��Ŀ����
9������Ԫ�����ڱ������ڵ�һ����
��1���١���Ԫ���н�������ǿ��Ԫ����Mg����Ԫ�ط��ű�ʾ����
��2��������γɵĻ�����ĵ���ʽΪ
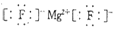 ��
����3������Ԫ�آڡ��ۡ�����ۺ������������ǿ������˳����HNO3��H2CO3��H2SiO3���ѧʽ����
��4����116��Ԫ�أ�Ԫ�ط���Lv�����й���ѧ����������ίԱ������Ϊ���W������Ԫ����Ԫ�����ڱ��е�λ���ǵ������ڵ�VIA�壬���ڽ���Ԫ�أ���������ǽ�������������Ԫ�ش�������������Ӧ��ˮ������仯ѧʽΪH2LvO4��
��5������������ΪN��R2+��������ΪA����ng������ͬ��̬�������������ĵ��ӵ����ʵ���ԼΪ$\frac{n}{��A+16��}$����A-N+8��mol��
���� ��Ԫ�������ڱ���λ�ÿ�֪���١���ֱ�ΪH��C��N��F��Mg��Al��Si��S��
��1��ֻ��Mg��AlΪ������ͬ���ڴ������ҽ����Լ�����
��2��������γɵĻ�����ΪMgF2��ֻ�����Ӽ���
��3���ǽ�����Խǿ����Ӧ��ۺ����������Խǿ��
��4����116��Ԫ�ص�����������Ϊ6����Oͬ���壬���Ϊ+6�ۣ�
��6������������ΪN��R2+��������ΪA��R��������ΪA-n��������ΪRO�����n=$\frac{m}{M}$���㣮
��� �⣺��1��ֻ��Mg��AlΪ������ͬ���ڴ������ҽ����Լ�������١���Ԫ���н�������ǿ��Ԫ����Mg���ʴ�Ϊ��Mg��
��2��������γɵĻ�����ΪMgF2��ֻ�����Ӽ�������ʽΪ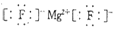 ���ʴ�Ϊ��
���ʴ�Ϊ��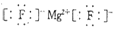 ��
��
��3���ǽ�����Խǿ����Ӧ��ۺ����������Խǿ����ڡ��ۡ�����ۺ������������ǿ������˳����HNO3��H2CO3��H2SiO3���ʴ�Ϊ��HNO3��H2CO3��H2SiO3��
��4����116��Ԫ�ص�����������Ϊ6����Oͬ���壬��������ϡ�������ԭ������Ϊ86�����Ԫ����Ԫ�����ڱ��е�λ���ǵ������ڵ�VIA�壬Ϊ����Ԫ�أ����Ϊ+6�ۣ�����Ԫ�ش�������������Ӧ��ˮ������仯ѧʽΪH2LvO4��
�ʴ�Ϊ���������ڵ�VIA�壻������ H2LvO4��
��6������������ΪN��R2+��������ΪA��R��������ΪA-n��������ΪRO��ng������ͬ��̬�������������ĵ��ӵ����ʵ���ԼΪ$\frac{n}{��A+16��}$����A-N+8��mol��
�ʴ�Ϊ��$\frac{n}{��A+16��}$����A-N+8����
���� ���⿼��λ�á��ṹ�����ʣ�Ϊ��Ƶ���㣬����Ԫ�ص�λ�á�Ԫ�ص����ʡ�Ԫ�ػ�����֪ʶ��Ԫ��������Ϊ���Ĺؼ������ط�����Ӧ�������Ŀ��飬ע�������֪ʶ��Ӧ�ã���Ŀ�ѶȲ���

 �ظ���ʦ�㲦ϵ�д�
�ظ���ʦ�㲦ϵ�д�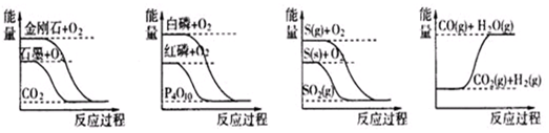
�ݴ��ж�����˵������ȷ���ǣ�������
| A�� | ʯīת��Ϊ���ʯ�����ȷ�Ӧ | |
| B�� | ��ͬ�����£����ױȺ����ȶ� | |
| C�� | ��ͬ�����£�������S��g����S ��s���������Ƚϣ�S ��s�������ϸ� | |
| D�� | CO��g��+H2O��g��=CO2��g��+H2 ��g�������ȷ�Ӧ |
| ������ | ����� | ǿ����� | ������� | �ǵ���� | |
| A | ���� | ˮú�� | ���� | ���� | HD |
| B | ��� | ������Һ | �Ȼ�����Һ | NH3•H2O | �������� |
| C | ���� | ʯ��ˮ | ���ᱵ | Al ��OH��3 | NH3 |
| D | ������ | �������� | ���Լ� | �⻯�� | �Ҵ� |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | 5.6gFe����1L 0.3moL/L�����У�ת�Ƶĵ�����Ϊ0.3NA | |
| B�� | ��������ƽ��ÿĦ��ˮ������ຬ��2NA����� | |
| C�� | 12gʯī��״�ṹ�к���ƽ���������ε���ĿΪNA | |
| D�� | 1molN2��3molH2��һ�������³�ַ�Ӧ�γɵ�N-H����ĿΪ6NA |
 �ڻ�ѧ��Ӧ�У���������ѧ��Ӧ�ķ��Ӽ���ײ��֮Ϊ��Ч��ײ����Щ���ӳ�Ϊ����ӣ�ʹ��ͨ���ӱ�ɻ���������ṩ����������л�ܣ��䵥λ��kJ•mol-1��ʾ��������۲�ͼ��Ȼ��ش����⣮
�ڻ�ѧ��Ӧ�У���������ѧ��Ӧ�ķ��Ӽ���ײ��֮Ϊ��Ч��ײ����Щ���ӳ�Ϊ����ӣ�ʹ��ͨ���ӱ�ɻ���������ṩ����������л�ܣ��䵥λ��kJ•mol-1��ʾ��������۲�ͼ��Ȼ��ش����⣮
 ������������ͭ����Һ��NaOH���Ի�������Ӧ�Ļ�ѧ����ʽ
������������ͭ����Һ��NaOH���Ի�������Ӧ�Ļ�ѧ����ʽ +4Cu��OH��2+2NaOH$\stackrel{��}{��}$NaOOC-COONa+2Cu2O��+6H2O��
+4Cu��OH��2+2NaOH$\stackrel{��}{��}$NaOOC-COONa+2Cu2O��+6H2O��
 ��
��