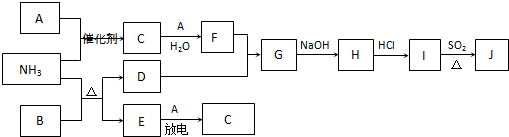
��Ŀ����
��12�֣����п�ͼ�漰������������Ԫ���У���һ��Ԫ���⣬�����Ϊ1��18��Ԫ�ء���֪��A��FΪ��ɫ���嵥�ʣ�BΪ���д̼�����ζ�����壬CΪ��ɫ�����EΪ��ɫ�������ʣ����ַ�Ӧ�IJ���δ��ȫ����
��ش��������⣺
��1��B�ĵ���ʽΪ ��
��2��J��K��ͬ�ֽ������Ȼ����KΪ��ɫ������д��SO2��ԭJ����K�����ӷ���ʽ ��
��3������β���г�����D��B��D�ڼ��Ⱥʹ������ڵ������£�������Ӧ�������ֶԿ�������Ⱦ�����ʡ���д���÷�Ӧ�Ļ�ѧ����ʽ ��
��4��������Ϊ�������滯ѧ���о��ɹ���ʹB��D�ķ�Ӧ�ڴ����������ʱ��Ч�ʴ����ߣ��Ӷ�ʹ��Ⱦ��D��ת���ʴ����ߡ�����Ӧ�û�ѧ�������۶Դ˹۵�������ۣ� ��
��1��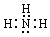 ��2��2Cu2����2Cl����SO2��2H2O��2CuCl����4H����SO42��
��2��2Cu2����2Cl����SO2��2H2O��2CuCl����4H����SO42��
��3��6NO��4NH3 5N2��6H2O
5N2��6H2O
��4�����о�ֻ�����ѧ��Ӧ���ʣ�����ʹ��ѧƽ�ⷢ���ƶ� ��ÿ��3�֣���12�֣�
����

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