��Ŀ����
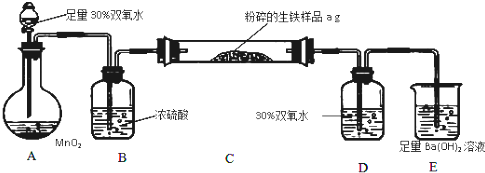
�����г����⣬����������Ԫ�أ���̼Ԫ�غ���Ԫ�أ�����̼��Ҫ��̼��������̬���ڣ���ʹ�������ܼ�Ӳ���࣬������������;����һ���������ֵ�ԭ�ϣ�ij��ȤС����ư���ͼ��ʾ��ʵ��װ�òⶨ�����еĺ�̼���ͺ�������
��ش��������⣺
��1���������������к�Ԫ�أ���ʹ���������ȴ��ԣ���Ԫ�������������п��ܴ��ڵļ�̬�� ��������ȷ�𰸵ı�ţ�
A��-2 B.0 C��+4 D��+6
��2����Ҫ���ȵ�װ���� ����װ�ñ�ţ���
��3������Dװ�ã��������ⶨ��������̼�� ���ƫ�ߡ���ƫ�͡�����Ӱ�족��
��4���ձ�E�з�����Ӧ�����ӷ�Ӧ����ʽΪ
��5����C���е���Ʒ��ַ�Ӧ��E�����յij������ˡ�ϴ�ӡ������������Ϊb�ˣ�����������̼Ԫ�ص���������Ϊ ��
��6����ͬѧ���Ӧ����C��D֮���һ��װ�ã���װ�õ������� ������ͬѧ�����D��E֮��ҲӦ���ٽ�һ��װ�ã���װ�õ������� ��
��7����Ӧ��ɺ������������к�����Ԫ�أ�������Ƽ�ʵ�鷽����д��ʵ�鲽�衢���� ��

��ش��������⣺
��1���������������к�Ԫ�أ���ʹ���������ȴ��ԣ���Ԫ�������������п��ܴ��ڵļ�̬��
A��-2 B.0 C��+4 D��+6
��2����Ҫ���ȵ�װ����
��3������Dװ�ã��������ⶨ��������̼��
��4���ձ�E�з�����Ӧ�����ӷ�Ӧ����ʽΪ
��5����C���е���Ʒ��ַ�Ӧ��E�����յij������ˡ�ϴ�ӡ������������Ϊb�ˣ�����������̼Ԫ�ص���������Ϊ
��6����ͬѧ���Ӧ����C��D֮���һ��װ�ã���װ�õ�������
��7����Ӧ��ɺ������������к�����Ԫ�أ�������Ƽ�ʵ�鷽����д��ʵ�鲽�衢����
���㣺̽�����ʵ���ɻ�������ʵĺ���
ר�⣺ʵ��̽�������ݴ�����
��������1�����Ļ��ϼ�һ��Ϊ���ۣ���̼�ķǽ���С����ķǽ����ԣ����Sһ����ʾ���ۣ�
��2��Aװ����˫��ˮ�ڶ�������Ϊ����������������������������ȣ���װ��C�е�̼Ԫ������Ϊ������̼����Ԫ������Ϊ����������Ҫ���ȣ�
��3�����û��˫��ˮ����������������Ҳ��������������Ӧ���ɳ�����ʹ̼�İٷֺ���ƫ�ߣ�
��4��װ��A�й��������ڶ�������Ϊ����������������������ͨ��Ũ��������װ��C�е�̼Ԫ������Ϊ������̼����Ԫ������Ϊ����������������ͨ������������Һ�����������������գ�������̼ͨ��Eװ������̼�ᱵ������
��5����E�г���������Ϊbg����̼�ᱵ������������̼Ԫ���غ��֪̼�ᱵ�����ʵ����������̼�����ʵ�����ȣ��ݴ˼���������̼������������������̼������Ϊ��������������������̼����������Ϊ��
��6�����ݶ�������������ˮ��������C��D֮���һ��������װ�ã����������̼����Ҫ����������������������D��E֮��ҲӦ���ٽ�һ����SO2�Ѿ�������װ�ã�
��7��������˫��ˮ���ն��������ҽ������������������������һ������D�к�����������ɼ��������к�����Ԫ�أ��ݴ˷�����
��2��Aװ����˫��ˮ�ڶ�������Ϊ����������������������������ȣ���װ��C�е�̼Ԫ������Ϊ������̼����Ԫ������Ϊ����������Ҫ���ȣ�
��3�����û��˫��ˮ����������������Ҳ��������������Ӧ���ɳ�����ʹ̼�İٷֺ���ƫ�ߣ�
��4��װ��A�й��������ڶ�������Ϊ����������������������ͨ��Ũ��������װ��C�е�̼Ԫ������Ϊ������̼����Ԫ������Ϊ����������������ͨ������������Һ�����������������գ�������̼ͨ��Eװ������̼�ᱵ������
��5����E�г���������Ϊbg����̼�ᱵ������������̼Ԫ���غ��֪̼�ᱵ�����ʵ����������̼�����ʵ�����ȣ��ݴ˼���������̼������������������̼������Ϊ��������������������̼����������Ϊ��
��6�����ݶ�������������ˮ��������C��D֮���һ��������װ�ã����������̼����Ҫ����������������������D��E֮��ҲӦ���ٽ�һ����SO2�Ѿ�������װ�ã�
��7��������˫��ˮ���ն��������ҽ������������������������һ������D�к�����������ɼ��������к�����Ԫ�أ��ݴ˷�����
���
�⣺��1�����Ļ��ϼ�һ��Ϊ���ۣ���̼�ķǽ���С����ķǽ����ԣ����Sһ����ʾ���ۣ���ѡ��A��
��2��Aװ����˫��ˮ�ڶ�������Ϊ����������������������������ȣ���װ��C�е�̼Ԫ������Ϊ������̼����Ԫ������Ϊ����������Ҫ���ȣ���ѡ��C��
��3�����û��˫��ˮ����������������Ҳ��������������Ӧ���ɳ�����ʹ̼�İٷֺ���ƫ�ߣ���D��30% ˫��ˮ������������SO2���壬�ʴ�Ϊ��ƫ�ߣ�
��4��������̼������������Ӧ����̼�ᱵ��ˮ����Ӧ���ӷ���ʽΪCO2+Ba2++2OH-=BaCO3��+H2O���ʴ�Ϊ��CO2+Ba2++2OH-=BaCO3��+H2O��
��5����E�еõ�bg̼�ᱵ������̼Ԫ���غ��֪̼�ᱵ�����ʵ����������̼�����ʵ�����ȣ���������̼������Ϊ
��12g/mol=
g��������̼����������Ϊ
��100%=
%���ʴ�Ϊ��
%��
��6�����ݶ�������������ˮ��������C��D֮���һ��������װ�ã����������̼����Ҫ����������������������D��E֮��ҲӦ���ٽ�һ����SO2�Ѿ�������װ�ã��ʴ�Ϊ����ֹD��Һ�嵹���� ����SO2�Ѿ�������
��7����Ϊ��˫��ˮ���ն��������ҽ���������������������������D�к�����������ɼ��������к�����Ԫ�أ�����Ϊ��ȡ����Dƿ��Һ���Թ��У��μ�����ʹ���ữ���ٵ���BaCl2��Һ�������ְ�ɫ������˵�������к�����Ԫ�أ�
�ʴ�Ϊ��ȡ����Dƿ��Һ���Թ��У��μ�����ʹ���ữ���ٵ���BaCl2��Һ�������ְ�ɫ������˵�������к�����Ԫ�أ�
��2��Aװ����˫��ˮ�ڶ�������Ϊ����������������������������ȣ���װ��C�е�̼Ԫ������Ϊ������̼����Ԫ������Ϊ����������Ҫ���ȣ���ѡ��C��
��3�����û��˫��ˮ����������������Ҳ��������������Ӧ���ɳ�����ʹ̼�İٷֺ���ƫ�ߣ���D��30% ˫��ˮ������������SO2���壬�ʴ�Ϊ��ƫ�ߣ�
��4��������̼������������Ӧ����̼�ᱵ��ˮ����Ӧ���ӷ���ʽΪCO2+Ba2++2OH-=BaCO3��+H2O���ʴ�Ϊ��CO2+Ba2++2OH-=BaCO3��+H2O��
��5����E�еõ�bg̼�ᱵ������̼Ԫ���غ��֪̼�ᱵ�����ʵ����������̼�����ʵ�����ȣ���������̼������Ϊ
| bg |
| 197g/mol |
| 12b |
| 197 |
| ||
| a |
| 1200b |
| 197a |
| 1200b |
| 197a |
��6�����ݶ�������������ˮ��������C��D֮���һ��������װ�ã����������̼����Ҫ����������������������D��E֮��ҲӦ���ٽ�һ����SO2�Ѿ�������װ�ã��ʴ�Ϊ����ֹD��Һ�嵹���� ����SO2�Ѿ�������
��7����Ϊ��˫��ˮ���ն��������ҽ���������������������������D�к�����������ɼ��������к�����Ԫ�أ�����Ϊ��ȡ����Dƿ��Һ���Թ��У��μ�����ʹ���ữ���ٵ���BaCl2��Һ�������ְ�ɫ������˵�������к�����Ԫ�أ�
�ʴ�Ϊ��ȡ����Dƿ��Һ���Թ��У��μ�����ʹ���ữ���ٵ���BaCl2��Һ�������ְ�ɫ������˵�������к�����Ԫ�أ�
���������⿼���ʵ�鷽�������װ�õ����⡢ʵ�������������ѧ����ȣ��Ѷ��еȣ�����ʵ��ԭ���ǽ���Ĺؼ����Ƕ�֪ʶ���ۺ����ã���Ҫѧ���߱���ʵ�Ļ���֪ʶ������֪ʶ�������⡢����������������������Ͷ�����̼������������������������ƣ���˸߿��ж�������������ļ���ıȽ϶࣬�������������Ӧ������һ���㣬��������������ʼ��Կ��飮

��ϰ��ϵ�д�
�����Ŀ
�������л���Ӧ���͵���ʶ�У�������ǣ�������
A�� +HO-NO2 +HO-NO2
 +H2O��ȡ����Ӧ +H2O��ȡ����Ӧ | |||
| B��CH2=CH2+Br2��CH2Br-CH2Br���ӳɷ�Ӧ | |||
C��2CH3CH2OH+O2
| |||
D��CH3COOH+CH3CH2OH
|
�����йع�ҵ����������ȷ���ǣ�������
| A����⾫��ͭʱ��ͬһʱ���������ܽ�ͭ����������������ͭ������С |
| B���ϳɰ���ҵ�У���NH3��ʱҺ�����������ڼӿ췴Ӧ���� |
| C�����Ṥҵ�У����ó�ѹ������ԭ���Ǵ������´���������� |
| D�������Ƽ��ǽ�CO2��NH3�Ⱥ�ͨ�뱥���Ȼ�����Һ�е�̼�����ƹ��壬��������̼���ƹ��� |
�����йط�ɢϵ��˵����ȷ���ǣ�������
| A�������ܷ��������ЧӦ����ɢϵ��Ϊ��Һ����Һ�ͽ��� |
| B����Fe��OH��3�����м����������ܽ� |
| C����ѪҺ����ԭ��ͬ��Һ�й�������Ĺ���ԭ������ |
| D�����������ʿ�ʹ����۳��⣬�����ִ��෴��ɽ����Ľ�����Ҳ�ܷ����۳� |
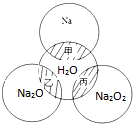 ��ͼ��ʾ����ԲȦ�ཻ����Ӱ���ּ��ұ���ʾԲȦ�������������Ӧ���Ƽ�������������ʵ�����Ϊ0.1mol��ˮ������Ϊ100g������˵����ȷ���ǣ�������
��ͼ��ʾ����ԲȦ�ཻ����Ӱ���ּ��ұ���ʾԲȦ�������������Ӧ���Ƽ�������������ʵ�����Ϊ0.1mol��ˮ������Ϊ100g������˵����ȷ���ǣ�������| A�����ұ���Ӧ������������ԭ��Ӧ |
| B������Ӧ�е���������Na2O2����ԭ����H20 |
| C������Ӧ��������1molNa2O2����ת��2moL�ĵ��� |
| D�����ұ���Ӧ��������Һ�����������ֱ�Ϊ��1����2����3����2��1����2=��3 |