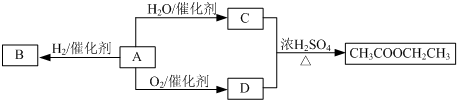
��Ŀ����
8��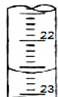 ��ijѧ����0.20mol•L-1�ı�NaOH��Һ�ζ�δ֪Ũ�ȵ����ᣬ�����Ϊ���¼�����
��ijѧ����0.20mol•L-1�ı�NaOH��Һ�ζ�δ֪Ũ�ȵ����ᣬ�����Ϊ���¼�������������ˮϴ�Ӽ�ʽ�ζ��ܣ�������ע��NaOH��Һ����0���̶�������
�ڹ̶��õζ��ܲ�ʹ�ζ��ܼ������Һ��
�۵���Һ������0����0���̶������£������¶���
����ȡ20.00mL����Һע��ྻ����ƿ�У�������3�η�̪��Һ
���ñ�Һ�ζ����յ㣬���µζ���Һ�������
��ش�
��1�����ϲ����д�����ǣ����ţ���
��2��������У��ڼ��µζ���Һ�����ʱ���ζ��ܼ��������ݣ������²ⶨ���ƫС���ƫ����ƫС������Ӱ�족��
��3����ͼ��ij�εζ�ʱ�ĵζ����е�Һ�棬�����Ϊ22.60mL
��4�������������ݣ���������������Һ��Ũ�ȣ�0.2mol/L
| �ζ����� | ���������ml�� | ���ռ������ml�� | |
| �ζ�ǰ���� | �ζ������ | ||
| ��һ�� | 20.00 | 0.40 | 20.40 |
| �ڶ��� | 20.00 | 4.00 | 24.00 |
| ������ | 20.00 | 2.00 | 24.10 |
��5��ij�¶�ʱ��ˮ�����ӻ�����KW=10-13�������¶���pH=11��Ba��OH��2��ҺaL��pH=1��H2SO4��ҺbL��ϣ�������Һ���Ϊ����֮�ͣ����ù���������Բ��ƣ������û����ҺΪ���ԣ���a��b=10��1
��6����֪��25���ˮ��Һ�У�AgX��AgY��AgZ��������ˮ����Ksp��AgX��=1.8��10-10��Ksp��AgY��=1.0��10-12��Ksp��AgZ��=8.7��10-17����AgX��AgY��AgZ���ߵ��ܽ�ȣ�mol/L��S��AgX����S��AgY����S��AgZ���Ĵ�С˳��Ϊ��s��AgX����s��AgY����s��AgZ��������AgY�ı�����Һ�м���������AgX���壬��c��Y-����С���������С�����䡱����
���� ��1���ζ�����װҺǰҪ������ϴ��
��2������c�����⣩=$\frac{c��������V������}{V�����⣩}$��������������V��������Ӱ�죬�Դ��ж�Ũ�ȵ���
��3�����ݵζ��ܵĽṹ�;�ȷ��Ϊ0.01mL�����
��4���ȸ������ݵ���Ч�ԣ���ȥ��3�����ݣ�Ȼ�����1��2��ƽ������V��NaOH�������Ÿ��ݸ���c�����⣩=$\frac{c��������V������}{V�����⣩}$�����㣮
��5�����û����ҺΪ���ԣ�һ������n��H+��=n��OH-�������¶���ˮ�����ӻ�����KW=10-13����pH=11����Һ��c��OH-��=$\frac{1{0}^{-13}}{1{0}^{-11}}$mol/L=0.01mol/L��pH=1��������������Ũ��Ϊ0.1mol/L���ݴ���ʽ���㣻
��6���ṹ���Ƶ��������Σ��ܶȻ�ԽС���ܽ��ԽС��AgY�ܵ��ܶȻ���AgXС���ӳ���ƽ���ƶ��ĽǶȷ�����
��� �⣺��1���ζ�����װҺǰҪ������ϴ�����Ԣٴ���
�ʴ�Ϊ���٣�
