��Ŀ����
10��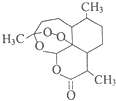 �����أ��ṹ��ʽ��ͼ����һ�ְ�ɫ��״���壬��������������֯�����ǡ�������Ψһ��Ч��ű������ҩ����й�Ů��ѧ��������ƾ�������Ѵ���������ȡ�����ػ��2015��ŵ����ҽѧ�������й��������ص�˵������ȷ���ǣ�������
�����أ��ṹ��ʽ��ͼ����һ�ְ�ɫ��״���壬��������������֯�����ǡ�������Ψһ��Ч��ű������ҩ����й�Ů��ѧ��������ƾ�������Ѵ���������ȡ�����ػ��2015��ŵ����ҽѧ�������й��������ص�˵������ȷ���ǣ�������| A�� | �����ط����к����Ѽ��������ȹ����� | |
| B�� | �������ڼ���ʱ����NaOH��ˮ��Һ��Ӧ | |
| C�� | ������������ˮ�������ܼ� | |
| D�� | �����Ѵ���������ȡ�����أ��õ�����ȡԭ�� |
���� �����غ����������ɷ���ˮ�ⷴӦ�����й�����������ǿ�����ԣ�������ɱ���������Դ˽����⣮
��� �⣺A���ɽṹ��ʽ��֪�����ط����к����Ѽ��������ȹ����ţ���A��ȷ��
B�������������ڼ���ʱ����NaOH��ˮ��Һ��Ӧ����B��ȷ��
C�����������������������������ʣ�������ˮ����C����
D�����������������ѣ������Ѵ���������ȡ�����أ��õ�����ȡԭ������D��ȷ��
��ѡC��
���� ���⿼���л���Ľṹ�����ʣ�Ϊ�߿��������ͣ�������ѧ���ķ��������Ŀ��飬ע������л���Ľṹ�����ŵ����ʣ�Ϊ��������Ŀ�Ĺؼ����ѶȲ���

��ϰ��ϵ�д�
 ȫ�Ų��Ծ�ϵ�д�
ȫ�Ų��Ծ�ϵ�д� ���100��1�ž�ϵ�д�
���100��1�ž�ϵ�д�
�����Ŀ
20������˵����ȷ���ǣ�������
| A�� | �㷶pH��ֽ������ΧΪ0��14�����������Ƚϲ����pH��ֽ�IJ�����Χ��խ�����������ȱȹ㷶pH��ֽ�� | |
| B�� | ���������һ�ֳ��õĻ�ѧ�Լ�����������ؾ�����ʱ��Ӧ���в���������ĥ���Եõ�ϸС���壬�����ڼӿ췴Ӧ���ܽ������ | |
| C�� | ȡ�����Ȼ��ܾ����Ũ�����ܽ⣬Ȼ��μ�ˮ����Һ����ɫ����������Һ������ˮ������Һ��ɫ��Ϊ��ɫ | |
| D�� | ̽���¶ȶԻ�ѧ��Ӧ���ʵ�Ӱ��ʵ��ʱ��Ӧ�Ƚ���ȡ��Na2S2O3��H2SO4��Һ��ϣ�Ȼ��������Ӧ�¶ȵ���ˮԡ�з�Ӧ����¼ÿ�鷴Ӧ����ʱ�� |
1��������ʵ��Ӧ������ͽ��;���ȷ���ǣ�������
| ��ʵ | ���� | ���� | |
| A | �Ȼ�����Һ�еμ����������� | ������ɫ���� | 2Fe3++H2S=2Fe2++S��+2H+ |
| B | Fe��NO3��2��Һ��ϡ���� | ��Һ��ɫ���� | Fe2++H2O?Fe��OH��2+2H+ ������ˮ�⣬dz��ɫ���� |
| C | ��֪CH4��ȼ����Ϊ890kJ/mol | ��������ȼ�ϣ�1.6g CH4��ȫȼ�գ�����89kJ | CH4��g��+2O2��g��=CO2��g��+2H2O��g�� ��H=-890kJ/mol |
| D | Mg��OH��2����Һ�еμ�FeCl3��Һ��Mg��OH��2������ȫ��Ӧ | ��ɫ������Ϊ���ɫ���� | $\frac{{k}_{sp}^{3}[Mg��OH��_{2}]}{{k}_{sp}^{2}[Fe��OH]_{3}]}$��105 |
| A�� | A | B�� | B | C�� | C | D�� | D |
5�����ж�ʵ������Ľ�������۾���ȷ���ǣ�������
| ѡ�� | ʵ����� | ʵ������ | ��������� |
| A | ��ij��Һ�м���BaCl2��Һ | �а�ɫ�������� | ������������ˮ��BaSO4������Һ��һ������SO42- |
| B | ������AgNO3��Һ�еμ�����NaCl��Һ���ٵμ�����ϡNaI��Һ | ��ʼ�а�ɫ�������ɣ����л�ɫ�������� | Ksp��AgI����Ksp��AgCl�� |
