��Ŀ����
17��A��B��C��D��E�����ֶ����ڵ�����Ԫ�أ����ǵ�ԭ��������������A��D������C��ԭ�Ӹ�����Ϊ1��1��2��1�γɻ����A��B��ɵ���̬�����������B������������ˮ���ﷴӦ����һ����M��E��C��������������ͬ����1��M�к��еĻ�ѧ�������Ӽ����ۼ���EԪ�������ڱ��е�λ���ǵ������ڢ�A�壮
��2����100mL 0.1mol•L-1 D������������Һ��ͨ��224mL A2E����״���£����壬������Һ�ʼ��ԣ�ԭ�������⻯����Һˮ��̶ȴ��ڵ���̶ȣ���Һ������Ũ���ɴ�С��˳��Ϊc��Na+����c��HS-����c��OH-����c��H+����c��S2-����
��3��B��C���γ��⻯����ȶ�����ǿ������˳����H2O��NH3�������Ļ�ѧʽ����
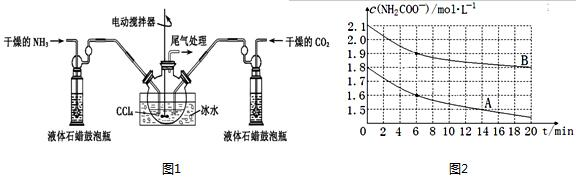
��4����������Ѱ����ʵĴ����͵缫���ϣ���A2��B2Ϊ�缫��Ӧ���HCl-NH4Cl��ҺΪ�������Һ��������ȼ�ϵ�أ�д���õ�ص������缫��Ӧʽ��N2+8H++6e-�T2NH4+���ŵ�ʱ��Һ�е�H+�����������������������
���� A��B��C��D��E���ֶ�����Ԫ�أ����ǵ�ԭ��������������A��D������C��ԭ�Ӹ�����Ϊ1��1�Ĺ��������2��1�γɻ�����ˮ����AΪHԪ�أ�CԪ������Ԫ�أ�D����Ԫ�أ�A��B��ɵ���̬�����������B������������ˮ���ﷴӦ����һ����M����BΪNԪ�أ�E��C��������������ͬ����E����Ԫ�أ��ɴ˷������
��� �⣺��1��������к��еĻ�ѧ�������Ӽ����ۼ�����Ԫ�������ڱ��е�λ���ǵ������ڢ�A���ʴ�Ϊ�����Ӽ����ۼ����������ڢ�A�壻
��2����100mL 0.1mol•L-1����������Һ��ͨ��224mLH2S����״���£����壬����ǡ����ȫ��Ӧ�������⻯�ƣ����⻯����Һˮ��̶ȴ��ڵ��룬������Һ�ʼ��ԣ�ˮ��̶ȴ��ڵ���̶ȣ�c��OH-����c��H+������������Ũ�ȹ�ϵΪ��c��Na+����c��HS-����c��OH-����c��H+����c��S2-����
�ʴ�Ϊ�����⻯����Һˮ��̶ȴ��ڵ���̶ȣ�c��Na+����c��HS-����c��OH-����c��H+����c��S2-����
��3���ǽ�����Խǿ�⻯��Խ�ȶ�������N��O���γ��⻯����ȶ�����ǿ������˳���ǣ�H2O��NH3���ʴ�Ϊ��H2O��NH3��
��4����N2��H2Ϊ�缫��Ӧ���HCl-NH4Cl��ҺΪ�������Һ��������ȼ�ϵ�أ�����������ԭ��Ӧ�������������õ����ӣ���������������NH4+�������缫��ӦΪN2+8H++6e-�T2NH4+��ԭ������������������ƶ����ʴ�Ϊ��N2+8H++6e-�T2NH4+������
���� ���⿼����Ԫ�ص��ƶϡ��ε�ˮ���Լ�����Ũ�ȴ�С�Ƚϡ�ԭ��أ���Ŀ�Ѷ��еȣ�ע���������ʽ�δ��ڵ����ˮ���������̣�

| A�� | Na2SO4 | B�� | NaOH | C�� | HCl | D�� | H2SO4 |
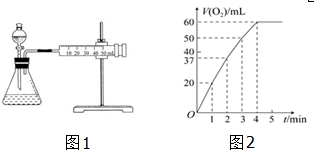
| t/min | 0 | 2 | 4 | 6 | 8 |
| V��O2��/mL | 0 | 9.9 | 17.2 | 22.4 | 26.5 |
��1��6min�ڹ�������ķֽ����ʣ�����ȷ��0.01��
��2��6min�ڹ�������ֽ��ת���ʣ�
��3��6min�ڹ�����������ʵ���Ũ�ȣ�
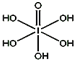 ±��Ԫ�صĵ��ʺͻ�����ܶ࣬���ǿ���������ѧ���ʽṹ�����ʵ����֪ʶȥ��ʶ���������ǣ�
±��Ԫ�صĵ��ʺͻ�����ܶ࣬���ǿ���������ѧ���ʽṹ�����ʵ����֪ʶȥ��ʶ���������ǣ���1���ڲ�̫ϡ����Һ�У���������Զ����ӵϣ�HF��2��ʽ���ڵģ�ʹ�������ӵϵ��������������
��2��������±��ṩ�ĵ�һ�����������жϣ����п������ɽ��ȶ��ĵ��������ӵ�±��ԭ����I��
| �� | �� | �� | �� | |
| ��һ�����ܣ�kJ/mol�� | 1681 | 1251 | 1140 | 1008 |
��4������ˮ�е��ܽ����ȻС�����ڵ⻯����Һ���ܽ��ȴ������������������Һ�з������з�ӦI-+I2=I3-��I3-���ӵ�����ԭ����Χ�Ҽ����ӶԶ���Ϊ2����KI3���Ƶģ�����CsICl2�ȣ���֪CsICl2���ȶ��������ֽ⣬���������ɾ����ܸ�������ʣ�����������Aʽ������
A��CsICl2=CsCl+ICl B��CsICl2=CsI+Cl2
��5����֪CaF2������ܶ�Ϊ��g/cm3��NAΪ�����ӵ�������CaF2�����ı߳�Ϊa pm����CaF2����Է����������Ա�ʾΪ$\frac{{a}^{3}���ѡ�1{0}^{-30}��{N}_{A}}{4}$���ú�a��ʽ�ӱ�ʾ����
| A�� | 1��1 | B�� | 2��1 | C�� | 3��1 | D�� | 4��1 |
| A�� | ������ʳ��ˮ����ʢ�е�ʯ����ƿ������Ȳ | |
| B�� | ����ȩ����������Һ�У�������������� | |
| C�� | ���Ҵ���3mol•L-1��H2SO4��Ϲ��ȵ�170����������ϩ | |
| D�� | ���ȷ��м���NaOH��Һ�����ȳ�ַ�Ӧ���ټ���AgNO3��Һ�����۲쵽��ɫ��������֤���ȷ��к�����Ԫ�� |