��2��������У��ڼ��µζ���Һ�����ʱ���ζ��ܼ��������ݣ����V������ƫС������c�����⣩=$\frac{c��������V������}{V�����⣩}$��������֪c������ƫС��
�ʴ�Ϊ��ƫС��
��3���ζ�ʱ�ĵζ����е�Һ�棬�����Ϊ22.60mL��
�ʴ�Ϊ��22.60��
��4�����εζ����ĵ����Ϊ��20.00mL��20.00mL��22.10����ȥ��3�����ݣ�Ȼ�����1��2��ƽ������V��NaOH��=20.00mL��c�����⣩=$\frac{c��������V������}{V�����⣩}$=$\frac{0.20mol•{L}^{-1}��20.00mL}{20.00mL}$=0.2000mol•L-1��
�ʴ�Ϊ��0.2��
��5�����û����ҺΪ���ԣ���n��H+��=n��OH-�������¶���ˮ�����ӻ�����KW=10-13����pH=11����Һ��c��OH-��=$\frac{1{0}^{-13}}{1{0}^{-11}}$mol/L=0.01mol/L��
pH=1��������������Ũ��Ϊ0.1mol/L��
��bL��0.1mol/L=aL��0.01mol/L��
��ã�a��b=10��1
�ʴ�Ϊ��10��1��
��6�����ݻ�ѧʽ���Ƶ����ʵ��ܶȻ�����ԽС������Խ���ܣ��ܽ��ԽС����֪�����ܽ�ȣ�mol/L���Ĵ�С˳��ΪS��AgX����S��AgY����S��AgZ����
AgX��AgY�ܽ�ȴ�����AgX������Һ��c��Ag+���ϴ�����AgY�ı�����Һ�м�������AgX�Ĺ��壬��c��Ag+������AgY�ij����ܽ�ƽ�����ƣ�����c��Y-������С��
�ʴ�Ϊ��s��AgX����s��AgY����s��AgZ������С��
���� ���⿼���к͵ζ�����ҺpH�ļ��㡢���ܵ���ʵ��ܽ�ƽ�⼰����ת���ı��ʣ�����ѧ���ķ��������������Ŀ��飬��Ŀ�Ѷ��еȣ�����ע���ܶȻ����������ú����⣮

 һ����ʦȨ����ҵ��ϵ�д�
һ����ʦȨ����ҵ��ϵ�д������Ż����ˮ���� �����ڿ����в��ױ���ʴ���������Dz����ý����� �������ڳ�ʪ�Ŀ����пɷ���������ʴ�� ������ȱ�ƻ�����������ɡ�ȱ��������ƶѪ�� ����ͭ������֡�Ӳ�����ǺϽ� ��Al��Fe��һ������������ijЩ�����ﷴӦ��
| A�� | �٢ڢ� | B�� | �ڢۢ� | C�� | �ۢܢݢ� | D�� | ȫ����ȷ |
�ٰѱ����Ƴ�ʯ���顡
���ں�ˮ�м���ʯ���飬���ˣ�ϴ�ӳ����
�۽������������ᷴӦ���ᾧ�����ˡ�
�����Ȼ����������и��ᄃ�塡
�ݵ����������������
����˵����ȷ���ǣ�������
| A�� | þԪ����Ԫ�����ڱ���λ�ڵ������ڡ��ڢ�B�� | |
| B�� | �ڢڲ�ϴ����Ϻ���ϴ��Һ�еμ�̼������Һ�ɼ�������Ƿ�ϴ�Ӹɾ� | |
| C�� | �ڹ�ҵ�����Ͽ���NaOH��Һ����ʯ���� | |
| D�� | �����Ҳ���Բ��õ��þ���ˮ��Һ�ķ��� |
��1������װ����ͼ��ʾ��
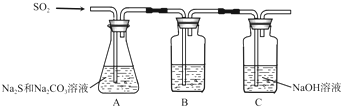
��װ��B�������Ǽ���װ��A��SO2������Ч�ʣ�B���Լ���Ʒ�졢��ˮ��KMnO4��Һ������SO2����Ч�ʵ͵�ʵ��������B����Һ��ɫ�ܿ���ɫ��
