ϊ
��3��װ��C��ʵ��Ŀ������֤�����Ƿ����Ư���ԣ�Ϊ��C�Т����η��룺
| a | b | c | d | |
| �� | �������ɫ���� | �������ɫ���� | ʪ�����ɫ���� | ʪ�����ɫ���� |
| �� | ��ʯ�� | �轺 | Ũ���� | ��ˮ�Ȼ��� |
| �� | ʪ�����ɫ���� | ʪ�����ɫ���� | �������ɫ���� | �������ɫ���� |
��5����������������ˮ�������������������������ClO2���ȶ�������NaOH��Һ��H2O2��Ӧ��ת��Ϊ�Ƚ��ȶ����������ƣ��÷�Ӧ�Ļ�ѧ����ʽΪ
��ClO2����������ˮ�л����ClO2������FeCl2���仹ԭ������V L���ѻ���Ϊ��״����ClO2����һ��������ˮ���ټ���a mol FeCl2�ɽ�������ClO2ǡ�ó�ȥ���������ˮ�в���ClO2�����ʵ�����Ϊ
��6��ijͬѧ�Լ��Ʊ���Ư�ۣ������Ʊ���������ˮ���ټ����̪���Ժ�ɫ��һ���ֱ����ɫ����ͬѧ�ݴ��жϣ��Լ��Ʊ���Ư�۳ɷ�ΪCa��OH��2��Ca��ClO��2����ȤС����Ϊ��ͬѧ���жϲ���ȷ��ԭ����
���㣺������ʵ�����Ʒ�
ר�⣺ʵ�������
��������1��������ƾ���ǿ�������ԣ��ܹ��������ᣬ���ݻ��ϼ�������Ŀ��ͬ��ƽ����ʽ��
��2����Ӧ�������к��������Ȼ��⡢ˮ������װ��B���ǰ�ȫƿ�����ʵ�����ʱC���Ƿ�����������������ʱB�еģ�ѹǿ����B�г���©����Һ���������γ�ˮ����
��3��Ϊ����֤�����Ƿ����Ư���ԣ�Ҫ��֤����������Ư���ԣ�ʪ�����ɫ�����У�������ˮ��Ӧ���ɴ��������Ư���ԣ�
��4������������ǿ���壬�ܹ��û��壬��������ǿ�ڵ⣬�ܹ��û��⣬������������ǿ�ڵ⣬�ܹ��û��⣬��ϵ����л������ܽ��Լ���ɫ���
��5��ClO2��NaOH��Һ��H2O2��Ӧ��ת��Ϊ�Ƚ��ȶ����������ƣ�NaClO2�������У�Cl�Ļ��ϼ۴�+4�۽�Ϊ+3�ۣ�����˫��ˮ�ǻ�ԭ���������к�����������Ԫ�صĻ��ϼ۴�-1�۽�Ϊ-2�ۣ����ݵ�ʧ�����غ���ƽ����ʽ��
����Ԫ�ػ��ϼ۱仯��ϵ�ʧ�����غ������㣻
��6�����Ǵ�����Ƶ�ˮ�ⷴӦ��ˮ���������ʣ�
��2����Ӧ�������к��������Ȼ��⡢ˮ������װ��B���ǰ�ȫƿ�����ʵ�����ʱC���Ƿ�����������������ʱB�еģ�ѹǿ����B�г���©����Һ���������γ�ˮ����
��3��Ϊ����֤�����Ƿ����Ư���ԣ�Ҫ��֤����������Ư���ԣ�ʪ�����ɫ�����У�������ˮ��Ӧ���ɴ��������Ư���ԣ�
��4������������ǿ���壬�ܹ��û��壬��������ǿ�ڵ⣬�ܹ��û��⣬������������ǿ�ڵ⣬�ܹ��û��⣬��ϵ����л������ܽ��Լ���ɫ���
��5��ClO2��NaOH��Һ��H2O2��Ӧ��ת��Ϊ�Ƚ��ȶ����������ƣ�NaClO2�������У�Cl�Ļ��ϼ۴�+4�۽�Ϊ+3�ۣ�����˫��ˮ�ǻ�ԭ���������к�����������Ԫ�صĻ��ϼ۴�-1�۽�Ϊ-2�ۣ����ݵ�ʧ�����غ���ƽ����ʽ��
����Ԫ�ػ��ϼ۱仯��ϵ�ʧ�����غ������㣻
��6�����Ǵ�����Ƶ�ˮ�ⷴӦ��ˮ���������ʣ�
���
�⣺��1��Ư�۹����Ũ���ᷴӦ�����Ȼ��ơ�������ˮ����ѧ����ʽΪ��Ca��ClO��2+4HCl��Ũ���TCaCl2+2Cl2��+2H2O��
�ʴ�Ϊ��Ca��ClO��2+4HCl��Ũ���TCaCl2+2Cl2��+2H2O��
��2����Ӧ�������Ȼ����ˮ�����������е����ʣ�װ��B�б���ʳ��ˮ�������dz�ȥCl2�е�HCl��װ��B���ǰ�ȫƿ�����ʵ�����ʱC���Ƿ�����������������ʱB�еģ�ѹǿ����B�г���©����Һ���������γ�ˮ����
�ʴ�Ϊ����ȥCl2�е�HCl �Ͱ�ȫƿ��
��3��װ��C��ʵ��Ŀ������֤�����Ƿ����Ư���ԣ���֤�����Ƿ����Ư���ԣ�Ҫ��֤����������Ư���ԣ�ʪ�����ɫ�����У�������ˮ��Ӧ���ɴ��������Ư���ԣ�ѡ����abc�Ģ��ж��Ǹ��������ͨ��ʪ�����ɫ����������֤������Ư���ԣ�����C��I��II��III���η���ʪ�����ɫ��������ˮ�Ȼ��ơ��������ɫ����������ѡd��
�ʴ�Ϊ��d��
��4�������������廯�Ʒ�Ӧ�����嵥�ʣ�Һ���嵥�ʺ͵⻯����Һ�еĵ⻯�ط�Ӧ���ɵⵥ�ʣ��ⵥ�������ڱ������Ϻ�ɫ�����ܶ�С��ˮ��������ˮ����Һ�ֲ㣻
������Cl2Ҳ�ɽ�I-����ΪI2������ͨ��E����Һ��Ϊ���㣬�ϲ㣨���㣩Ϊ�Ϻ�ɫ������˵�����������ǿ�ڵ⣻
�ʴ�Ϊ��E����Һ��Ϊ���㣬�ϲ㣨���㣩Ϊ�Ϻ�ɫ��������Cl2Ҳ�ɽ�I-����ΪI2��
��5��ClO2��NaOH��Һ��H2O2��Ӧ��ת��Ϊ�Ƚ��ȶ����������ƣ�NaClO2�������У�Cl�Ļ��ϼ۴�+4�۽�Ϊ+3�ۣ�����˫��ˮ�ǻ�ԭ���������к�����������Ԫ�صĻ��ϼ۴�-1�۽�Ϊ-2�ۣ����ݵ�ʧ�����غ㡢ԭ�Ӹ����غ�÷���ʽΪ��2ClO2+2NaOH+H2O2=2NaClO2+O2��+2H2O��
����ClO2�ܱ�FeCl2��ԭΪCl-��ClԪ�ػ��ϼ۽�����5�ۣ�FeCl2������ΪFeCl3��FeԪ�ػ��ϼ�������1�ۣ��裺������ˮ�в���ClO2�����ʵ���Ϊx��
���ݵ����غ㣬��a��3-2��=5x�����x=0.2amol��ClO2������m=nM=0.2amol��67.5g/mol=13.5ag��
�ʴ�Ϊ��2ClO2+2NaOH+H2O2=2NaClO2+O2��+2H2O��13.5a��
��6�����Ǵ�����Ƶ�ˮ�ⷴӦ��ˮ���������ʣ������������ǿ�������Σ���Һ��ˮ�ⷴӦ�ʼ��ԣ������̪��ʺ�ɫ��ˮ����һ������������Ư���ԣ�ʹ��Һ�����ɫ��Ư�۳ɷ���Ca��OH��2��Ca��ClO��2��
�ʴ�Ϊ�������������ǿ�������Σ���Һ��ˮ�ⷴӦ�ʼ��ԣ������̪��ʺ�ɫ��ˮ����һ������������Ư���ԣ�ʹ��Һ�����ɫ���ʲ���ȷ������Ca��OH��2��
�ʴ�Ϊ��Ca��ClO��2+4HCl��Ũ���TCaCl2+2Cl2��+2H2O��
��2����Ӧ�������Ȼ����ˮ�����������е����ʣ�װ��B�б���ʳ��ˮ�������dz�ȥCl2�е�HCl��װ��B���ǰ�ȫƿ�����ʵ�����ʱC���Ƿ�����������������ʱB�еģ�ѹǿ����B�г���©����Һ���������γ�ˮ����
�ʴ�Ϊ����ȥCl2�е�HCl �Ͱ�ȫƿ��
��3��װ��C��ʵ��Ŀ������֤�����Ƿ����Ư���ԣ���֤�����Ƿ����Ư���ԣ�Ҫ��֤����������Ư���ԣ�ʪ�����ɫ�����У�������ˮ��Ӧ���ɴ��������Ư���ԣ�ѡ����abc�Ģ��ж��Ǹ��������ͨ��ʪ�����ɫ����������֤������Ư���ԣ�����C��I��II��III���η���ʪ�����ɫ��������ˮ�Ȼ��ơ��������ɫ����������ѡd��
�ʴ�Ϊ��d��
��4�������������廯�Ʒ�Ӧ�����嵥�ʣ�Һ���嵥�ʺ͵⻯����Һ�еĵ⻯�ط�Ӧ���ɵⵥ�ʣ��ⵥ�������ڱ������Ϻ�ɫ�����ܶ�С��ˮ��������ˮ����Һ�ֲ㣻
������Cl2Ҳ�ɽ�I-����ΪI2������ͨ��E����Һ��Ϊ���㣬�ϲ㣨���㣩Ϊ�Ϻ�ɫ������˵�����������ǿ�ڵ⣻
�ʴ�Ϊ��E����Һ��Ϊ���㣬�ϲ㣨���㣩Ϊ�Ϻ�ɫ��������Cl2Ҳ�ɽ�I-����ΪI2��
��5��ClO2��NaOH��Һ��H2O2��Ӧ��ת��Ϊ�Ƚ��ȶ����������ƣ�NaClO2�������У�Cl�Ļ��ϼ۴�+4�۽�Ϊ+3�ۣ�����˫��ˮ�ǻ�ԭ���������к�����������Ԫ�صĻ��ϼ۴�-1�۽�Ϊ-2�ۣ����ݵ�ʧ�����غ㡢ԭ�Ӹ����غ�÷���ʽΪ��2ClO2+2NaOH+H2O2=2NaClO2+O2��+2H2O��
����ClO2�ܱ�FeCl2��ԭΪCl-��ClԪ�ػ��ϼ۽�����5�ۣ�FeCl2������ΪFeCl3��FeԪ�ػ��ϼ�������1�ۣ��裺������ˮ�в���ClO2�����ʵ���Ϊx��
���ݵ����غ㣬��a��3-2��=5x�����x=0.2amol��ClO2������m=nM=0.2amol��67.5g/mol=13.5ag��
�ʴ�Ϊ��2ClO2+2NaOH+H2O2=2NaClO2+O2��+2H2O��13.5a��
��6�����Ǵ�����Ƶ�ˮ�ⷴӦ��ˮ���������ʣ������������ǿ�������Σ���Һ��ˮ�ⷴӦ�ʼ��ԣ������̪��ʺ�ɫ��ˮ����һ������������Ư���ԣ�ʹ��Һ�����ɫ��Ư�۳ɷ���Ca��OH��2��Ca��ClO��2��
�ʴ�Ϊ�������������ǿ�������Σ���Һ��ˮ�ⷴӦ�ʼ��ԣ������̪��ʺ�ɫ��ˮ����һ������������Ư���ԣ�ʹ��Һ�����ɫ���ʲ���ȷ������Ca��OH��2��
���������⿼�����������Ʊ������ʣ���ȷ�Ʊ���ԭ���������������ǽ���ؼ���ע��������ԭ����ʽ����д����ؼ��㣬��Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
���з���ʽ��д����ȷ���ǣ�������
A��Si+O2
| ||||
B��SiO2+2C
| ||||
C��SiO2+CaO
| ||||
| D��SiO2+2Na++2OH-�TNa2SiO3+H2O |
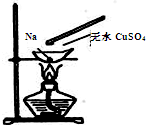 ijѧϰ������������һ��ʵ�飺���������зŻƶ�����С��һ������ƣ��������ۻ�ʱ���ò�����պȡ������ˮCuSO4��ĩ���ۻ����ƽӴ���˲�����ҫ�۵Ļ�ͬʱ�к�ɫ�������ɣ��ݴ��ж�����˵���в���ȷ���ǣ�������
ijѧϰ������������һ��ʵ�飺���������зŻƶ�����С��һ������ƣ��������ۻ�ʱ���ò�����պȡ������ˮCuSO4��ĩ���ۻ����ƽӴ���˲�����ҫ�۵Ļ�ͬʱ�к�ɫ�������ɣ��ݴ��ж�����˵���в���ȷ���ǣ�������| A��������Ӧ���û���Ӧ |
| B��������Ӧ�Ƿ��ȷ�Ӧ |
| C����������ˮ�����£�Na������CuSO4��Ӧ������Cu |
| D������ʵ��֤��CuSO4���л�ԭ�� |
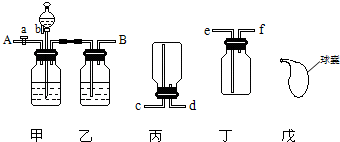 ������ͼװ�ÿ��Դ�CO��CO2�Ļ���������Ⱥ����������CO��CO2���壮
������ͼװ�ÿ��Դ�CO��CO2�Ļ���������Ⱥ����������CO��CO2���壮