��Ŀ����
9��������������������մ���Һ���������Ժͽ�ǿ�Ļ�ԭ�ԣ�����֯��Ư������ȼ������������еĻ�ԭ������������ƣ�Na2S203�������������ƺ����ͨ�����Ϸ�Ӧ�Ƶã�װ����ͼI��ʾ��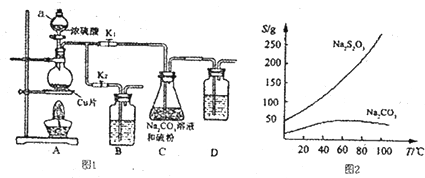
��֪��Na2S203��������Һ�в����ȶ����ڣ��й����ʵ��ܽ��������ͼ2��ʾ��
��1��Na2S203•5H20���Ʊ���
����1����ͼ���Ӻ�װ�ú�δװҩƷ�������A��Cװ�������ԵIJ�����_�ر�K2��K1����D�м�ˮ��û����ĩ�ˣ�����ë����˫����ס��ƿ��D�е���������ð������ȴ���γ�1��ˮ����˵�����������ã�
����2������ҩƷ����K1���ر�K2�����ȣ�װ��B��D�е�ҩƷ��ѡ�����������е�ACD�����ţ���
A��NaOH��Һ B��ŨH2S04C������KMnO4��Һ D������NaHCO3��Һ
����3��C�л��Һ��������������Ӧһ��ʱ�����۵������٣�
����4������C�еĻ��Һ������Һ��������Ũ�������ȹ��ˣ��ٽ���Һ��ȴ�ᾧ���ˡ�ϴ�ӡ���ɣ��õ���Ʒ��
��2��Na2S2O3���ʵļ��飺��������������ˮ�еμ�����Na2S2O3��Һ����ˮ��ɫ��dz����鷴Ӧ����Һ�к����������д���÷�Ӧ�Ļ�ѧ����ʽNa2S2O3+4Cl2+5H2O=Na2SO4+H2SO4+8HCl��
��3������Na2S203��Һ�ⶨ��ˮ��Ba2+Ũ�ȣ��������£�ȡ��ˮ25.00mL�������ʵ�����ȼ�������K2Cr2O7��Һ����BaCrO4���������ˡ�ϴ�Ӻ�������ϡ�����ܽ⣮��ʱCr42-ȫ��ת��ΪCr2O72-���ټӹ���KI��Һ����ַ�Ӧ���������Һ��ָʾ������0.010mol•L-1��Na2S2O3��Һ���еζ�����Ӧ��ȫʱ������Na2S2O3��Һ18.00Ml�����ַ�Ӧ�����ӷ���ʽΪ��Cr2072-+6I-+14H+�T2Cr3++3I2+7H20��I2+2S2O32-�TS4O62-+2I-����÷�ˮ��Ba2+�����ʵ���Ũ��Ϊ0.0024mol/L��
���� ��1������1���������������������ʣ�����װ�������ԣ�
����2��װ��B��D�������ǽ���β����������ֹβ���ж���������Ⱦ������
����3�������������������Һ�в��ȶ���Ӧ������ҺΪ�����ԣ���������ҺpH�ӽ���С��7��
����4������Һ�л�þ��壬��Ҫ����Ũ�������ȹ��ˣ��ٽ���Һ��ȴ�ᾧ�����ˡ�ϴ�ӡ���ɣ��õ���Ʒ��
��2������Ŀ��Ϣ��֪��Na2S2O3����ˮ������Ӧ����Na2SO4��H2SO4����������ԭΪHCl��
��3���������֪��BaCrO4�������ܽ�ת��ΪCr2O2-7����Ԫ���غ㼰��֪����ʽ�ɵù�ϵʽ��2Ba2+��2BaCrO4��Cr2O2-7��3I2��6Na2S2O3��������ĵ�Na2S2O3���ù�ϵʽ������Һ��n��Ba2+������������c��Ba2+����
