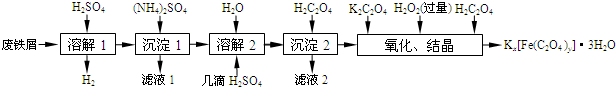
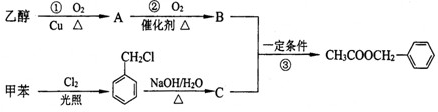
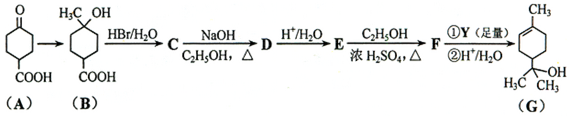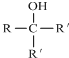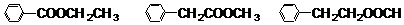��Ŀ����
��֪�����ǻ�ͬʱ����ͬһ��̼ԭ����ʱ�ṹ�Dz��ȶ��ģ�����������ˮ��Ӧ�����磺CH3CH��OH��2��CH3CHO+H2O�����з���ʽΪC9H8O2Br2������M����֪E�Ļ�ѧʽΪC7H5O2Na��A����Է�������Ϊ46����һ�������¿ɷ�������һϵ�з�Ӧ��
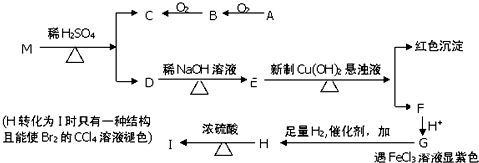
��ش���������
��1��B�й����ŵ����� ��A�ĺ˴Ź��������� �����շ壻G��H�ķ�Ӧ���� ��
��2��M�Ľṹ��ʽ
��3��д�����з�Ӧ�Ļ�ѧ����ʽ
��A��B ��H��I
��4��ͬʱ��������������G��ͬ���칹��Ľṹ��ʽ�� �֣�д������һ��
A����������
B���ܷ���ˮ�ⷴӦ
C���ܷ���������Ӧ
D����FeCl3��Һ��Ӧ����ɫ��

��ش���������
��1��B�й����ŵ�����
��2��M�Ľṹ��ʽ
��3��д�����з�Ӧ�Ļ�ѧ����ʽ
��A��B
��4��ͬʱ��������������G��ͬ���칹��Ľṹ��ʽ��
A����������
B���ܷ���ˮ�ⷴӦ
C���ܷ���������Ӧ
D����FeCl3��Һ��Ӧ����ɫ��
���㣺�л�����ƶ�
ר�⣺
������A����Է�������Ϊ46��A��������B��B�ɼ���������C����B��������C������AΪCH3CH2OH��BΪCH3CHO��CΪCH3COOH��E�Ļ�ѧʽΪC7H5O2Na������������ͭ��Һ��Ӧ����ש��ɫ������˵��E�к���-CHO��Hת��ΪIʱ����ʹ������Ȼ�̼��Һ��ɫ��˵����H�еĴ��ǻ���ˮ������̼̼˫��������ֻ��һ�ֽṹ��˵��H�еĴ��ǻ��ں���һ��ȡ����������Ԫ���Ķ�λ���������и����ʵ�ת����ϵ����֪D��E��F�б����ϵ�ȡ�������ڶ�λ����EΪ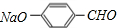 �����������ǿ�ڷӣ��ڼ��������£������л����ת����֪FΪ
�����������ǿ�ڷӣ��ڼ��������£������л����ת����֪FΪ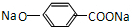 ��GΪ
��GΪ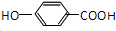 ��HΪ
��HΪ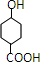 ����IΪ
����IΪ ��DӦΪBr2CH-
��DӦΪBr2CH- -OH��MΪ��
-OH��MΪ��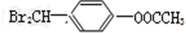 ���ݴ˴��⣮
���ݴ˴��⣮
 �����������ǿ�ڷӣ��ڼ��������£������л����ת����֪FΪ
�����������ǿ�ڷӣ��ڼ��������£������л����ת����֪FΪ ��GΪ
��GΪ ��HΪ
��HΪ ����IΪ
����IΪ ��DӦΪBr2CH-
��DӦΪBr2CH- -OH��MΪ��
-OH��MΪ�� ���ݴ˴��⣮
���ݴ˴��⣮���
�⣺A����Է�������Ϊ46��A��������B��B�ɼ���������C����B��������C������AΪCH3CH2OH��BΪCH3CHO��CΪCH3COOH��E�Ļ�ѧʽΪC7H5O2Na������������ͭ��Һ��Ӧ����ש��ɫ������˵��E�к���-CHO��Hת��ΪIʱ����ʹ������Ȼ�̼��Һ��ɫ��˵����H�еĴ��ǻ���ˮ������̼̼˫��������ֻ��һ�ֽṹ��˵��H�еĴ��ǻ��ں���һ��ȡ����������Ԫ���Ķ�λ���������и����ʵ�ת����ϵ����֪D��E��F�б����ϵ�ȡ�������ڶ�λ����EΪ �����������ǿ�ڷӣ��ڼ��������£������л����ת����֪FΪ
