��Ŀ����
��10�֣�����һ������ˮ��Һ��ֻ���ܺ������������е������֣�K+��NH4+��Cl����Mg2+��Ba2+��CO32����SO42������ȡ3��100 mL����Һ�ֱ��������ʵ�飺
��һ�ݼ���AgNO3��Һ�г���������
�ڶ��ݼ�������NaOH��Һ���Ⱥ��ռ�����״����20.16 L���壻
�����ݼ�������BaCl2��Һ�����ˡ�ϴ�ӡ�����õ���ɫ�������ٽ���������������ϴ�ӣ����ֳ��������ܽ⡣
��������ʵ�飬�Իش��������⣺
��1���ɵ�һ�ݽ��е�ʵ���Cl���Ƿ���ڵ��ж��� ���һ�����ڡ�����һ�������ڡ�����ȷ��������
��2���ɵڶ��ݽ��е�ʵ���֪�û������Ӧ���� �������ӷ��ţ��������ʵ���Ũ��Ϊ ��
��3��������ʵ�������յõ��ij����ɷ�Ϊ �����ѧʽ��
��4���ۺ�����ʵ�飬����Ϊ���½�����ȷ���� ��
A���û��Һ��һ����NH4+��CO32����SO42����K+�����ܺ�Cl��
B���û��Һ��һ����NH4+��CO32����SO42�������ܺ�K+��Cl��
C���û��Һ��һ����NH4+��CO32����SO42�������ܺ�Mg2+��K+��Cl��
D���û��Һ��һ����NH4+��SO42�������ܺ�Mg2+��K+��Cl��
��ÿ��2�֣���10�֣���1������ȷ���� ��2��NH4+��9 mol ��L��1�� ��3��BaSO4�� ��4��B
��������
�����������һ�ݼ���AgNO3��Һ�г�����������֪ԭ��Һ�����ٺ���Cl����CO32����SO42���е�һ�֣��ڶ��ݼ�������NaOH��Һ���Ⱥ��ռ�����״����20.16 L���壬��֪ԭ��Һ�к���NH4+�������ʵ���Ϊ20.16L/22.4L/mol=0.9mol�������ʵ���Ũ��Ϊ0.9mol/0.1L=9mol/L�������ݼ�������BaCl2��Һ�����ˡ�ϴ�ӡ�����õ���ɫ�������ٽ���������������ϴ�ӣ����ֳ��������ܽ⣬��֪ԭ��Һ�к���CO32����SO42��������̼�ᱵ�����ᱵ���������̼�ᱵ�ܽ⣬ʣ��������ᱵ��ԭ��Һ�в�����Mg2+��Ba2+��ԭ��Һ��һ������NH4+��CO32����SO42����һ��������Mg2+��Ba2+�����ܺ���K+��Cl����
���㣺���ӵļ��飬���ӵĹ��棬���ӷ�Ӧ

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д����й��ڴ��������ǿ����ʡ�������ʺͷǵ���ʵ���ȷ�����
������ | ����� | ǿ����� | ������� | �ǵ���� | |
A | ���� | ���� | ���� | ��ˮ | �ɱ� |
B | �������� | ������Һ | ���ᱵ | ���� | ˮ |
C | ���� | Ũ���� | ������ | ˮ | ������̼ |
D | ���� | ���� | ���� | ���� | ���� |
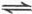 4NO��g��+6H2O��g��������Ӧ���ʷֱ���v��NH3����v��O2����v��NO����v��H2O��[mol/��L��min��]��ʾ�������й�ϵ����ȷ����
4NO��g��+6H2O��g��������Ӧ���ʷֱ���v��NH3����v��O2����v��NO����v��H2O��[mol/��L��min��]��ʾ�������й�ϵ����ȷ���� 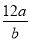
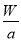 mol
mol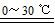 2K2FeO4+6KCl+8H2O�������йظ÷�Ӧ˵����ȷ����
2K2FeO4+6KCl+8H2O�������йظ÷�Ӧ˵����ȷ����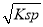 mol/L;
mol/L;