��Ŀ����
15��������ĸ��ʾ�������м���Ԫ�ص�λ�ã���ش��������⣮| A | |||||||
| B | C | D | |||||
| E | F | G | H | I | |||
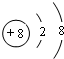 ��E��I�γɵĻ�����ĵ���ʽΪ
��E��I�γɵĻ�����ĵ���ʽΪ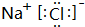 ��
����2��E��G��H��Ԫ�ص�����������Ӧˮ�������Һ�����Ӧ�����ӷ���ʽΪAl��OH��3+OH-=AlO2-+2H2O��Al��OH��3+3H+=Al3++3H2O��OH-+H+=H2O��
��3��F��I�γɵĻ������д��ڵĻ�ѧ�����������Ӽ�����A��I����Ԫ���У���ȫ�ɷǽ���Ԫ���γɵ�һ�����ӻ�����Ļ�ѧʽ��NH4NO3��
��4��4.8g����F�����״����2.24L��B�����������ǡ�÷�Ӧ��
���� ��Ԫ�������ڱ���λ�ã���֪AΪ�⣬BΪ̼��CΪN��DΪO��EΪNa��FΪMg��GΪAl��HΪS��IΪCl��
��1��O2-�����Ӻ��������Ϊ10�����������Ϊ2��8��E��I�γɵĻ�����ΪNaCl�����������������ӹ��ɣ�
��2��E��G��H��Ԫ�ص�����������Ӧˮ����ֱ�ΪNaOH��Al��OH��3��H2SO4��
��3��F��I�γɵĻ�����ΪMgCl2����þ�����������ӹ��ɣ���A��I����Ԫ���У���ȫ�ɷǽ���Ԫ���γɵ�һ�����ӻ�����Ϊ����淋ȣ�
��4��������Ӧ2Mg+CO2$\frac{\underline{\;��ȼ\;}}{\;}$2MgO+C�����ݷ���ʽ������Ҫ������̼�������
��� �⣺��Ԫ�������ڱ���λ�ã���֪AΪ�⣬BΪ̼��CΪN��DΪO��EΪNa��FΪMg��GΪAl��HΪS��IΪCl��
��1��O2-�����Ӻ��������Ϊ10�����������Ϊ2��8�����ӽṹʾ��ͼΪ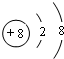 ��E��I�γɵĻ�����ΪNaCl�����������������ӹ��ɣ�����ʽΪ
��E��I�γɵĻ�����ΪNaCl�����������������ӹ��ɣ�����ʽΪ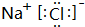 ��
��
�ʴ�Ϊ��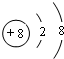 ��
��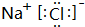 ��
��
��2��E��G��H��Ԫ�ص�����������Ӧˮ����ֱ�ΪNaOH��Al��OH��3��H2SO4�����Ӧ���ӷ���ʽΪ��Al��OH��3+OH-=AlO2-+2H2O��Al��OH��3+3H+=Al3++3H2O��OH-+H+=H2O��
�ʴ�Ϊ��Al��OH��3+OH-=AlO2-+2H2O��Al��OH��3+3H+=Al3++3H2O��OH-+H+=H2O��
��3��F��I�γɵĻ�����ΪMgCl2����þ�����������ӹ��ɣ��������Ӽ�����A��I����Ԫ���У���ȫ�ɷǽ���Ԫ���γɵ�һ�����ӻ�����ΪNH4NO3�ȣ�
�ʴ�Ϊ�����Ӽ���NH4NO3��
��4��������Ӧ2Mg+CO2$\frac{\underline{\;��ȼ\;}}{\;}$2MgO+C��4.8gMg�����ʵ���Ϊ$\frac{4.8g}{24g/mol}$=0.2mol���ɷ���ʽ��֪��Ҫ������̼Ϊ0.2mol��$\frac{1}{2}$=0.1mol������Ҫ������̼�����Ϊ0.1mol��22.4L/mol=2.24L��
�ʴ�Ϊ��2.24��
���� ���⿼��Ԫ�����ڱ���Ԫ�������ɵ��ۺ�Ӧ�ã���������Ԫ�����ڱ��Ľṹ�����ضԻ�ѧ����Ŀ��飬ע��Ի���֪ʶ���������գ�

| A�� | 18 | B�� | 8 | C�� | 10 | D�� | 16 |
| A�� | ͨ������KMnO4��Һ�� | B�� | ͨ����������ˮ�� | ||
| C�� | ��ȼ | D�� | ͨ��H2����� |
| A�� | CH4+Cl2$\stackrel{����}{��}$CH3Cl+HCl | |
| B�� | CH2=CH2+HBr��CH3CH2Br | |
| C�� | 2CH3CH2OH+2Na��2 CH3CH2ONa+H2�� | |
| D�� | 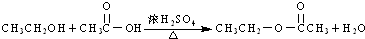 |
| A�� | N2��H2��NH3���߹��� | |
| B�� | ������������ܶȲ��ٷ����仯 | |
| C�� | N2��H2��NH3�����ʵ���Ũ����� | |
| D�� | ��λʱ������0.1 mol N2��ͬʱ����0.2 mol NH3 |
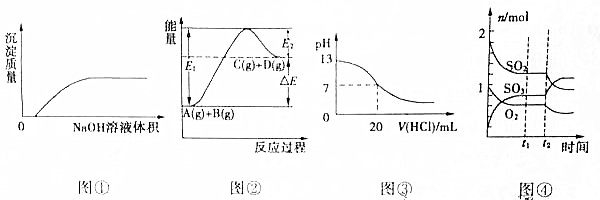
| A�� | ͼ�ٱ�ʾ��NaOH��Һ����Ba��HCO3��2��Һ�� | |
| B�� | ͼ�ڱ�ʾ��Ӧ����ܼ���С����������ܼ��� | |
| C�� | ͼ�۱�ʾ25��ʱ��0.1mol/L�������20mL0.1mol/LNaOH��Һ�У���Һ��pH�����������ı仯���� | |
| D�� | ͼ�ܱ�ʾһ�������µķ�Ӧ2SO2��g��+O2?2SO3��g����H��0��t2ʱ�̸ı������ |
| A�� | ���������ζ | B�� | ���ۺ���ά�ؾ��������� | ||
| C�� | ���������ˮ�� | D�� | �������C��H��OԪ�� |