�����������ǿ�ڷӣ��ڼ��������£������л����ת����֪FΪ ��GΪ
��GΪ ��HΪ
��HΪ ����IΪ
����IΪ ��DӦΪBr2CH-
��DӦΪBr2CH- -OH��MΪ��
-OH��M�� ��
��
��1��BΪCH3CHO������ȩ����AΪCH3CH2OH������3�����ʲ�ͬ��H���˴Ź���������3�����շ壬G��H��GΪ ��ͨ���ӳɷ�Ӧ������
��ͨ���ӳɷ�Ӧ������ ��
��
�ʴ�Ϊ��ȩ���� 3���ӳɷ�Ӧ��
��2�������Ϸ�����֪MΪ ��
��
�ʴ�Ϊ�� ��
��
��3����AΪCH3CH2OH���ڼ��������´�������CH3CHO
����Ӧ�ķ���Ϊ2CH3CH2OH+O2��2CH3CHO��
�ʴ�Ϊ2CH3CH2OH+O2��2CH3CHO��
��HΪ ��ͨ����ȥ��Ӧ������
��ͨ����ȥ��Ӧ������ ����Ӧ�ķ���ʽΪ
����Ӧ�ķ���ʽΪ ��
��
�ʴ�Ϊ�� ��
��
��4��GΪ ����������֪G��ͬ���칹��Ӧ����-COO-��-CHO���Լ����ǻ���������������
����������֪G��ͬ���칹��Ӧ����-COO-��-CHO���Լ����ǻ��������������� ���ڡ��䡢�ԣ��������֣�
���ڡ��䡢�ԣ��������֣�
�ʴ�Ϊ��3�� ���ڡ��䡢����дһ�֣���
���ڡ��䡢����дһ�֣���
 �����������ǿ�ڷӣ��ڼ��������£������л����ת����֪FΪ
�����������ǿ�ڷӣ��ڼ��������£������л����ת����֪FΪ ��GΪ
��GΪ ��HΪ
��HΪ ����IΪ
����IΪ ��DӦΪBr2CH-
��DӦΪBr2CH- -OH��MΪ��
-OH��M�� ��
����1��BΪCH3CHO������ȩ����AΪCH3CH2OH������3�����ʲ�ͬ��H���˴Ź���������3�����շ壬G��H��GΪ
 ��ͨ���ӳɷ�Ӧ������
��ͨ���ӳɷ�Ӧ������ ��
���ʴ�Ϊ��ȩ���� 3���ӳɷ�Ӧ��
��2�������Ϸ�����֪MΪ
 ��
���ʴ�Ϊ��
 ��
����3����AΪCH3CH2OH���ڼ��������´�������CH3CHO
����Ӧ�ķ���Ϊ2CH3CH2OH+O2��2CH3CHO��
�ʴ�Ϊ2CH3CH2OH+O2��2CH3CHO��
��HΪ
 ��ͨ����ȥ��Ӧ������
��ͨ����ȥ��Ӧ������ ����Ӧ�ķ���ʽΪ
����Ӧ�ķ���ʽΪ ��
���ʴ�Ϊ��
 ��
����4��GΪ
 ����������֪G��ͬ���칹��Ӧ����-COO-��-CHO���Լ����ǻ���������������
����������֪G��ͬ���칹��Ӧ����-COO-��-CHO���Լ����ǻ��������������� ���ڡ��䡢�ԣ��������֣�
���ڡ��䡢�ԣ��������֣��ʴ�Ϊ��3��
 ���ڡ��䡢����дһ�֣���
���ڡ��䡢����дһ�֣���
���������⿼���л�����ƶϣ��漰����ȩ�����ᡢ±�����������ӵ����ʵȣ��Ѷ��еȣ�����ʱ��Ҫ�Ը������Ϣ�������ã����չ����ŵ������ǹؼ�����ȡ���Ʒ������Ʒ����ϵķ����ƶϣ�

��ϰ��ϵ�д�
�����Ŀ
���ֽ�����Ϸ�ĩ15g�������������ᷴӦǡ������11.2L��������״������������������Ľ��������Ϊ��������
| A��Mg��Al |
| B��Al��Zn |
| C��Cu��Zn |
| D��Mg��Ag |
���з�Ӧ�����ӷ���ʽ�У���д��ȷ���ǣ�������
| A���������������ᷴӦBa2++OH-+H++SO42-=BaSO4��+H2O |
| B�����۸�ϡ���ᷴӦ2Fe+6H+=2Fe3++3H2�� |
| C���Ȼ�����Һ��̼������Һ��ӦCa2++CO32-=CaCO3�� |
| D��̼��Ƹ����ᷴӦCO32-+2H+=H2O+CO2�� |