| C | ���е�������Ũ��ˮ�������� | ��Һ�ֲ㣬�ϲ�ʳȺ�ɫ���²㼸����ɫ | ������ˮ����ȡ����Ӧ��ʹ��ˮ��ɫ |
| D | �������м���Ũ���� | ��ڣ����ȣ�������ͣ��ų��̼������� | Ũ���������ˮ�Ժ�ǿ�����ԣ���Ӧ������C��SO2��CO2�� |
| A�� | A | B�� | B | C�� | C | D�� | D |
15����֪AB3�������л��ܼ���������ˮ��������˵����ȷ���ǣ�������
| A�� | AB3�ǷǼ��Է��� | B�� | Aԭ���¶Ե��� | ||
| C�� | Aԭ��Ϊsp3�ӻ� | D�� | AB3Ϊ�����νṹ |
2���������ӻ��������Һ���ܴ��������һ���ǣ�������
| A�� | Al3+��SO42-��NH3•H2O | B�� | Na+��OH-��Cl2 | ||
| C�� | K+��Ca2+��CO32- | D�� | H+��Na+��NO3- |
5���Թ�����Ϊ�����������������V2O5���ǽӴ�����������Ĵ������ӷϷ������л���V2O5�ȱ�����Ⱦ����
����������Դ�ۺ����ã��Ϸ���������Ҫ�ɷ�Ϊ��
������һ�ַϷ��������չ���·�ߣ�
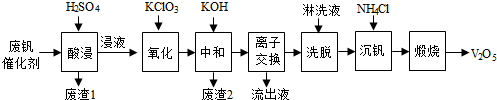
�ش��������⣺
��1���������ʱV2O5ת��ΪVO2+����Ӧ�����ӷ���ʽΪV2O5+2H+=2VO2++H2O��ͬʱV2O4ת��VO2+��������1������Ҫ�ɷ���SiO2��
��2��������������ʹ3 mol��VO2+��ΪVO2+������Ҫ������KClO3����Ϊ0.5mol��
��3�����к͡�����֮һ��ʹ����V4O124-��ʽ��������Һ�У�������2���к���Fe��OH��3��Al��OH��3��
��4�������ӽ������͡�ϴ�ѡ��ɼ�ʾΪ��4ROH+V4O124-$?_{ϴ��}^{���ӽ���}$R4V4O12+4OH-����ROHΪǿ���������ӽ�����֬����Ϊ�����ϴ��Ч�ʣ���ϴҺӦ�óʼ��ԣ���ᡱ������С�����
��5��������Һ����������������K+��
��6�����������õ�ƫ����泥�NH4VO3��������д�������ա��з�����Ӧ�Ļ�ѧ����ʽ2NH4VO3$\frac{\underline{\;����\;}}{\;}$V2O5+H2O��+2NH3����
����������Դ�ۺ����ã��Ϸ���������Ҫ�ɷ�Ϊ��
| ���� | V2O5 | V2O4 | K2SO4 | SiO2 | Fe2O3 | Al2O3 |
| ��������/% | 2.2��2.9 | 2.8��3.1 | 22��28 | 60��65 | 1��2 | ��1 |

�ش��������⣺
��1���������ʱV2O5ת��ΪVO2+����Ӧ�����ӷ���ʽΪV2O5+2H+=2VO2++H2O��ͬʱV2O4ת��VO2+��������1������Ҫ�ɷ���SiO2��
��2��������������ʹ3 mol��VO2+��ΪVO2+������Ҫ������KClO3����Ϊ0.5mol��
��3�����к͡�����֮һ��ʹ����V4O124-��ʽ��������Һ�У�������2���к���Fe��OH��3��Al��OH��3��
��4�������ӽ������͡�ϴ�ѡ��ɼ�ʾΪ��4ROH+V4O124-$?_{ϴ��}^{���ӽ���}$R4V4O12+4OH-����ROHΪǿ���������ӽ�����֬����Ϊ�����ϴ��Ч�ʣ���ϴҺӦ�óʼ��ԣ���ᡱ������С�����
��5��������Һ����������������K+��
��6�����������õ�ƫ����泥�NH4VO3��������д�������ա��з�����Ӧ�Ļ�ѧ����ʽ2NH4VO3$\frac{\underline{\;����\;}}{\;}$V2O5+H2O��+2NH3����
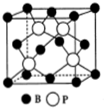 �����ס�����ͬ��Ԫ�أ�����Ԫ�ص��ʼ��仯������ũҩ�����ʵȷ�������ҪӦ�ã�
�����ס�����ͬ��Ԫ�أ�����Ԫ�ص��ʼ��仯������ũҩ�����ʵȷ�������ҪӦ�ã