��Ŀ����
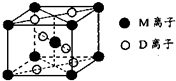 ��2013?����ģ�⣩A��B��D��E��G��M�������ֳ���Ԫ�أ����ǵĺ˵���������������У�Ԫ��M�Ļ�̬3d�������2�����ӣ�A�Ļ�̬ԭ��L���������K���������2����E�� ��������ͬ����Ԫ�صļ������а뾶��С��D��Gͬ���壻B��D�γɵĻ������ж��֣�����һ���Ǻ���ɫ���壮
��2013?����ģ�⣩A��B��D��E��G��M�������ֳ���Ԫ�أ����ǵĺ˵���������������У�Ԫ��M�Ļ�̬3d�������2�����ӣ�A�Ļ�̬ԭ��L���������K���������2����E�� ��������ͬ����Ԫ�صļ������а뾶��С��D��Gͬ���壻B��D�γɵĻ������ж��֣�����һ���Ǻ���ɫ���壮��1��M�Ļ�̬ԭ�Ӽ۲�����Ų�ʽΪ
3d24s2
3d24s2
��Ԫ��B��D��G�ĵ�һ�������ɴ�ʱС��˳����N��O��S
N��O��S
����Ԫ�ط��ű�ʾ������2���ü۲���ӶԻ�������Ԥ�⣬GD32-�����幹����
������
������
�������ֱ�������3��M��D�γɵ�һ�ֳȺ�ɫ���徧���ṹ��ͼ��ʾ���仯ѧʽΪ
TiO2
TiO2
����Ԫ�ط��ű�ʾ������4����֪������EB�ṹ�뵥�������ƣ������ʿ���E ���Ȼ�����NaB3�ڸ����·�Ӧ�Ƶã������ɵ���B2���÷�Ӧ��ѧ����ʽΪ
AlCl3+3NaN3�T3NaCl+AlN+4N2��
AlCl3+3NaN3�T3NaCl+AlN+4N2��
������8.4gB2���ɣ���ת�Ƶ�����Ϊ1.204��1023��0.2NA
1.204��1023��0.2NA
��������A��B��D��E��G��M�������ֳ���Ԫ�أ����ǵĺ˵���������������У�Ԫ��M�Ļ�̬3d�������2�����ӣ���M��TiԪ�أ�A�Ļ�̬ԭ��L���������K���������2������A��CԪ�أ�B��D�γɵĻ������ж��֣�����һ���Ǻ���ɫ���壬����ɫ�����Ƕ���������B��ԭ������С��D������B��NԪ�أ�D��OԪ�أ�D��Gͬ���壬G��ԭ������С��M������G��SԪ�أ�E�ļ�������ͬ����Ԫ�صļ������а뾶��С����E��AlԪ�أ�
����⣺A��B��D��E��G��M�������ֳ���Ԫ�أ����ǵĺ˵���������������У�Ԫ��M�Ļ�̬3d�������2�����ӣ���M��TiԪ�أ�A�Ļ�̬ԭ��L���������K���������2������A��CԪ�أ�B��D�γɵĻ������ж��֣�����һ���Ǻ���ɫ���壬����ɫ�����Ƕ���������B��ԭ������С��D������B��NԪ�أ�D��OԪ�أ�D��Gͬ���壬G��ԭ������С��M������G��SԪ�أ�E�ļ�������ͬ����Ԫ�صļ������а뾶��С����E��AlԪ�أ�
��1��M��TiԪ�أ���۵�����3d��4s�ϵ��ӣ��������̬ԭ�Ӽ۲�����Ų�ʽΪ3d24s2��ͬһ�����У�Ԫ�صĵ�һ����������ԭ�����������������������ƣ�����VAA��Ԫ�ش��ڵ�VIA��Ԫ�أ�ͬһ�����У�Ԫ�صĵ�һ����������ԭ���������������С���������һ�����ܵĴ�С˳���ǣ�N��O��S��
�ʴ�Ϊ��3d24s2��N��O��S��
��2���ü۲���ӶԻ�������Ԥ�⣬SO32-�м۲���Ӷ�=3+
(6+2-3��2)=4��������һ���µ��Ӷԣ����������幹���������ͣ��ʴ�Ϊ�������ͣ�
��3���þ�����Tiԭ�Ӹ���=1+8��
=2��Oԭ�Ӹ���=2+4��
=4�������ѡ���ԭ�Ӹ�����=2��4=1��2�����仯ѧʽΪ��TiO2���ʴ�Ϊ��TiO2��
��4��Al���Ȼ�����NaB3�ڸ����·�Ӧ����AlN��N2������Ԫ���غ�֪���������Ȼ��ƣ����Ը÷�Ӧ����ʽΪ��AlCl3+3NaN3�T3NaCl+AlN+4N2����ÿ����112g����ת�Ƶ��ӵ����ʵ�����
mol������8.4gB2���ɣ���ת�Ƶ�����=
NA=0.2NA=1.204��1023���ʴ�Ϊ��AlCl3+3NaN3�T3NaCl+AlN+4N2����1.204��1023��0.2NA��
��1��M��TiԪ�أ���۵�����3d��4s�ϵ��ӣ��������̬ԭ�Ӽ۲�����Ų�ʽΪ3d24s2��ͬһ�����У�Ԫ�صĵ�һ����������ԭ�����������������������ƣ�����VAA��Ԫ�ش��ڵ�VIA��Ԫ�أ�ͬһ�����У�Ԫ�صĵ�һ����������ԭ���������������С���������һ�����ܵĴ�С˳���ǣ�N��O��S��
�ʴ�Ϊ��3d24s2��N��O��S��
��2���ü۲���ӶԻ�������Ԥ�⣬SO32-�м۲���Ӷ�=3+
| 1 |
| 2 |
��3���þ�����Tiԭ�Ӹ���=1+8��
| 1 |
| 8 |
| 1 |
| 2 |
��4��Al���Ȼ�����NaB3�ڸ����·�Ӧ����AlN��N2������Ԫ���غ�֪���������Ȼ��ƣ����Ը÷�Ӧ����ʽΪ��AlCl3+3NaN3�T3NaCl+AlN+4N2����ÿ����112g����ת�Ƶ��ӵ����ʵ�����
| 8 |
| 3 |
8.4��
| ||
| 112 |
���������⿼�����ʽṹ�����ʣ���ȷ�ƶ�Ԫ���ǽⱾ��ؼ���ע�⣨4�����е������е�Ԫ�صĻ��ϼۣ�Ϊ�״��㣮

��ϰ��ϵ�д�
 ��������ϵ�д�
��������ϵ�д� ����˼ά����ѵ����ʱ��ѧ��ϵ�д�
����˼ά����ѵ����ʱ��ѧ��ϵ�д�
�����Ŀ
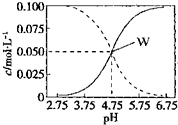 ��2013?����ģ�⣩25��ʱ��c��CH3COOH��+c��
��2013?����ģ�⣩25��ʱ��c��CH3COOH��+c��