��Ŀ����
3��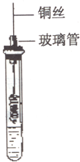 �Ҵ���һ����Ҫ���л�����ԭ�ϣ������������ס������Ϊԭ�Ͼ����͡������Ƴɣ��Ҵ������;��ӹ������γɵij���ȼ�ϼ��Ҵ����ͣ�����й�֪ʶ���ش��������⣺
�Ҵ���һ����Ҫ���л�����ԭ�ϣ������������ס������Ϊԭ�Ͼ����͡������Ƴɣ��Ҵ������;��ӹ������γɵij���ȼ�ϼ��Ҵ����ͣ�����й�֪ʶ���ش��������⣺��1��д���Ҵ�������Ʒ�Ӧ�Ļ�ѧ����ʽ2CH3CH2OH+2Na��2CH3CH2ONa+H2��
��2�����Թ������2mL�Ҵ�����һ���������״��ͭ˿���ھƾ������м��ȣ�ʹͭ˿��������һ�����ɫ������ͭ��������������ʢ���Ҵ����Թ����ͼ����ȡ��ͭ˿�����Թ۲쵽ͭ˿�����ɺ�ɫ������ʵ��ʵ�����Ҵ���������Ӧ����ѧ����ʽΪ��2CH3CH2OH+O2$��_{��}^{����}$2CH3CHO+2H2O ����÷�Ӧ��������Լ��ǣ�������Һ������������ͭ
��3�����е�˵���У���ȷ����BC
A���Ҵ�������һ�ִ�������ڿ�������Դ
B ʹ���Ҵ����ͣ����Խ�ʡʯ����Դ������β�����ŷţ���������
C���Ҵ������ᷴӦ���ɵ�����������һ����ɫ�����ܶȱ�ˮС����״Һ��
D������CH3CH218OH��CH3COOH����������Ӧ�������������в���18O��
���� ��1���Ҵ�������Ʒ�Ӧ�����Ҵ��ƺ�������
��2���Ҵ������ɫ������ͭ��Ӧ�õ���ȩ�ͺ�ɫ��ͭ�Լ�ˮ������ȩ�������ʿ��Ժ�������Һ�Լ����Ƶ�������ͭ����Һ֮�䷴Ӧ��
��3��A���Ҵ������ǽ��Ҵ������ͻ�ϻ��ܺ�����ʣ�
B �Ҵ����͵�ȼ�ղ�����ˮ�Ͷ�����̼��
C������������һ����ɫ�����ܶȱ�ˮС����״Һ�壻
D������CH3CH218OH��CH3COOH����������Ӧ��ԭ���ǣ������ǻ������⣮
��� �⣺��1���Ҵ�������Ʒ�Ӧ�����Ҵ��ƺ�������2CH3CH2OH+2Na��2CH3CH2ONa+H2�����ʴ�Ϊ��2CH3CH2OH+2Na��2CH3CH2ONa+H2����
��2��һ���������״��ͭ˿���ھƾ������м��ȣ�ʹͭ˿��������һ�����ɫ������ͭ��������������ʢ���Ҵ����Թ��������Ӧ��2CH3CH2OH+O2$��_{��}^{����}$2CH3CHO+2H2O������ȩ�������ʿ��Ժ�������Һ�Լ����Ƶ�������ͭ����Һ֮�䷴Ӧ���ֱ�������������Dz���ש��ɫ������
�ʴ�Ϊ����ɺ�ɫ��2CH3CH2OH+O2$��_{��}^{����}$2CH3CHO+2H2O��������Һ������������ͭ��
��3��A���Ҵ������ǽ��Ҵ������ͻ�ϻ��ܺ�����ʣ���һ�ֻ����ʴ���
B �Ҵ����͵�ȼ�ղ�����ˮ�Ͷ�����̼��ʹ���Ҵ����ͣ����Խ�ʡʯ����Դ������β�����ŷţ���������������ȷ��
C���Ҵ������ᷴӦ���ɵ���������������������һ����ɫ�����ܶȱ�ˮС����״Һ�壬����ȷ��
D������CH3CH218OH��CH3COOH����������Ӧ��ԭ���ǣ������ǻ������⣬���������к�18O��
��ѡBC��
���� ���⿼��ѧ���Ҵ��������Լ�Ӧ��֪ʶ��ע��֪ʶ��Ǩ��Ӧ���ǹؼ����Ѷ��еȣ�

 ��Ȥ����¹�֪��ϵ�д�
��Ȥ����¹�֪��ϵ�д� Ӣ��СӢ������Ĭдϵ�д�
Ӣ��СӢ������Ĭдϵ�д� �����ҵ���������ͯ������ϵ�д�
�����ҵ���������ͯ������ϵ�д�| A�� | SO2ʹ���Ը��������Һ��ɫ | |
