��Ŀ����
ij�����Һ�п��ܺ������������е������֣�K+��Na+��Fe2+��Fe3+��SO42-��NO2-��CO32- ��ȡ����20mL����Һ��������ʵ�飮��������ʵ�飬����˵����ȷ���ǣ�������
�ٵ�һ�ݼ�������ϡ���ᣬ���κ����������
�ڵڶ��ݼ�������NaOH�������裬���ˣ�ϴ�ӣ����գ����õ�x g���壻
�۵����ݵμ�0.1mol��L-1����KMnO4��Һ����Ӧ���յ㣬�����������ΪVmL��
�����ýྻ�IJ�˿պȡ�û����Һ����dzɫ���������գ����ֻ�����ֻ�ɫ��
�ٵ�һ�ݼ�������ϡ���ᣬ���κ����������
�ڵڶ��ݼ�������NaOH�������裬���ˣ�ϴ�ӣ����գ����õ�x g���壻
�۵����ݵμ�0.1mol��L-1����KMnO4��Һ����Ӧ���յ㣬�����������ΪVmL��
�����ýྻ�IJ�˿պȡ�û����Һ����dzɫ���������գ����ֻ�����ֻ�ɫ��
| A��ԭ�����Һ��ֻ����Na+��Fe3+��SO42-�������ܴ���K+��CO32- |
| B����ʵ������ƶ�ԭ�����Һ���Ƿ���SO42- |
| C����ʵ��١��ڿ��ж�ԭ�����Һ���Ƿ���Fe3+ |
| D����ʵ��ۿ��ж�ԭ�����Һ�д���Fe2+ |
���㣺���������ӵļ���,���������ӵļ���
ר�⣺
�������ٵ�һ�ݼ����������ᣬ���κ����������˵����Һ�в���CO32-���ӣ�Ҳ����NO2-���ӣ���NO2-+H+�THNO2��4HNO2�T4NO��+2H2O+O2����������ų���������Һ�ĵ����ԣ�������ֻ��SO42-���ӣ���һ������SO42-���ӣ�
�ڵڶ��ݼ�������NaOH��Һ���ɳ�����˵����Һ�к���Fe2+��Fe3+���ӣ�����߶��У��������õ�xg���壬��֪�ܹ����е���Ԫ�ص����ʵ�����
�۵����ݵμ�0.10mol?L-1����KMnO4��Һ�����������ΪVmL��˵����Һ�к��л�ԭ��Fe2+���ӣ��������ĵĸ�����ص����ʵ�����ȷ��Fe2+���ӵ����ʵ���������ȷ���Ƿ���Fe3+���ӣ�
�����ýྻ�IJ�˿պȡ�û����Һ����dzɫ���������գ����ֻ�����ֻ�ɫ��˵������Na+���ӣ�������ȷ���Ƿ���K+���ӣ���ͨ����ɫ�ܲ����۲죮
������������Һ��һ������Na+���ӡ�SO42-���ӣ�����CO32-���ӡ�����NO2-���ӣ�Fe2+��Fe3+����������һ�֣�
�ڵڶ��ݼ�������NaOH��Һ���ɳ�����˵����Һ�к���Fe2+��Fe3+���ӣ�����߶��У��������õ�xg���壬��֪�ܹ����е���Ԫ�ص����ʵ�����
�۵����ݵμ�0.10mol?L-1����KMnO4��Һ�����������ΪVmL��˵����Һ�к��л�ԭ��Fe2+���ӣ��������ĵĸ�����ص����ʵ�����ȷ��Fe2+���ӵ����ʵ���������ȷ���Ƿ���Fe3+���ӣ�
�����ýྻ�IJ�˿պȡ�û����Һ����dzɫ���������գ����ֻ�����ֻ�ɫ��˵������Na+���ӣ�������ȷ���Ƿ���K+���ӣ���ͨ����ɫ�ܲ����۲죮
������������Һ��һ������Na+���ӡ�SO42-���ӣ�����CO32-���ӡ�����NO2-���ӣ�Fe2+��Fe3+����������һ�֣�
���
�⣺A��ֻ��ͨ����ɫ�ܲ����۲����ȷ���Ƿ���K+���ӣ���A����
