��Ŀ����
16��Fe2+��Fe3+��O22-��CN-��F-���л����ӵ��γɵĻ�������й㷺��Ӧ�ã�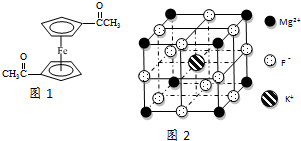
��1��C��N��Oԭ�ӵĵ�һ�������ɴ�С��˳����N��O��C��
��2��Fe2+��̬��������Ų�ʽΪ1s22s22p63s23p63d6��
��3����������ï���dz������Ϳ��������ṹ��ͼ1��ʾ����������̼ԭ�ӵ��ӻ���ʽ��sp2��sp3��
��4�������K3Fe��CN��6�����ڵ��Ӵ������������������廥Ϊ�ȵ������һ�ַ��ӵĻ�ѧʽΪCO��N2����֪��CN��2��ֱ���ͷ��ӣ������жԳ��ԣ���CN��2�Цм��ͦҼ��ĸ�����Ϊ4��3��
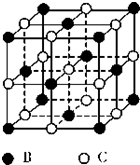
��5��F-��������Fe3+�γ�[FeF6]3-����������Mg2+��K+�γ�һ��������ϵ�����Ӿ��壬�˾���Ӧ���ڼ������ṹ��ͼ2��ʾ���þ���Ļ�ѧʽΪKMgF3��
���� ��1��ͬһ����Ԫ�أ�Ԫ�صĵ�һ����������ԭ������������������ƣ�����IIA�塢��VA��Ԫ�ص�һ�����ܴ���������Ԫ�أ�
��2���ȸ��ݹ���ԭ��д��Fe��������Ų�ʽ��Ȼ��д��Fe2+��̬��������Ų�ʽ��
��3�����ݷ�����̼ԭ�ӵijɼ�������ж�̼ԭ�ӵ��ӻ���ʽ��
��4��ԭ�Ӹ�����ͬ�Ҽ۵�������ͬ�����ǵȵ����壻��CN��2��ֱ���ͷ��ӣ����жԳ��ԣ��ṹʽΪ��N��C-C��N��1��������1���Ҽ���1�������к���1���Ҽ���2���м���
��5�����ݾ�̯�����㾧����F-��Mg2+��K+������Ŀȷ����ѧʽ��
��� �⣺��1��ͬһ����Ԫ�أ�Ԫ�صĵ�һ����������ԭ������������������ƣ�����IIA�塢��VA��Ԫ�ص�һ�����ܴ���������Ԫ�أ�����C��N��O����Ԫ�صĵ�һ���������ɴ�С��˳����N��O��C��
�ʴ�Ϊ��N��O��C��
��2������26��Ԫ�أ���ԭ�Ӻ�����26�����ӣ���������Ų�ʽΪ��1s22s22p63s23p63d64s2��ʧȥ2�����ӱ��Fe2+�����̬��������Ų�ʽ��1s22s22p63s23p63d6��
�ʴ�Ϊ��1s22s22p63s23p63d6��
��3��������Cԭ����̼̼˫����̼̼������ʽ���ڣ���̼ԭ�ӵ��ӻ���ʽ��sp2��sp3��
�ʴ�Ϊ��sp2��sp3��
��4��CN-�к�������ԭ���Ҽ۵�������10��������CN-��Ϊ�ȵ������һ�ַ��ӵĻ�ѧʽ��CO��N2��
��CN��2��ֱ���ͷ��ӣ����жԳ��ԣ��ṹʽΪ��N��C-C��N��1��������1���Ҽ���1�������к���1���Ҽ���2���м������ԣ�CN��2�Цм��ͦҼ��ĸ�����Ϊ4��3��
�ʴ�Ϊ��CO��N2��4��3��
��5��������F-������Ŀ=12��$\frac{1}{4}$=3��Mg2+������Ŀ=8��$\frac{1}{8}$=1��K+������Ŀ=1���ʾ��廯ѧʽΪ��KMgF3��
�ʴ�Ϊ��KMgF3��
���� ���⿼�������ʽṹ�����ʣ��漰��һ�����ܡ���������Ų����ӻ�������ȵ����塢��������ȣ��Ѷ��еȣ�ע�����þ�̯�����о����йؼ��㣮

| A�� | ���ᶡ�� | B�� | �������� | C�� | ������� | D�� | �������� |
��������
| A�� | X�ֱ����������Ԫ�ؾ����γ�����2�ֻ����� | |
| B�� | M�ֱ��w��X��Y��Z�γɻ����������ͬ���ϼ� | |
| C�� | M��X��Z����Ԫ����ɵĻ����ﺬ�����Ӽ� | |
| D�� | M��X��Y��ɻ������Y��Z��ɻ�������������Ļ�ѧ��������ȫ��ͬ |
| A�� | Al3+��Ba2+��Cl-��NO3- | B�� | K+��OH-��Cl-��SO42- | ||
| C�� | Ca2+��Mg2+��NO3-��HCO3- | D�� | Na+��Cl-��CO32-��SO32- |
 ��A��B��C��D��E����ԭ���������������Ԫ�أ�ԭ��������������30����A�Ļ�̬ԭ��2p�ܼ���3�������ӣ�C�ĺ���û�гɵ����ӣ�B��C��Ԫ���γɵĻ����ᄃ���ṹ��ͼ��ʾ��D��3p�ܼ���1�������ӣ�����B�����������ڣ�EԪ ���ڵؿ��к����ӵ���λ����
��A��B��C��D��E����ԭ���������������Ԫ�أ�ԭ��������������30����A�Ļ�̬ԭ��2p�ܼ���3�������ӣ�C�ĺ���û�гɵ����ӣ�B��C��Ԫ���γɵĻ����ᄃ���ṹ��ͼ��ʾ��D��3p�ܼ���1�������ӣ�����B�����������ڣ�EԪ ���ڵؿ��к����ӵ���λ����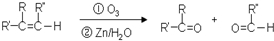 �л���A�Ľṹ��ʽΪ
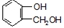
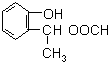
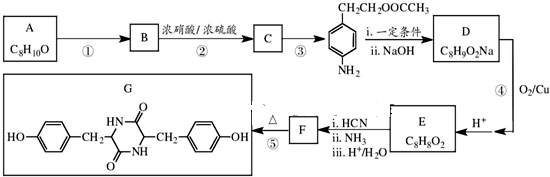
�л���A�Ľṹ��ʽΪ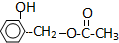 ��G�ķ���ʽΪC7H12O������A��H��Ϊ�л����ת����ϵ���£�
��G�ķ���ʽΪC7H12O������A��H��Ϊ�л����ת����ϵ���£�
 ���Է�����ȥ��Ӧ
���Է�����ȥ��Ӧ ����F��G�ķ�Ӧ����Ϊ��ȥ��Ӧ��
����F��G�ķ�Ӧ����Ϊ��ȥ��Ӧ��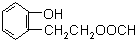 ��
�� �������������칹����
�������������칹���� ��
�� ��
�� 
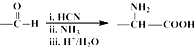 ��NO2$\stackrel{Fe?HCl}{��}$NH2
��NO2$\stackrel{Fe?HCl}{��}$NH2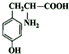 ��
��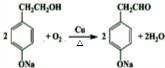 ��
��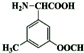 ��
��