��Ŀ����
��14�֣���ij�ܱ������м���0.6molA��0.2molC��һ������B�������壬һ�������·�����Ӧ��������Ũ����ʱ��仯���ͼ��ʾ������t0-t1��c��B��δ����������ͼΪ��Ӧ��ϵ�з�Ӧ������ʱ��仯����������ĸ��ζ�ֻ�ı�һ���������������¶ȡ�Ũ�ȡ�ѹǿ��ÿ�θı���������ͬ������֪t3��t4��Ϊʹ�ô�����
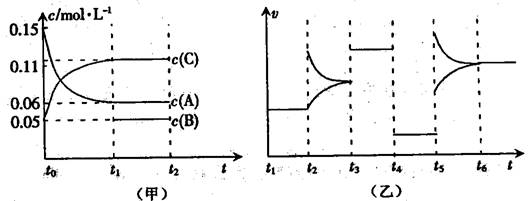
��1����t1=5s����t0��t1����CŨ�ȱ仯��ʾ�ķ�Ӧ����Ϊv(C)=________mol/(L•s)��
��2����t2��t3�Σ�C�����������С���˽�v������___v���棩���>������=����<������
��3��t4��t5�θı������Ϊ_______��B����ʼ���ʵ���Ϊ_______��
��4��t5��t6��������A�����ʵ�������С0.03mol�����˹����������������Ƚ�������ΪakJ��д���÷�Ӧ���Ȼ�ѧ����ʽ��__________________________________��
��5���÷�Ӧ�Ļ�ѧƽ�ⳣ��K�ɱ�ʾΪ_______________��[�ú�c(A) ��c(B) ��c(C)��ʽ�ӱ�ʾ]
������ͼ�в�����t1��t6 ����K�ı仯����ͼ��
��1��0.012 ��2�֣� ��2��<��2�֣�
��3����ѹ����������������� ��2�֣� 0.08mol��2�֣�
��4��3A(g) B(g)+2C(g)����H=+100a kJ/mol��2�֣�
B(g)+2C(g)����H=+100a kJ/mol��2�֣�
��5��K=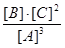 ����ͼ���2�֣�
����ͼ���2�֣�

��������

 ������ҵ��ͬ����ϰ��ϵ�д�
������ҵ��ͬ����ϰ��ϵ�д�| t | 2 |
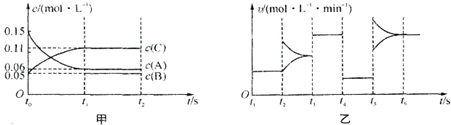
| A����t0=15 s������C��Ũ�ȱ仯��ʾ��t0��t1�ε�ƽ����Ӧ����Ϊ0.004mol?L-1?s-1 |
| B��t4��t5�θı������һ��Ϊ��Сѹǿ |
| C�����������ݻ�Ϊ2 L��B����ʼ���ʵ���Ϊ0.02 mol |
| D��t5��t6��Σ�������A�����ʵ���������0.03 mol�����˹����������������Ƚ�������Ϊa kJ���÷�Ӧ���Ȼ�ѧ����ʽΪ3A��g��?B��g��+2C��g����H=+100akJ?mol-1 |




 ��1��A����ʼ���ʵ���Ϊ
��1��A����ʼ���ʵ���Ϊ