��Ŀ����
��֬������͵������������еĻ����л���ѧ���ʣ�
����ij��Ȼ��֬�������ʽΪC57H106O6��1mol����֬ˮ��ɵõ�1mol���͡�1mol������֬����A��2molһ��ֱ������֬���ᣮ���ⶨA����Է�������Ϊ280��ԭ�Ӹ�����ΪC��H��O=9��16��1��
��֪��RCH=CHR�������KMnO4��Һ���Ⱥ��ữ������˫�������������RCH=CHR��
RCOOH+R��COOH�������ø÷�Ӧ�IJ��ﷴ�ƺ�̼̼˫��������Ľṹ���ڴ��������£�1mol������֬����A��1mol H2��Ӧ�����õ�B��C�Ļ���B��C��Ϊͬ���칹�壮��B��C�Ļ���������KMnO4��Һ�����ữ�õ��������ֲ��
HOOC-��CH2��10-COOH��CH3-��CH2��7-COOH��HOOC-��CH2��7-COOH��CH3-��CH2��4-COOH��
��1��д��A�ķ���ʽ ��
��2��д��B��C�Ľṹ��ʽ�ֱ��� �� ��
�����Ǧ�-�����ᣬ��C��54.8%��H��5.58%��N��7.11%�����ຬO���ҵ���Է�������������200���ҷ����в����ڼ���-CH3��������FeCl3��Һ������ɫ��Ӧ��1mol���������3mol NaOH��ȫ��Ӧ��
��3���л����ҵ���Է�������Ϊ ��
��4��д�����������������л����ҵ���һ�ֵĽṹ��ʽ ��
��Ϊij�ǣ�ȼ�ձ�����O2������CO2��H2O�����ʵ�������ȣ�������Է��������������ʽ��Է���������5����0.1mol�����ܻ�ԭ21.6g����0.1mol��������24g���ᷢ��������Ӧ��
��5���������ʽ ��
��6����������ֱ�����ӣ�д����������Һ��Ӧ�Ļ�ѧ����ʽ ��
����ij��Ȼ��֬�������ʽΪC57H106O6��1mol����֬ˮ��ɵõ�1mol���͡�1mol������֬����A��2molһ��ֱ������֬���ᣮ���ⶨA����Է�������Ϊ280��ԭ�Ӹ�����ΪC��H��O=9��16��1��
��֪��RCH=CHR�������KMnO4��Һ���Ⱥ��ữ������˫�������������RCH=CHR��
| �ټ���KMnO4��Һ���� |
| ���ữ |
HOOC-��CH2��10-COOH��CH3-��CH2��7-COOH��HOOC-��CH2��7-COOH��CH3-��CH2��4-COOH��
��1��д��A�ķ���ʽ
��2��д��B��C�Ľṹ��ʽ�ֱ���
�����Ǧ�-�����ᣬ��C��54.8%��H��5.58%��N��7.11%�����ຬO���ҵ���Է�������������200���ҷ����в����ڼ���-CH3��������FeCl3��Һ������ɫ��Ӧ��1mol���������3mol NaOH��ȫ��Ӧ��
��3���л����ҵ���Է�������Ϊ
��4��д�����������������л����ҵ���һ�ֵĽṹ��ʽ
��Ϊij�ǣ�ȼ�ձ�����O2������CO2��H2O�����ʵ�������ȣ�������Է��������������ʽ��Է���������5����0.1mol�����ܻ�ԭ21.6g����0.1mol��������24g���ᷢ��������Ӧ��
��5���������ʽ
��6����������ֱ�����ӣ�д����������Һ��Ӧ�Ļ�ѧ����ʽ
���㣺�л�����ƶ�,�й��л������ʽȷ���ļ���
ר�⣺������,�л���Ļ�ѧ���ʼ��ƶ�
��������1����ˮ����Ա�ʾ�ɣ�C57H106O6+3H2O��C3H8O3�����ͣ�+A+2X��A����Է�������Ϊ280��ԭ�Ӹ�����ΪC��H��O=9��16��1����A�ķ���ʽΪC9nH16nOn��n=