B����1����һ�ݼ����������ᣬ���κ����������˵����Һ�в���CO32-���ӣ�Ҳ����NO2-���ӣ���NO2-+H+�THNO2��4HNO2�T4NO��+2H2O+O2����������ų���������Һ�ĵ����ԣ�������ֻ��SO42-���ӣ���һ������SO42-���ӣ���B����
C������NaOH��Һ���ɳ�����˵����Һ�к���Fe2+��Fe3+���ӣ�����߶��У����Ը���ʵ��١��ڲ����ж�ԭ�����Һ���Ƿ���Fe3+����C����
D���μ�0.10mol?L-1����KMnO4��Һ�����������ΪVmL�����������ط�Ӧ��ֻ��Fe2+���ӣ�����֤��һ������Fe2+���ӣ���D��ȷ��
��ѡD��
B����1����һ�ݼ����������ᣬ���κ����������˵����Һ�в���CO32-���ӣ�Ҳ����NO2-���ӣ���NO2-+H+�THNO2��4HNO2�T4NO��+2H2O+O2����������ų���������Һ�ĵ����ԣ�������ֻ��SO42-���ӣ���һ������SO42-���ӣ���B����
C������NaOH��Һ���ɳ�����˵����Һ�к���Fe2+��Fe3+���ӣ�����߶��У����Ը���ʵ��١��ڲ����ж�ԭ�����Һ���Ƿ���Fe3+����C����
D���μ�0.10mol?L-1����KMnO4��Һ�����������ΪVmL�����������ط�Ӧ��ֻ��Fe2+���ӣ�����֤��һ������Fe2+���ӣ���D��ȷ��
��ѡD��
���������⿼�����Ӽ�����Ŀ������ʱע�����Ŀ���ҳ���Ӧ�ĵ������������ʵ����ʽ����ƶϣ���Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
�����й�˵����ȷ���ǣ�������
| A����NH3ͨ���ȵ�CuSO4��Һ����ʹCu2+��ԭ��Cu |
| B�������£�������ˮ���ܴ�������C1-��Fe3+��NO3-��Na+��SO32- |
| C��Ũ�Ⱥ������ͬ��NaOH��CH3COOH���ϣ���Һ��c��Na+����c��CH3COO-�� |
| D���ŵ練ӦΪH2+2NiO��OH��=2Ni��OH��2�ĵ�أ����ʱ��������NiO��OH������ԭ |
�����½�������Һ�������Ϻ�������Һ��pHһ������7���ǣ�������
| A��pH=4�������pH=10�İ�ˮ |
| B��pH=4�Ĵ�����Һ��pH=10������������Һ |
| C��0.1mol?L-1�������0.1mol?L-1������������Һ |
| D��0.1mol?L-1�������0.1mol?L-1������������Һ |
NAΪ����٤����������������������ǣ�������
| A����״���£�11.2 L�����к���0.5 NAԭ�� |
| B�����³�ѹ�£�46g NO2��N2O4��������к���ԭ������Ϊ3NA |
| C����״���£�0.1 mol Cl2�μӷ�Ӧ��ת�Ƶĵ�����Ŀһ��Ϊ0.2 NA |
| D��1 mol Na������O2��Ӧ������Na2O��Na2O2�Ļ���ת�Ƶ�������NA�� |


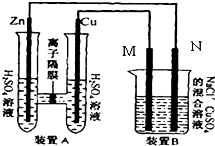 ��ͼװ��B����Ũ�Ⱦ�Ϊ0.1mol/L��NaCl��CuSO4�����Һ����Һ���Ϊ500ml��M��N��Ϊʯī�缫����װ��A��Zn����������6.5gʱ��N����������
��ͼװ��B����Ũ�Ⱦ�Ϊ0.1mol/L��NaCl��CuSO4�����Һ����Һ���Ϊ500ml��M��N��Ϊʯī�缫����װ��A��Zn����������6.5gʱ��N����������