��� �⣺��1������1���������������������ʣ�����װ�������ԣ��������Ϊ���ر�K2��K1����D�м�ˮ��û����ĩ�ˣ�����ë����˫����ס��ƿ��D�е���������ð������ȴ���γ�1��ˮ����˵�����������ã�
�ʴ�Ϊ���ر�K2��K1����D�м�ˮ��û����ĩ�ˣ�����ë����˫����ס��ƿ��D�е���������ð������ȴ���γ�1��ˮ����˵�����������ã�
����2��װ��B��D�������ǽ���β����������ֹβ���ж���������Ⱦ����������������л�ԭ�ԣ����������Ը��������Һ�������գ�����������������������Һ��̼��������Һ��Ӧ�����գ���ѡ��ACD��
����3�������������������Һ�в��ȶ���Ӧ������ҺΪ�����ԣ����Կ�����ҺpH�ӽ���С��7��
����4������Һ�л�þ��壬��Ҫ����Ũ�������ȹ��ˣ��ٽ���Һ��ȴ�ᾧ�����ˡ�ϴ�ӡ���ɣ��õ���Ʒ��
�ʴ�Ϊ����ȴ�ᾧ��
��2������Ŀ��Ϣ��֪��Na2S2O3����ˮ������Ӧ����Na2SO4��H2SO4����������ԭΪHCl����Ӧ����ʽΪ��Na2 S2O3+4Cl2+5H2O=Na2SO4+H2SO4+8HCl��
�ʴ�Ϊ��Na2S2O3+4Cl2+5H2O=Na2SO4+H2SO4+8HCl��
��3���������֪��BaCrO4�������ܽ�ת��ΪCr2O2-7����Ԫ���غ㼰��֪����ʽ�ɵù�ϵʽ��2Ba2+��2BaCrO4��Cr2O2-7��3I2��6Na2S2O3�����ĵ�Na2S2O3Ϊ0.018L��0.01mol/L����n��Ba2+��=0.018L��0.01mol/L��$\frac{1}{3}$=0.00006mol������Һ��c��Ba2+��=$\frac{0.00006mol}{0.025L}$=0.0024mol/L��
�ʴ�Ϊ��0.0024mol/L��
���� ���⿼��ʵ���Ʊ�������ƣ��漰�����Լ��顢��ʵ��װ�ü�����ķ������ۡ����ʵķ����ᴿ��������ԭ��Ӧ�ζ�����3����ע�����ù�ϵʽ���м��㣬�Ѷ��еȣ�


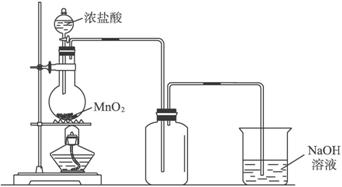
��1���Ʊ�����ѡ�õ�ҩƷΪ���������̺�Ũ���ᣬ��ط�Ӧ�Ļ�ѧ����ʽΪ��MnO2+4HCl��Ũ��$\frac{\underline{\;����\;}}{\;}$MnCl2+Cl2��+2H2O��
��2��װ��B�б���ʳ��ˮ�������dz�ȥCl2�е�HCl��ͬʱװ��B���ǰ�ȫƿ�����ʵ�����ʱC���Ƿ�����������д����������ʱB�е�����B�г���©����Һ���������γ�ˮ����
��3��װ��F�ձ���ʢ�ŵ��Լ���NaOH��Һ�����з�����Ӧ�����ӷ���ʽ��2OH-+Cl2=Cl-+ClO-+H2O��
��4��װ��C��ʵ��Ŀ������֤�����Ƿ����Ư���ԣ�Ϊ��C�Т����η���D��
| ѡ�� | �� | �� | �� |
| A | �������ɫ���� | ��ʯ�� | ʪ�����ɫ���� |
| B | �������ɫ���� | �轺 | ʪ�����ɫ���� |
| C | ʪ�����ɫ���� | Ũ���� | �������ɫ���� |
| D | ʪ�����ɫ���� | ��ˮ�Ȼ��� | �������ɫ���� |
H+��aq��+OH-��aq��=H2O��l������H=-57.3kJ•mol-1����֪����Һ�з�Ӧ�У�
