��Ŀ����
����˵������ȷ���ǣ�������
��ijԪ��ԭ�ӵ������ֻ��һ�����ӣ�����±�ؽ��ʱ��һ���γ����Ӽ�
�����ӻ�����һ���������Ӽ����ۻ�������һ�����������Ӽ�
��VIIA�嵥�ʴ��ϵ��£��۷е�Խ��Խ�ߣ���IA�嵥�ʵ��۷е�ҲԽ��Խ��
��H2O���Ӽ�������������H2O���ӱ�H2S�����ȶ�
��������״̬��ˮ��Һ�о����ܵ�������ʳ�Ϊ�ǵ����
��HClO4���Ա�H2SO4����ǿ��˵���ȵķǽ����Ա���ǿ
��������ܶȣ�Li��Na��K��Rb��Cs��
��ijԪ��ԭ�ӵ������ֻ��һ�����ӣ�����±�ؽ��ʱ��һ���γ����Ӽ�
�����ӻ�����һ���������Ӽ����ۻ�������һ�����������Ӽ�
��VIIA�嵥�ʴ��ϵ��£��۷е�Խ��Խ�ߣ���IA�嵥�ʵ��۷е�ҲԽ��Խ��
��H2O���Ӽ�������������H2O���ӱ�H2S�����ȶ�
��������״̬��ˮ��Һ�о����ܵ�������ʳ�Ϊ�ǵ����
��HClO4���Ա�H2SO4����ǿ��˵���ȵķǽ����Ա���ǿ
��������ܶȣ�Li��Na��K��Rb��Cs��
| A���٢ڢ� | B���٢ۢ� |
| C���ڢ� | D���ڢ� |
���㣺ͬһ������Ԫ�����ʵݱ������ԭ�ӽṹ�Ĺ�ϵ,�ǽ�����Ԫ�����ڱ��е�λ�ü������ʵݱ�Ĺ���,���ӻ�����Ľṹ����������,����Ĵ��ڶ��������ʵ�Ӱ��,�������ǵ����,�����������
ר�⣺
��������ijԪ��ԭ�ӵ������ֻ��һ�����ӣ�����±�ؽ��ʱ����һ���γ����Ӽ��������γɹ��ۼ���
�����ӻ�����һ���������Ӽ������ܺ��й��ۼ����ۻ�������һ�����������Ӽ���
��VIIA�嵥�ʴ��ϵ��£��۷е�Խ��Խ�ߣ���IA�嵥�ʵ��۷е���ϵ������ͣ�
�ܷ��ӵ��ȶ����뻯ѧ���йأ�
��������״̬��ˮ��Һ�о����ܵ���Ļ������Ϊ�ǵ���ʣ�
��Ԫ�صķǽ�����Խǿ��������������ˮ��������Խǿ��
��Na��K��λ�õߵ���
�����ӻ�����һ���������Ӽ������ܺ��й��ۼ����ۻ�������һ�����������Ӽ���
��VIIA�嵥�ʴ��ϵ��£��۷е�Խ��Խ�ߣ���IA�嵥�ʵ��۷е���ϵ������ͣ�
�ܷ��ӵ��ȶ����뻯ѧ���йأ�
��������״̬��ˮ��Һ�о����ܵ���Ļ������Ϊ�ǵ���ʣ�
��Ԫ�صķǽ�����Խǿ��������������ˮ��������Խǿ��
��Na��K��λ�õߵ���
���
�⣺��ijԪ��ԭ�ӵ������ֻ��һ�����ӣ�����±�ؽ��ʱ����һ���γ����Ӽ��������γɹ��ۼ�����HX���ʴ���
�����ӻ�����һ���������Ӽ������ܺ��й��ۼ����ۻ�������һ�����������Ӽ���ֻ�����ۼ�������ȷ��
��VIIA�嵥�ʴ��ϵ��£��۷е�Խ��Խ�ߣ���IA�嵥�ʵ��۷е���ϵ������ͣ��ʴ���
�ܷ��ӵ��ȶ����뻯ѧ���йأ�����Ӽ��������أ��ʴ���
��������״̬��ˮ��Һ�о����ܵ���Ļ������Ϊ�ǵ���ʣ���ƾ��ȣ��ʴ���
��Ԫ�صķǽ�����Խǿ��������������ˮ��������Խǿ������HClO4���Ա�H2SO4����ǿ��˵���ȵķǽ����Ա���ǿ������ȷ��
