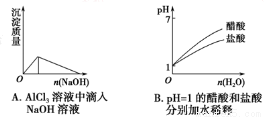��Ŀ����
��16�֣��±�ΪԪ�����ڱ���һ���֣�����Ԫ�آ١����ڱ��е�λ�ã����û�ѧ��������
�� ���� | IA | 0 | ||||||
1 | �� | ��A | ��A | ��A | ��A | ��A | ��A | |
2 | �� | �� | �� | �� | ||||
3 | �� | �� | �� | �� | �� | |||
��1���ݡ��ޡ��ߵ����Ӱ뾶�ɴ�С��˳��Ϊ ���������ӷ��ţ�
��2���ࡢ�ᡢ�����ۺ������������ǿ����Ϊ �����û�ѧʽ��ʾ��
��3��д�����ڿ����м������ɵIJ���ĵ���ʽ�� ��
��4���ɱ���Ԫ���γɵ����ʿɷ�����ͼ�еķ�Ӧ������B��C��G�ǵ��ʣ�BΪ����ɫ���壬D��Һ�Լ��ԡ�

��д��D��Һ��G��Ӧ�Ļ�ѧ����ʽ ��
�ڻ����X�е�ij���ʲ�����ˮ���������������������ڼ��д�����ڼ�����Һ�еĵ��뷽��ʽ ��
��5����֪ijЩ��ͬ��Ԫ�ص�����Ҳ��һ���������ԣ���Ԫ�آ���Ԫ�آߵ��������������Ƶ����ʡ�д��Ԫ�آڵ�����������NaOH��Һ��Ӧ�����ӷ���ʽ�� ��
��6���飨As���Ļ����������ɱ�漰ҽ�ơ�
��As��ԭ�ӽṹʾ��ͼΪ ��
��Y�ɢܢޢ�����Ԫ����ɣ��������г�������������As����Y��ˮ��Һ��Ӧ��������As����ۺ����ᣬ�÷�Ӧ�Ļ�ѧ����ʽΪ ��
��1��F->Na+>Al3+��2�֣�����2��HClO4>H2SO4>H3PO4��2�֣�����3��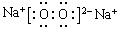 ��2�֣���
��2�֣���
��4����2Al+2NaOH+2H2O=2NaAlO2+3H2����2�֣����� Al(OH)3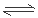 AlO2-+H++H2O��2�֣���
AlO2-+H++H2O��2�֣���
��5��Be(OH)2+2OH-=BeO22-+2H2O��2�֣���
��6����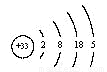 ��2�֣����� 5NaClO+2As+3H2O=2H3AsO4+5NaCl
��2�֣����� 5NaClO+2As+3H2O=2H3AsO4+5NaCl
��������
�����������1���ݺ�Ԫ����F����Ԫ����Na���ߺ�Ԫ����Al�����ǵ����ӵ��Ӳ�ṹ��ͬ�����ڵ��Ӳ�ṹ��ͬ��������˵���˵����Խ�����Ӱ뾶��ԽС���������Ӱ뾶�ɴ�С��˳��ΪF->Na+>Al3+����2���ࡢ�ᡢ������Ԫ�طֱ���P��S��Cl��Ԫ�صķǽ�����Խǿ��������������Ӧ��ˮ��������Ծ�Խǿ������Ԫ�صķǽ����ԣ�Cl>S>P��������ۺ������������ǿ����ΪHClO4>H2SO4>H3PO4����3����Ԫ��Na�ڿ�����ȼ�ղ���Na2O2������ʽ�ǣ�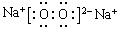 ����4���������ʵ����ʼ��ת����ϵ��֪��A��NaCl��B��Cl2��C��H2��D��NaOH��G��Al��F��NaAlO2�������X�к��е�������ˮ�IJ��ǹ�����Al(OH)3����D��Һ��G��Ӧ�Ļ�ѧ����ʽ��2Al+2NaOH +2H2O= 2NaAlO2 +3H2�����ڻ����X�е�ij���ʲ�����ˮ���������������������ڼ��д�����ڼ�����Һ�еĵ��뷽��ʽ��Al(OH)3
����4���������ʵ����ʼ��ת����ϵ��֪��A��NaCl��B��Cl2��C��H2��D��NaOH��G��Al��F��NaAlO2�������X�к��е�������ˮ�IJ��ǹ�����Al(OH)3����D��Һ��G��Ӧ�Ļ�ѧ����ʽ��2Al+2NaOH +2H2O= 2NaAlO2 +3H2�����ڻ����X�е�ij���ʲ�����ˮ���������������������ڼ��д�����ڼ�����Һ�еĵ��뷽��ʽ��Al(OH)3 AlO2-+H++H2O����5������Ԫ�������ڱ��е�λ�ÿ�֪��Ԫ����Be��Ԫ�آڵ�����������NaOH��Һ��Ӧ�����ӷ���ʽ�ǣ�Be(OH)2+2OH-=BeO22-+2H2O����6����33��Ԫ��As��ԭ�ӽṹʾ��ͼΪ
AlO2-+H++H2O����5������Ԫ�������ڱ��е�λ�ÿ�֪��Ԫ����Be��Ԫ�آڵ�����������NaOH��Һ��Ӧ�����ӷ���ʽ�ǣ�Be(OH)2+2OH-=BeO22-+2H2O����6����33��Ԫ��As��ԭ�ӽṹʾ��ͼΪ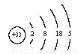 ����Y�ɢܢޢ�����Ԫ����ɣ��������г����������������������NaClO��As����Y��ˮ��Һ��Ӧ��������As����ۺ����ᣬ���ݵ����غ㼰ԭ���غ�ɵø÷�Ӧ�Ļ�ѧ����ʽΪ5NaClO+2As+3H2O=2H3AsO4+5NaCl��
����Y�ɢܢޢ�����Ԫ����ɣ��������г����������������������NaClO��As����Y��ˮ��Һ��Ӧ��������As����ۺ����ᣬ���ݵ����غ㼰ԭ���غ�ɵø÷�Ӧ�Ļ�ѧ����ʽΪ5NaClO+2As+3H2O=2H3AsO4+5NaCl��
���㣺����Ԫ�ؼ���������ƶϡ�Ԫ�ص�λ�á����ʵ��������ʡ�����ʽ��ԭ�ӽṹʾ��ͼ����ѧ����ʽ�����ӷ���ʽ�����뷽��ʽ����д�����Ӱ뾶�ıȽϡ���������ǿ���Ƚϵ�֪ʶ��

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