| B�� | SO2ʹ���з�̪������������Һ��ɫ | |
| C�� | SO2ʹƷ����Һ��ɫ | |
| D�� | SO2ʹ��ˮ��ɫ |
| A�� | ��Ӧʱ��������pH���� | B�� | ����ܷ�ӦΪ2H2+O2=2H2O | ||
| C�� | O2������������ԭ��Ӧ | D�� | ����ʱOH-���ƶ� |
| A�� | CH4��g��+2O2��g��=CO2��g��+2H2O��l����H=+890 kJ/mol | |
| B�� | CH4��g��+2O2��g��=CO2��g��+2H2O��g����H=-890kJ/mol | |
| C�� | CH4��g��+2O2��g��=CO2��g��+2H2O��l����H=-445kJ/mol | |
| D�� | CH4��g��+2O2��g��=CO2��g��+2H2O��l����H=-890kJ/mol |
| A�� | �����붡�� | B�� | �������춡�� | C�� | ��������� | D�� | 12C��13C |
��OH-�ĵ���ʽ
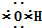
����ϩ�Ľṹ��ʽ��CH2=CH2
�ۼ���Ľṹʽ
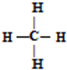
����ϩ�Ľṹʽ��C2H4��
| A�� | �٢� | B�� | �ڢ� | C�� | �ڢ� | D�� | �٢� |
| A�� | ���ͻ����� | B�� | ���������屽 | C�� | �����������Ҵ� | D�� | �Ҵ������� |
���Ʊ�TiCl4
ʵ�������÷�ӦTiO2 ��s��+CCl4��g��$\frac{\underline{\;\;��\;\;}}{\;}$TiCl4��g��+CO2��g��������ˮ�����������Ʊ�TiCl4��װ��ͼ���й���Ϣ���£�

| ���� | �۵�/�� | �е�/�� | ���� |
| CCl4 | -23 | 76 | ��TiCl4���� |
| TiCl4 | -25 | 136 | ����ʪ������������ |
��1������A�����������θ���ܣ�
��2��ʵ�鿪ʼǰ�IJ�������������װ�������ڼ���װ�������ԣ��ۼ�װҩƷ����ͨN2һ��ʱ�䣬������ȼ�ƾ��ƣ�ʵ�������������D�е�Һ̬���������ò��������������� �������
��3��װ��E�ܷ��ܣ���ܡ����ܡ�������װ��A��������װ��A�������տ����е����������ܱ�֤��Ӧ�����������½���
��4��TiCl4����ȡ���캽�չ�ҵ����--�ѺϽ����Ҫԭ�ϣ�ij�ѺϽ��Ԫ�ػ���Al��Si�ȣ���֪�ڳ����£�����һ����ǿ��ǿ��Ľ����������ʵ��������е�SiԪ��ȡ�����Ͻ���Ʒ���Թ��У��μӹ���NaOH��Һ�������ú�ȡ�ϲ���Һ������ε���ϡ���������������г������������ճ�������ȫ�ܽ⣬��֤����Ʒ����Si
���Ʊ�TiO2���ⶨTiO2������������
��TiCl4�м�ˮ�����ȣ�ˮ��õ�����TiO2•xH2O�������ˡ�ˮϴ��ȥ���е�Cl-���ٺ�ɡ����ճ�ȥˮ�ֵõ�����TiO2��
��5��д������TiO2•xH2O�Ļ�ѧ����ʽTiCl4+��x+2��H2O$\frac{\underline{\;\;��\;\;}}{\;}$TiO2•xH2O��+4HCl��
��6��һ�������£���TiO2�ܽⲢ��ԭΪTi3+����NH4Fe��SO4��2����Һ�ζ�Ti3+��ȫ������Ti4+���ζ�����ʱ����ȡ����TiO2����0.2g������0.1mol•L-1 NH4Fe��SO4��2����Һ20mL��
| TiCl4 | Mg | MgCl2 | Ti | |
| �۵�/�� | -25.0 | 648.8 | 714 | 1667 |
| �е�/�� | 136.4 | 1090 | 1412 | 3287 |
�ڸõζ��������õ���ָʾ����KSCN��Һ��
����TiO2��������Ϊ80%��