��Ŀ����
��Ӧ4NH3��������5O2������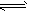 4NO��������6H2O��������10L�ܱ������н��У�����Ӻ�ˮ���������ʵ���������0��45mol����˷�Ӧ��ƽ������
4NO��������6H2O��������10L�ܱ������н��У�����Ӻ�ˮ���������ʵ���������0��45mol����˷�Ӧ��ƽ������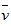 (X)(��Ӧ����������ʻ�������������)�ɱ�ʾΪ
(X)(��Ӧ����������ʻ�������������)�ɱ�ʾΪ
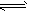 4NO��������6H2O��������10L�ܱ������н��У�����Ӻ�ˮ���������ʵ���������0��45mol����˷�Ӧ��ƽ������
4NO��������6H2O��������10L�ܱ������н��У�����Ӻ�ˮ���������ʵ���������0��45mol����˷�Ӧ��ƽ������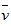 (X)(��Ӧ����������ʻ�������������)�ɱ�ʾΪ
(X)(��Ӧ����������ʻ�������������)�ɱ�ʾΪ [ ]
A��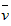 (NH3)=0��0100mol��L-1��s-1
(NH3)=0��0100mol��L-1��s-1
B��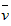 (O2)=0��0010mol��L-1��s-1
(O2)=0��0010mol��L-1��s-1
C��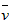 (NO)=0��0010mol��L-1��s-1
(NO)=0��0010mol��L-1��s-1
D��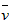 (H2O)=0��045mol��L-1��s-1
(H2O)=0��045mol��L-1��s-1
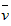 (NH3)=0��0100mol��L-1��s-1
(NH3)=0��0100mol��L-1��s-1B��
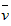 (O2)=0��0010mol��L-1��s-1
(O2)=0��0010mol��L-1��s-1 C��
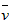 (NO)=0��0010mol��L-1��s-1
(NO)=0��0010mol��L-1��s-1D��
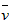 (H2O)=0��045mol��L-1��s-1
(H2O)=0��045mol��L-1��s-1 C

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
��Ӧ4NH3������+5O2������?4NO������+6H2O����������2L���ܱ������н��У�1���Ӻ�NH3������0.12mol����ƽ��ÿ����Ũ�ȱ仯��ȷ���ǣ�������
| A��NO��0.001Ħ/�� | B��H2O��0.002Ħ/�� | C��NH3��0.002Ħ/�� | D��O2��0.002Ħ/�� |
 4NO������+6H2O��������2�����ܱ������н��У�1���Ӻ�NH3������0.12Ħ������ƽ��ÿ����Ũ�ȱ仯��ȷ���ǣ�������
4NO������+6H2O��������2�����ܱ������н��У�1���Ӻ�NH3������0.12Ħ������ƽ��ÿ����Ũ�ȱ仯��ȷ���ǣ�������