��������ܶȣ�Li��K��Na��Rb��Cs����ѡC��
�����ӻ�����һ���������Ӽ������ܺ��й��ۼ����ۻ�������һ�����������Ӽ���ֻ�����ۼ�������ȷ��
��VIIA�嵥�ʴ��ϵ��£��۷е�Խ��Խ�ߣ���IA�嵥�ʵ��۷е���ϵ������ͣ��ʴ���
�ܷ��ӵ��ȶ����뻯ѧ���йأ�����Ӽ��������أ��ʴ���
��������״̬��ˮ��Һ�о����ܵ���Ļ������Ϊ�ǵ���ʣ���ƾ��ȣ��ʴ���
��Ԫ�صķǽ�����Խǿ��������������ˮ��������Խǿ������HClO4���Ա�H2SO4����ǿ��˵���ȵķǽ����Ա���ǿ������ȷ��
��������ܶȣ�Li��K��Na��Rb��Cs����ѡC��
���������⿼����ۺϣ��漰��ѧ�������ʵĹ�ϵ���ǽ�����ǿ���жϡ�ͬһ����Ԫ�����ʵݱ���ɵ�֪ʶ�㣬���ؿ�������ж����������þ��������������Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
���к��ǻ���OH�����л��ﲻ���ڴ����ǣ�������
A�� |
| B��CH3OH |
C��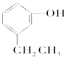 |
D�� |
�����£����и���������ָ����Һ��һ���ܴ���������ǣ�������
| A������������Һ�У�Na+��K+��MnO4-��AlO2- | ||
| B��0.1 mol?L-1 FeCl3��Һ��K+��NH4+��I-��SCN- | ||
C��
| ||
| D�����ܽ�Al2O3����Һ��Na+��K+��Cl-��CO32- |
��ӦA��g��+3B��g��?2C��g��+2D��g���ڲ�ͬ����²�÷�Ӧ���ʣ����з�Ӧ���������ǣ�������
| A���ԣ�A��=0.1 mol?L-1?s-1 |
| B���ԣ�B��=0.6 mol?L-1?s-1 |
| C���ԣ�C��=0.5 mol?L-1?s-1 |
| D���ԣ�D��=0.4 mol?L-1?s-1 |
�������ʱ��������⣬���ִ���ѧ��ҵ�У�������ѧ��Ӧ���ʵ�����Ҫ�������ǣ�������
| A����Ӧ���¶� |
| B������ |
| C����Ӧ��ϵ��ѹǿ |
| D����Ӧ���Ũ�� |
һ�������·�ӦN2��g��+3H2��g��?2NH3��g ����10L���ܱ������н��У����2min�ڣ�N2�����ʵ�����20mol��С��8mol����2min��N2�ķ�Ӧ����Ϊ��������
| A��1.2mol/��L?min�� |
| B��1mol/��L?min�� |
| C��0.6mol/��L?min�� |
| D��0.4mol/��L?min�� |
�������ӷ���ʽ��д����ȷ���ǣ�������
| A����̼��������Һ�е����������� HCO3-+OH-=CO32-+H2O |
| B����̼�������Һ�е�����������ʯ��ˮ 2HCO3-+Ca2++2OH-=CO32-+2H2O+CaCO3�� |
| C������������Һ��ͨ������Ķ������� ClO-+SO2+H2O=HSO3-+HClO |
| D������ͭ������������Һ��� Cu2++SO42-+Ba2++2OH-=Cu��OH��2��+BaSO4�� |