CH3COOH��aq��+NaOH��aq��=CH3COONa��aq��+H2O��l������H=-Q1kJ•mol-1��
$\frac{1}{2}$H2SO4��Ũ��+NaOH��aq��=$\frac{1}{2}$Na2SO4��aq��+H2O��l������H=-Q2 kJ•mol-1
HNO3��aq��+KOH��aq���TKNO3��aq��+H2O��l������H=-Q3 kJ•mol-1��
��Q1��Q2��Q3�Ĺ�ϵ��ȷ���ǣ�������
| A�� | Q1=Q2=Q3 | B�� | Q2��Q1��Q3 | C�� | Q2��Q3��Q1 | D�� | Q2=Q3��Q1 |
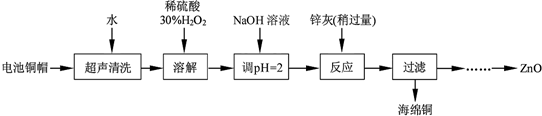
��1��Ϊȷ������п�ң���Ҫ�ɷ�ΪZn��ZnO������Ϊ�����������������ʵ������ⶨ��ȥH2O2����Һ��Cu2+�ĺ�����ʵ�����Ϊ��ȷ��ȡһ������ĺ���Cu2+����Һ�ڴ�����ƿ�У�������ˮϡ�ͣ�����pH=3��4���������KI����Na2S2O3����Һ�ζ����յ㣮���������е����ӷ���ʽ���£�2Cu2++4I-�T2CuI����ɫ����+I2��I2+2S2O32-�T2I-+S4O62-
��Na2S2O3����ҺӦװ�ڼ�ʽ������ʽ���ʽ���ζ����ڣ�
�ڵζ�ѡ�õ�ָʾ��Ϊ������Һ���ζ��յ�۲쵽������Ϊ�μ����һ�Σ���ɫ����ɫ�����ɫ���Ұ�����ڲ��䣮
�����ζ�ǰ��Һ��H2O2û�г��������ⶨ��Cu2+�ĺ�������ƫ�ߣ��ƫ�ߡ���ƫ�͡������䡱����
��2����֪pH��11ʱZn��OH��2������NaOH��Һ����[Zn��OH��4]2-���±��г��˼����������������������pH��Χ����ʼ������pH����������Ũ��Ϊ1.0mol•L-1���㣩��
ʵ���п�ѡ�õ��Լ���30% H2O2��1.0mol•L-1HNO3��1.0mol•L-1 NaOH��
| ��ʼ������pH | ��ȫ������pH | |
| Fe3+ | 1.1 | 3.2 |
| Fe2+ | 5.8 | 8.8 |
| Zn2+ | 5.9 | 8.9 |
�ټ�H2O2������Fe2+��
�ڼ�����������Һ������PH��4���ң�
�۹��ˣ�
�ܼ�����������Һ������PH��8.9-11֮�䣻
�ݹ��ˡ�ϴ�ӡ�����
��900�����գ�
| A�� | �Ͻ������ٺ������ֽ��� | |
| B�� | Ư�����ڿ����г��ڴ�� | |
| C�� | ����ʦ����������ʴʯӢ��������Ʒ | |
| D�� | NaOH��Һ����������ĥ�ڲ�������ϸ���Լ�ƿ�� |
| A�� | p��Ne����p��N2����p��O2�� | B�� | p��O2����p��Ne����p��N2�� | C�� | p��N2����p��O2����p��Ne�� | D�� | p��N2����p��Ne����p��O2�� |

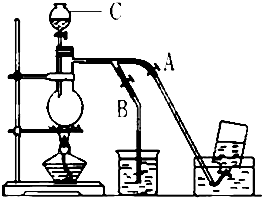 ��ͼ��ʾ��û��ͨ������������Ʊ�����ʱ��Ƶ�װ�ã�ͼ��a��b�ǿɿ��Ƶĵ������У��������ڱ����Ȼ�����Һ�е��ܽ�Ƚ�С����
��ͼ��ʾ��û��ͨ������������Ʊ�����ʱ��Ƶ�װ�ã�ͼ��a��b�ǿɿ��Ƶĵ������У��������ڱ����Ȼ�����Һ�е��ܽ�Ƚ�С����