��Ŀ����
 ��������й�����ʵ�飬�ش��й����⣮
��������й�����ʵ�飬�ش��й����⣮��1����˿��������ȼ�յ�ʵ�飺
��ȼϵ������״ϸ��˿�˵Ļ�����Ҫȼ��ʱ������ʢ�������ļ���ƿ�У��۲������������������
��2����ͼ��̽��������ʴ������ʵ�飺
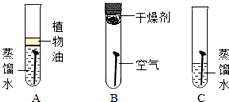
��A��ֲ���͵�������
��һ��ʱ���B������û����ʴ����C��������ʴ��ͨ��B��C�Ա�˵��������ʴ������֮һ��
���㣺�����ĵ绯ѧ��ʴ�����,���Ļ�ѧ����
ר�⣺
��������1���������Ļ�ѧ���ʽ������ȼ���������ش��жϣ�
��2����������������ǣ������Ϳ����Լ�ˮ�Ӵ���ֲ���ͺ�ˮ�ǻ������ܵģ��ݴ˻ش�
��2����������������ǣ������Ϳ����Լ�ˮ�Ӵ���ֲ���ͺ�ˮ�ǻ������ܵģ��ݴ˻ش�
���
�⣺��1�������Ż��ϸߣ�������ȼ��ʵ��ʱ����ȼϵ������״ϸ��˿�˵Ļ�����Ҫȼ��ʱ������ʢ�������ļ���ƿ�У��۲��������������������ȼ��˿������ȼ�ջ������䣬�ų��������ȣ�����Ϊ�˷�ֹ����ƿ��ը�ѣ�ƿ������ˮ��
�ʴ�Ϊ��ƿ������ˮ����ȼ��˿��
��2����̽��������Ҫ���Ʊ�����ֲ���͵������Ǹ�������������ֻ��ˮ�������½����ܷ����⣬�ʴ�Ϊ������������
��B������ֻ�������Ӵ���C�������������Լ�ˮ�Ӵ���һ��ʱ���B�е����������⣬����C�е����⣬ͨ��B��C�Ա�˵��������ʴ������֮һ����ˮ�Ӵ���
�ʴ�Ϊ����ˮ�Ӵ���
�ʴ�Ϊ��ƿ������ˮ����ȼ��˿��
��2����̽��������Ҫ���Ʊ�����ֲ���͵������Ǹ�������������ֻ��ˮ�������½����ܷ����⣬�ʴ�Ϊ������������
��B������ֻ�������Ӵ���C�������������Լ�ˮ�Ӵ���һ��ʱ���B�е����������⣬����C�е����⣬ͨ��B��C�Ա�˵��������ʴ������֮һ����ˮ�Ӵ���
�ʴ�Ϊ����ˮ�Ӵ���
���������⿼��ѧ����������������������Ϳ����Լ�ˮ�Ӵ���ע����Ʊ�������ʵ���е�Ӧ�ã��ѶȲ���

��ϰ��ϵ�д�
 ��������ܸ�ϰϵ�д�
��������ܸ�ϰϵ�д�
�����Ŀ
���������ʷֱ�ӵ������ɫ����������ʹ����ɫ��������
| A�����Ƶ���ˮ | B��84����Һ |
| C��Һ�� | D����������Һ |
����������ȷ���ǣ�������
��Na2O��Na2O2���ܺ�ˮ��Ӧ���ɼ���Ƕ��Ǽ���������
��Na2CO3��Һ��NaHCO3��Һ���ܸ�CaCl2��Һ��Ӧ�õ���ɫ����
�����ڳ����²����ױ�����
��Na2O2��������������Na2O����
��ʯ����Һ�м���Na2O2��ĩ���ȱ�������ɫ��������������
������ŨNH4Cl��Һ��Ӧ���ų��������к�H2��NH3��
��Na2O��Na2O2���ܺ�ˮ��Ӧ���ɼ���Ƕ��Ǽ���������
��Na2CO3��Һ��NaHCO3��Һ���ܸ�CaCl2��Һ��Ӧ�õ���ɫ����
�����ڳ����²����ױ�����
��Na2O2��������������Na2O����
��ʯ����Һ�м���Na2O2��ĩ���ȱ�������ɫ��������������
������ŨNH4Cl��Һ��Ӧ���ų��������к�H2��NH3��
| A������ȷ | B���ڢܢݢ� |
| C���ڢݢ� | D���ܢݢ� |
���з����е�����ԭ���ӻ������������ͬ���ǣ�������
| A��BF3��NH3 |
| B��BeCl2��SCl2 |
| C��H2O��SO2 |
| D��SO3��SO2 |
����˵����ȷ���ǣ�������
| A��CaCl2������״̬�¿ɵ��磬����ǿ����� |
| B��û�е��ʲμӵĻ��Ϸ�Ӧһ���Ƿ�������ԭ��Ӧ |
| C��Ԫ�ش������̬ʱһ������ǿ������ |
| D����2KClO3+4HCl��Ũ���T2KCl+2ClO2+Cl2+2H2O�У��������������������ǻ�ԭ���� |
��һ�������£��������ӵĻ�ԭ��˳��Ϊ��Cl-��Br-��Fe2+��I-��SO2���ɴ��ж����¸���Ӧ����Һ�������������ǣ�������
| A��2Fe3++SO2+2H2O=2Fe2++SO42-+4H+ |
| B��I2+SO2+2H2O=4H++SO42-+2I- |
| C��2Fe2++Cl2=2Fe3++2Cl- |
| D��2Br-+4H++SO42-=SO2+Br2+2H2O |