��Ŀ����
��֪�ϳɰ���ӦΪ��N2+3H2?2NH3����һ���¶��£���2L�ܱ������У�����2molN2��5molH2��һ��������ʹ֮��Ӧ������2min��ﵽƽ��״̬�����NH3Ϊ0.4mol����
��1����N2��ʾ��2min�ڸ÷�Ӧ�ķ�Ӧ���ʣ�
��2����ʱ������ת���ʣ�
��3��ƽ��ʱNH3�����������������1λС����
��4��ƽ��ʱ�����е�ѹǿ�뿪ʼѹǿ֮�ȣ�
��5��ƽ��ʱ�����������ƽ��Ħ��������
��1����N2��ʾ��2min�ڸ÷�Ӧ�ķ�Ӧ���ʣ�
��2����ʱ������ת���ʣ�
��3��ƽ��ʱNH3�����������������1λС����
��4��ƽ��ʱ�����е�ѹǿ�뿪ʼѹǿ֮�ȣ�
��5��ƽ��ʱ�����������ƽ��Ħ��������
���㣺��ѧƽ��ļ���
ר�⣺��ѧƽ��ר��
���������ݻ�ѧƽ�������ʽ��ʽ����õ�ƽ�����ʵ�����
N2 +3H2 =2 NH3��
��ʼ����mol��2 5 0
�仯����mol��0.2 0.6 0.4
ƽ������mol��1.8 4.4 0.4
��Ϸ�Ӧ���ʡ�Ũ�ȡ�ת���ʡ�ƽ��Ħ������������㣬����ѹǿ֮�ȵ����������ʵ���֮�ȣ�
N2 +3H2 =2 NH3��
��ʼ����mol��2 5 0
�仯����mol��0.2 0.6 0.4
ƽ������mol��1.8 4.4 0.4
��Ϸ�Ӧ���ʡ�Ũ�ȡ�ת���ʡ�ƽ��Ħ������������㣬����ѹǿ֮�ȵ����������ʵ���֮�ȣ�
���
�⣺��2L�ܱ������У�����2molN2��5molH2��һ��������ʹ֮��Ӧ������2min����NH3Ϊ0.4mol����
N2 +3H2 =2 NH3��
��ʼ����mol��2 5 0
�仯����mol��0.2 0.6 0.4
ƽ������mol��1.8 4.4 0.4
��1����N2��ʾ��2min�ڸ÷�Ӧ�ķ�Ӧ����=
=0.05mol/L?min��
��N2��ʾ��2min�ڸ÷�Ӧ�ķ�Ӧ���ʷֱ���0.05mol/L?min��
��2������ת����=
��100%=12%��
��2min ��������ת����Ϊ12%��
��3��ƽ��ʱNH3���������=�������ʵ�������=
��100%=6.1%��
��ƽ��ʱNH3���������6.1%��
��4��ƽ��ʱ�����е�ѹǿ�뿪ʼѹǿ֮��=�������ʵ���֮��=��1.8+4.4+0.4������2+5��=33��35��
��ƽ��ʱ�����е�ѹǿ�뿪ʼѹǿ֮��33��35��
��5��ƽ��ʱ�����������ƽ��Ħ������=
=
=10g/mol��
��ƽ��ʱ�����������ƽ��Ħ������10g/mol��
N2 +3H2 =2 NH3��
��ʼ����mol��2 5 0
�仯����mol��0.2 0.6 0.4
ƽ������mol��1.8 4.4 0.4
��1����N2��ʾ��2min�ڸ÷�Ӧ�ķ�Ӧ����=
| ||
| 2min |
��N2��ʾ��2min�ڸ÷�Ӧ�ķ�Ӧ���ʷֱ���0.05mol/L?min��
��2������ת����=
| 0.6mol |
| 5mol |
��2min ��������ת����Ϊ12%��
��3��ƽ��ʱNH3���������=�������ʵ�������=
| 0.4mol |
| 1.8mol+4.4mol+0.4mol |
��ƽ��ʱNH3���������6.1%��
��4��ƽ��ʱ�����е�ѹǿ�뿪ʼѹǿ֮��=�������ʵ���֮��=��1.8+4.4+0.4������2+5��=33��35��
��ƽ��ʱ�����е�ѹǿ�뿪ʼѹǿ֮��33��35��
��5��ƽ��ʱ�����������ƽ��Ħ������=
| m |
| n |
| 1.8mol��28g/mol+4.4mol��2g/mol+0.4mol��17g/mol |
| 1.8mol+4.4mol+0.4mol |
��ƽ��ʱ�����������ƽ��Ħ������10g/mol��
���������⿼���˻�ѧƽ��ļ���Ӧ�ã�ƽ������ʽ���㷽���ǽ���ؼ�����Ŀ�ϼ�

��ϰ��ϵ�д�
�����Ŀ
����˵����ȷ���ǣ�NAΪ�����ӵ�������ֵ��������
| A��124g P4���е�P-P���ĸ���Ϊ4NA |
| B��12gʯī�к���C-C���ĸ���Ϊ1.5NA |
| C��12g���ʯ�к���C-C���ĸ���Ϊ2NA |
| D��60g SiO2�к�Si-O���ĸ���Ϊ2NA |
��25��ʱ���ܱ�������X��Y��Z��������ij�ʼŨ�Ⱥ�ƽ��Ũ�����±���
����˵����ȷ���ǣ�������
| ���� | X | Y | Z |
| ��ʼŨ�ȣ�mol/L�� | 0.1 | 0.2 | 0 |
| ƽ��Ũ�ȣ�mol/L�� | 0.05 | 0.05 | 0.1 |
| A����Ӧ�ﵽƽ��ʱ��X��ת����Ϊ50% |
| B����Ӧ�ﵽƽ��ʱ��Y��ת����Ϊ25% |
| C��������������ʱ������Y�����ʵ�����ƽ��������Ӧ�������ƶ���ƽ�ⳣ��K��� |
| D��������������ʱ�����¶����ߣ�ƽ�ⳣ��K����˵���÷�Ӧ����Ӧ��H��0 |
 ��Ҫ�����
��Ҫ�����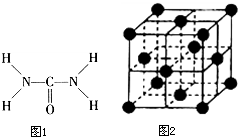 ��ͼ1����֪���صĽṹʽΪ�����ؿ��������л����ʣ���Ҫ�������������ᣬ�����غ���������ѧʽΪ[Fe��H2NCONH2��6]
��ͼ1����֪���صĽṹʽΪ�����ؿ��������л����ʣ���Ҫ�������������ᣬ�����غ���������ѧʽΪ[Fe��H2NCONH2��6]