=2������A�ķ���ʽΪ��C18H32O2������ԭ���غ�֪����ֱ������֬����ķ���ʽΪ��C18H36O2���ṹ��ʽΪ��CH3-��CH2��16-COOH��
��2��������ɷ�Ӧԭ������Ӧ�����ж�B��C�Ľṹ��ʽ��
��3���л������к�C��54.8%��H��5.58%��N��7.11%�����ຬO����O����������Ϊ1-54.8%-5.58%-7.11=32.51%�������л�����N��C����N��H����N��O����N��N��=
��
��
��
��9��11��4��1���������ʽΪC9H11NO4��ʽ��Ϊ9��12+11+14+4��16=197��200���������⣬���ҷ���ʽΪC9H11NO4��
��4�����Ǧ�-�����ᣬ�����в����ڼ���-CH3��������FeCl3��Һ������ɫ��Ӧ��˵�����з��ǻ���1mol A�������3molNaOH��ȫ��Ӧ��˵�������г����а������Ȼ�֮��Ӧ����2�����ǻ���
��5���ݡ����ȼ��ij�ǣ����ĵ����������ɵ�CO2��H2O�����ʵ�������ȡ��ɵó�ʵ��ʽΪCH2O��
��6�����������Ϣ�ж��京�еĹ��������༰��Ŀ��Ȼ��ȷ����Ľṹ��ʽ�����д������������Һ��Ӧ�Ļ�ѧ����ʽ��
| 280 |
| 12��9+1��16+16��1 |
��2��������ɷ�Ӧԭ������Ӧ�����ж�B��C�Ľṹ��ʽ��
��3���л������к�C��54.8%��H��5.58%��N��7.11%�����ຬO����O����������Ϊ1-54.8%-5.58%-7.11=32.51%�������л�����N��C����N��H����N��O����N��N��=
| 54.8% |
| 12 |
| 5.58% |
| 1 |
| 32.51% |
| 16 |
| 7.11% |
| 14 |
��4�����Ǧ�-�����ᣬ�����в����ڼ���-CH3��������FeCl3��Һ������ɫ��Ӧ��˵�����з��ǻ���1mol A�������3molNaOH��ȫ��Ӧ��˵�������г����а������Ȼ�֮��Ӧ����2�����ǻ���
��5���ݡ����ȼ��ij�ǣ����ĵ����������ɵ�CO2��H2O�����ʵ�������ȡ��ɵó�ʵ��ʽΪCH2O��
��6�����������Ϣ�ж��京�еĹ��������༰��Ŀ��Ȼ��ȷ����Ľṹ��ʽ�����д������������Һ��Ӧ�Ļ�ѧ����ʽ��
���
�⣺��1����ˮ����Ա�ʾ�ɣ�C57H106O6+3H2O��C3H8O3�����ͣ�+A+2X��A����Է�������Ϊ280��ԭ�Ӹ�����ΪC��H��O=9��16��1����A�ķ���ʽΪC9nH16nOn��n=
=2������A�ķ���ʽΪ��C18H32O2������ԭ���غ�֪����ֱ������֬����ķ���ʽΪ��C18H36O2���ṹ��ʽΪ��CH3-��CH2��16-COOH��
�ʴ�Ϊ��C18H32O2��
��2����B��C�Ļ���������KMnO4��Һ�����ữ�õ��������ֲ��HOOC-��CH2��10-COOH��CH3-��CH2��7-COOH��HOOC-��CH2��7-COOH��CH3-��CH2��4-COOH�����ݷ�Ӧԭ����RCH=CHR��
RCOOH+R��COOH����B��C��Ϊͬ���칹���֪��B��C�к��е�Cԭ����һ����ȣ�����Խ��ֽ������ࣺHOOC-��CH2��10-COOH��CH3-��CH2��4-COOH��CH3-��CH2��7-COOH��HOOC-��CH2��7-COOH�ֱ�ΪB��C�е�һ�������Ը�����ط�Ӧ���ɵIJ����B��C�Ľṹ��ʽΪ��CH3��CH2��7-CH=CH-��CH2��7-COOH��CH3��CH2��4-CH=CH-��CH2��10-COOH��
�ʴ�Ϊ��CH3��CH2��7-CH=CH-��CH2��7-COOH��CH3��CH2��4-CH=CH-��CH2��10-COOH��
