��Ŀ����
6����ͼ��һ��ҩ����ӺϳɵIJ���·��ͼ����Ӧ�����Ѿ���ȥ����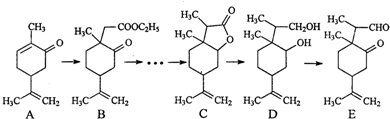
�ش��������⣺
��1���л���A�ж���ͬ���칹�壬������������������A��ͬ���칹��Ľṹ��ʽΪ
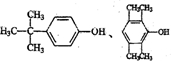 ����дһ�֣���
����дһ�֣��������ڷ����廯�������ʹFeCl3��Һ��ɫ���ۺ˴Ź���������ʾ��4�ֲ�ͬ��ѧ�������⣮
��2���л���B�����к�������������Ϊ�ʻ���ȩ����������̼ԭ����Ϊ2��
��3��C��LiBH4��ԭ���Եõ�D��C��D��ֱ����������������������ԭ��ԭ���DZ���̼̼˫���������ӳɻ�ԭ�������Ѻ�����������ԭ��Ӧ��
��4��E������������ͭ��Ӧ�Ļ�ѧ����ʽ���л���E��RCHO����ʾ��ΪRCHO+2Cu��OH��2$\stackrel{��}{��}$RCOOH+Cu2O��+2H2O��
��5����д��B��C�ĺϳ�·��ͼ��CH3I�����Լ���ѡ�����ϳ�·������ͼʾ�����£�
H2C�TCH2$\stackrel{HBr}{��}$CH3CH2Br$��_{��}^{NaOH��Һ}$CH3CH2OH
��֪����LiBH4���Խ�ȩ��ͪ�����ԭ�ɴ��������ܻ�ԭ���ᡢ�����Ρ�̼̼˫��������ֽ⣮
RCOR��$��_{THF}^{LiBH_{4}}$RCH��OH��R��R-COOR��$��_{THF}^{LiBH_{4}}$CH3CH2OH
��RCH2COOR��$��_{����}^{CH_{3}I}$RCH��CH3��COOR�䣮
���� ��1�������ڷ����廯���Ӧ���б���������ʹFeCl3��Һ��ɫ�����з��ǻ����ۺ˴Ź���������ʾ��4�ֲ�ͬ��ѧ�������⣬�ṹӦ�Գƣ�
��2������B�Ľṹ��ʽ�жϺ��еĹ����ţ�����̼ԭ�Ӽ�̼ԭ����Χ���ӵ�ԭ�ӻ�ԭ���Ÿ�����ͬ��
��3����������̼̼˫�������ӳɷ�Ӧ��
��4��E������������ͭ��Ӧ��E��ȩ�����������Ȼ���ͬʱ��������ͭ���ɣ�����Ԫ���غ���д��ѧ����ʽ��
��5��B��C��Ӧ���ڼ���������ˮ�⣬Ȼ����LiBH4��ԭ�õ��ǻ�����Ũ���������·���������Ӧ�������Ϣ�ڣ���CH3I�ڴ��������·�����Ӧ�ɵõ�C��
��� �⣺��1�������ڷ����廯���Ӧ���б���������ʹFeCl3��Һ��ɫ�����з��ǻ����ۺ˴Ź���������ʾ��4�ֲ�ͬ��ѧ�������⣬�ṹӦ�Գƣ����ܵĽṹ�� ��
��
�ʴ�Ϊ�� ��
��
��2���л���B�����к�������������Ϊ�ʻ���ȩ��������B�Ľṹ��ʽ��֪��������̼ԭ����Ϊ2���ʴ�Ϊ���ʻ���ȩ����2��
��3����������̼̼˫�������ӳɷ�Ӧ����Ϊ����̼̼˫���������ӳɻ�ԭ���������Ѻ�����������ԭ��Ӧ������LiBH4��ԭ��
�ʴ�Ϊ������̼̼˫���������ӳɻ�ԭ�������Ѻ�����������ԭ��Ӧ��
��4��E������������ͭ��Ӧ��E��ȩ�����������Ȼ���ͬʱ��������ͭ���ɣ���Ӧ�ķ���ʽΪRCHO+2Cu��OH��2$\stackrel{��}{��}$RCOOH+Cu2O��+2H2O��
�ʴ�Ϊ��RCHO+2Cu��OH��2$\stackrel{��}{��}$RCOOH+Cu2O��+2H2O��
��5��B��C��Ӧ���ڼ���������ˮ�⣬Ȼ����LiBH4��ԭ�õ��ǻ�����Ũ���������·���������Ӧ�������Ϣ�ڣ���CH3I�ڴ��������·�����Ӧ�ɵõ�C����Ӧ����Ϊ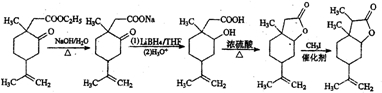 ��
��
�ʴ�Ϊ�� ��
��
���� ���⿼���л���ĺϳɣ�������ѧ���������������Ŀ��飬ע������л�������ŵ����ʣ�����ʱҪ��ϸ���⣬���������Ϣ����Ŀ�ѶȲ���

| A�� | 120�� | B�� | 104��30�� | C�� | 109��28�� | D�� | 60�� |
| A�� | ������ N-Cl���ǷǼ��Լ� | B�� | �����в����ڹ¶Ե��� | ||
| C�� | ���Ȼ������ӽṹ�������� | D�� | ��N-Cl �����ܴ����ķе�� |
| A�� | ������A��B��C��Ũ��֮��Ϊ1��2��2 | |
| B�� | ��λʱ������0.2 mol/L Bͬʱ����0.2 mol/L C | |
| C�� | ������ѹǿ����ʱ��仯 | |
| D�� | ������B��Ũ�Ȳ���ʱ��仯 |
| A�� | NH4++OH-��NH3•H2O | B�� | 2Na+2H2O��2Na++2OH-+H2�� | ||
| C�� | HCO3-+OH-��H2O+CO32- | D�� | 2Na+2H2O��2NaOH+H2�� |
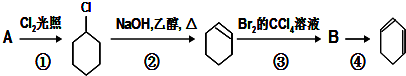
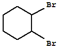 ��
�� +Cl2 $\stackrel{����}{��}$
+Cl2 $\stackrel{����}{��}$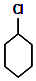 +HCl��
+HCl��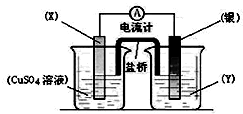 ����ͼװ��ʵ��ԭ��ط�Ӧ��Cu+2Ag+�TCu2++2Ag
����ͼװ��ʵ��ԭ��ط�Ӧ��Cu+2Ag+�TCu2++2Ag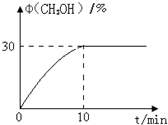 CO2����ϳɼ״��ļ������Լ������������ŷźͼ���ȼ��Σ��������Ҫ���壬��������Ҫ��Ӧ���£�CO2��g��+3H2��g���TCH3OH��g��+H2O��g�����ش��������⣺
CO2����ϳɼ״��ļ������Լ������������ŷźͼ���ȼ��Σ��������Ҫ���壬��������Ҫ��Ӧ���£�CO2��g��+3H2��g���TCH3OH��g��+H2O��g�����ش��������⣺