��Ϊ��ʹSO2������������ȫ���ڲ��ı�A����ҺŨ�ȡ�����������£����˼�ʱ���跴Ӧ���⣬���ɲ�ȡ�ĺ�����ʩ�ǿ���SO2�����١��ʵ������¶ȣ���д��������
��2�����豾ʵ�����õ�Na2CO3������NaCl��NaOH�����ʵ�鷽�����м��飮������ʱCaCO3������Һ��pH=10.2��
��ѡ�Լ���������ϡ���ᡢAgNO3��Һ��CaCl2��Һ��Ca��NO3��2��Һ����̪��Һ������ˮ��pH�ơ��ձ����Թܡ��ι�
| ��� | ʵ����� | Ԥ������ | ���� |
| �� | ȡ������Ʒ���Թ��У�������������ˮ��������ܽ⣬�μ�����ϡ���ᣬ�ٵμ�����AgNO3��Һ���� | �а�ɫ�������� | ��Ʒ��NaCl |
| �� | ��ȡ������Ʒ���ձ��У�������������ˮ����ֽ����ܽ⣬��ȡ������Ʒ���ձ��У�������������ˮ����ֽ����ܽ⣬�������CaCl2��Һ�����裬���ã���pH�Ʋⶨ�ϲ���ҺpH | �а�ɫ�������ɣ��ϲ���ҺpH����10.2 | ��Ʒ��NaOH |
��֪��Cr2O72-+6I-+14H+�T2Cr3++3I2+7H2O 2S2O32-+I2�TS4O62-+2I-��
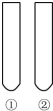 ���С��Լ����͡��Թ��е����ʡ�������ɡ�ʵ��Ŀ�ġ����ǣ�������
���С��Լ����͡��Թ��е����ʡ�������ɡ�ʵ��Ŀ�ġ����ǣ�������| ʵ��Ŀ�� | �Լ� | �Թ��е����� | |
| A | �ǻ��Ա����Ļ�����Ӱ�� | ������ˮ | �ٱ��ڱ�����Һ |
| B | ���Ա����Ļ�����Ӱ�� | ����KMnO4��Һ | �ٱ��ڼױ� |
| C | ��������û��̼̼˫�� | Br2��CCl2��Һ | �ٱ�����ϩ |
| D | ̼������Աȱ���ǿ | ʯ����Һ | �ٱ�����Һ��̼����Һ |
| A�� | A | B�� | B | C�� | C | D�� | D |
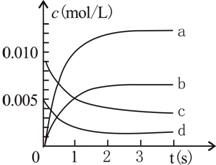 800��ʱ����2L�ܱ������ڼ���N0��02��������Ӧ��2N0��g��+O2��g��?2N02��g�������n��N0����ʱ��ı仯�������ش�
800��ʱ����2L�ܱ������ڼ���N0��02��������Ӧ��2N0��g��+O2��g��?2N02��g�������n��N0����ʱ��ı仯�������ش�| ʱ�䣨s�� | 0 | 1 | 2 | 3 | 4 | 5 |
| n��NO����mol�� | 0.020 | 0.010 | 0.008 | 0.007 | 0.007 | 0.007 |
��2��ͼ�б�ʾN02�仯��������b��
��3����˵���÷�Ӧ�Ѵﵽƽ��״̬����ad��
a�����������������ʵ������ֲ���
b������Ӧ���淴Ӧ�����ʶ�Ϊ0
c��������N0��02��N02���ʵ���֮��Ϊ2��1��2
d��������N0���������������䣮
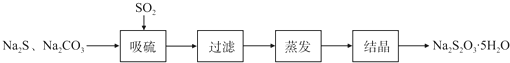
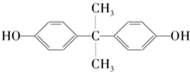 ��������̼�����Ĺ������£�
��������̼�����Ĺ������£�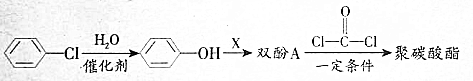
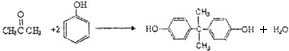 ��
��