��3��ij�л���A��C��54.8%��H��5.58%��N��7.11%�����ຬO����O����������Ϊ1-54.8%-5.58%-7.11=32.51%�������л�����N��C����N��H����N��O����N��N��=
��
��
��
��9��11��4��1�������ʽΪC9H11NO4��ʽ��Ϊ9��12+11+14+4��16=197��200���������⣬���ҵķ���ʽΪ��C9H11NO4����Է�������Ϊ197��
�ʴ�Ϊ��197��
��4�����Ǧ�-�����ᣬ�����в����ڼ���-CH3��������FeCl3��Һ������ɫ��Ӧ��˵�����з��ǻ���1mol ���������3molNaOH��ȫ��Ӧ��˵�������г����а������Ȼ�֮��Ӧ����2�����ǻ������ܵĽṹ�� �ȣ���Ӧ��ͬ���칹����Ҫ�����ڱ������ǻ���λ�ã����ǻ�����ʱ����2��ͬ���칹�壬���ǻ����ʱ����3��ͬ���칹�壬���ǻ����ʱ����һ�ֽṹ������6��ͬ���칹�壬
�ȣ���Ӧ��ͬ���칹����Ҫ�����ڱ������ǻ���λ�ã����ǻ�����ʱ����2��ͬ���칹�壬���ǻ����ʱ����3��ͬ���칹�壬���ǻ����ʱ����һ�ֽṹ������6��ͬ���칹�壬
�ʴ�Ϊ�� ��
��
��5����Ϊ����ȼ�����ĵ����������ɵĶ�����̼��ˮ�����ʵ�������ȣ�
���Ը��Ƿ�����C��H��O����ԭ�����ʵ���֮��Ϊ��1��2����2+1-2��=1��2��1���ʸ��ǵ����ʽΪ��CH2O��
�ʴ�Ϊ��CH2O��
��6��������Է��������������ʽ��Է���������5�������ķ���ʽΪ����CH2O��5=C5H10O5��
0.1mol�����ܻ�ԭ21.6g�����������ʵ���Ϊ��
=0.2mol�����л����к���1��ȩ����
0.1mol��������24g���ᷢ��������Ӧ��24g��������ʵ���Ϊ��
=0.4mol������к���4���ǻ���
������ֱ�����ӣ�����Ľṹ��ʽΪ��CH2OH-CHOH-CHOH-CHOH-CHO������������Һ��Ӧ�Ļ�ѧ����ʽΪ��CH2OH-CHOH-CHOH-CHOH-CHO+2Ag��NH3��2OH
CH2OH-CHOH-CHOH-CHOH-COONH4++2Ag��+3NH3+H2O��
�ʴ�Ϊ��CH2OH-CHOH-CHOH-CHOH-CHO+2Ag��NH3��2OH
CH2OH-CHOH-CHOH-CHOH-COONH4++2Ag��+3NH3+H2O��
| 280 |
| 12��9+1��16+16��1 |
�ʴ�Ϊ��C18H32O2��
��2����B��C�Ļ���������KMnO4��Һ�����ữ�õ��������ֲ��HOOC-��CH2��10-COOH��CH3-��CH2��7-COOH��HOOC-��CH2��7-COOH��CH3-��CH2��4-COOH�����ݷ�Ӧԭ����RCH=CHR��
| �ټ���KMnO4��Һ���� |
| ���ữ |
�ʴ�Ϊ��CH3��CH2��7-CH=CH-��CH2��7-COOH��CH3��CH2��4-CH=CH-��CH2��10-COOH��
��3��ij�л���A��C��54.8%��H��5.58%��N��7.11%�����ຬO����O����������Ϊ1-54.8%-5.58%-7.11=32.51%�������л�����N��C����N��H����N��O����N��N��=
| 54.8% |
| 12 |
| 5.58% |
| 1 |
| 32.51% |
| 16 |
| 7.11% |
| 14 |
�ʴ�Ϊ��197��
��4�����Ǧ�-�����ᣬ�����в����ڼ���-CH3��������FeCl3��Һ������ɫ��Ӧ��˵�����з��ǻ���1mol ���������3molNaOH��ȫ��Ӧ��˵�������г����а������Ȼ�֮��Ӧ����2�����ǻ������ܵĽṹ��
 �ȣ���Ӧ��ͬ���칹����Ҫ�����ڱ������ǻ���λ�ã����ǻ�����ʱ����2��ͬ���칹�壬���ǻ����ʱ����3��ͬ���칹�壬���ǻ����ʱ����һ�ֽṹ������6��ͬ���칹�壬
�ȣ���Ӧ��ͬ���칹����Ҫ�����ڱ������ǻ���λ�ã����ǻ�����ʱ����2��ͬ���칹�壬���ǻ����ʱ����3��ͬ���칹�壬���ǻ����ʱ����һ�ֽṹ������6��ͬ���칹�壬�ʴ�Ϊ��
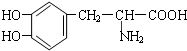 ��
����5����Ϊ����ȼ�����ĵ����������ɵĶ�����̼��ˮ�����ʵ�������ȣ�
���Ը��Ƿ�����C��H��O����ԭ�����ʵ���֮��Ϊ��1��2����2+1-2��=1��2��1���ʸ��ǵ����ʽΪ��CH2O��
�ʴ�Ϊ��CH2O��
��6��������Է��������������ʽ��Է���������5�������ķ���ʽΪ����CH2O��5=C5H10O5��
0.1mol�����ܻ�ԭ21.6g�����������ʵ���Ϊ��
| 21.6g |
| 108g/mol |
0.1mol��������24g���ᷢ��������Ӧ��24g��������ʵ���Ϊ��
| 24g |
| 60g/mol |
������ֱ�����ӣ�����Ľṹ��ʽΪ��CH2OH-CHOH-CHOH-CHOH-CHO������������Һ��Ӧ�Ļ�ѧ����ʽΪ��CH2OH-CHOH-CHOH-CHOH-CHO+2Ag��NH3��2OH
| �� |
�ʴ�Ϊ��CH2OH-CHOH-CHOH-CHOH-CHO+2Ag��NH3��2OH
| �� |
���������⿼���л��ƶϣ���Ŀ�ѶȽϴ������漰��֪ʶ��϶ࡢ�ۺ��Խ�ǿ���������ϴ��������ճ����л���ṹ������Ϊ���ؼ���ע����պ�ȷ���л������ʽ���ṹ��ʽ�ķ����������ֿ�����ѧ���ķ�����������������ѧ����������

��ϰ��ϵ�д�
�����Ŀ
����H+����Cl-����Al3+����K+����S2-����OH-����NO3-����NH4+ �ֱ����H2O�У������ϲ�Ӱ��ˮ�ĵ���ƽ����ǣ�������
| A���٢ۢݢߢ� | B���ڢܢ� |
| C���٢� | D���ڢܢޢ� |
Ǧ�����dz��õ�ֱ����Դ�����й�����ͼ��ʾװ�õ�˵���У���ȷ���ǣ�������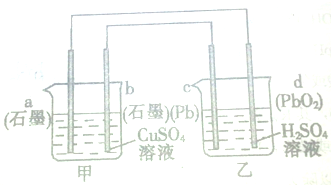

| A���缫bΪ���� |
| B���缫a�Ϸ����ķ�ӦΪCu2++2e-�TCu |
| C������ʱ��װ�ü�����Һ��pH��С |
| D�����·�е�����������Ϊ��b�����ߵ�c |
�����Ƽ��뵽����ϡ��Һ�У��������壬���г������ɵ��ǣ�������
| A��̼�������Һ |
| B���Ȼ�����Һ |
| C����������Һ |
| D���Ȼ����Һ |
��ͼ��ʾ��ʵ���������Ƶı�ע��ʵ�������ȷ���ǣ�������
A�� ��ʽ�ζ��� |
B�� ��������ȡ |
C��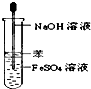 Fe��OH��2����ȡ |
D��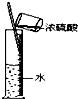 Ũ����ϡ�� |
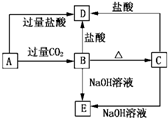 A��B��C��D��E���ֻ����������ij�ֳ���Ԫ�أ����ǵ�ת����ϵ��ͼ��ʾ������AΪ������Һ������ɫ��Ӧ��dz��ɫ������ɫ�ܲ�������BΪ���ܵİ�ɫ���壬A��E����������ͬ����ش��������⣺
A��B��C��D��E���ֻ����������ij�ֳ���Ԫ�أ����ǵ�ת����ϵ��ͼ��ʾ������AΪ������Һ������ɫ��Ӧ��dz��ɫ������ɫ�ܲ�������BΪ���ܵİ�ɫ���壬A��E����������ͬ����ش��������⣺