��Ŀ����
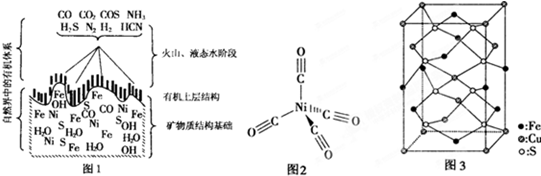
������Һ�о����ڵ�����е�ǰ�أ�������Һ�������ض������Χ������г��ܳ桢��桢���ࡢ�ݱ���ȶ�ֲ����˼��ѵ���Ȼ����ʵ���ң��Һ��ס�����ض����Χ������FeS����ͭ��п��ȿ��
��1���ӵ縺�ԽǶȷ�����C��N��OԪ�صķǽ�����������ǿ������˳��Ϊ ����һ�����ܴ�С��˳��Ϊ ��
��2��ͭԭ�Ӻ�������Ų�ʽΪ�� ��
��3��FeS��NaCl��Ϊ���Ӿ��壬�����ṹ���ƣ�ǰ���۵�Ϊ985��C������801��C����ԭ���� ��
��4���ӡ�����ض���γɵĿ�����ȡ�����轫��ת��Ϊ���ʻ��������ṹ��ͼ2���������������λ��Ϊ �����ʻ�����������ΪҺ̬��������CCl4�������л��ܼ�����̬���ʻ��������� ���壮
��5����ͭ��������Ҫ��ͭ��ȫ����
��ͭ�����������ģ���ͼ3Ϊ��ͭ��ľ�����
��д����ͭ��Ļ�ѧʽ ����ͭ��ľ���Ϊֱ�������Σ������������ⳤΪa cm����Ϊbcm����ͭ���Ħ������ΪMg/mol�������ӵ�����ΪNAmol-1�����ͭ������ܶ� g/cm3��

��1���ӵ縺�ԽǶȷ�����C��N��OԪ�صķǽ�����������ǿ������˳��Ϊ
��2��ͭԭ�Ӻ�������Ų�ʽΪ��
��3��FeS��NaCl��Ϊ���Ӿ��壬�����ṹ���ƣ�ǰ���۵�Ϊ985��C������801��C����ԭ����
��4���ӡ�����ض���γɵĿ�����ȡ�����轫��ת��Ϊ���ʻ��������ṹ��ͼ2���������������λ��Ϊ
��5����ͭ��������Ҫ��ͭ��ȫ����
| 2 |
| 3 |
��д����ͭ��Ļ�ѧʽ
���㣺�����ļ���,ԭ�Ӻ�������Ų�,Ԫ�ص����ܡ��縺�Եĺ��弰Ӧ��,�����ܵ�Ӧ��
ר�⣺ԭ�������ṹר��,��ѧ���뾧��ṹ
��������1��ͬһ�����У�Ԫ�صĵ縺������ԭ�����������������Ԫ�صķǽ���������ԭ�����������������һ������Ҳ����ԭ�����������������������ƣ�����IIA����VAԪ�ش�������Ԫ�صĵ�һ�����ܣ�
��2��ͭ��29��Ԫ�أ��������29�����ӣ����ݹ���ԭ����д���������Ų�ʽ��
��3�����Ӿ�����۵��뾧���ܳ����ȣ�
��4������ͼƬ֪����λ����4��������������ԭ���жϣ�
��5�����þ�̯��ȷ���仯ѧʽ��������=
���㣮
��2��ͭ��29��Ԫ�أ��������29�����ӣ����ݹ���ԭ����д���������Ų�ʽ��
��3�����Ӿ�����۵��뾧���ܳ����ȣ�
��4������ͼƬ֪����λ����4��������������ԭ���жϣ�
��5�����þ�̯��ȷ���仯ѧʽ��������=
| m |
| V |
���
�⣺��1��ͬһ�����У�Ԫ�صĵ縺������ԭ�����������������Ԫ�صķǽ���������ԭ�����������������һ������Ҳ����ԭ�����������������������ƣ�����IIA����VAԪ�ش�������Ԫ�صĵ�һ�����ܣ�����C��N��OԪ�صķǽ�����������ǿ������˳��Ϊ��O��N��C����һ�����ܴ�С��˳��ΪN��O��C
���ʴ�Ϊ��O��N��C��N��O��C��
��2��ͭ��29��Ԫ�أ��������29�����ӣ����ݹ���ԭ��֪���������Ų�ʽΪ��1s22s22p63s23p63d104s1��[Ar]3d104s1��
�ʴ�Ϊ��1s22s22p63s23p63d104s1��[Ar]3d104s1��
��3�����Ӿ�����۵��뾧���ܳ����ȣ�FeS��NaCl���۵�ֱ�Ϊ985��C��801��C����ԭ����FeS����ľ����ܱ�NaCl���徧���ܴʴ�Ϊ��FeS����ľ����ܱ�NaCl���徧���ܴ�
��4������ͼƬ֪����λ����4��������������ԭ���жϣ���̬���ʻ��������ڷ��Ӿ��壬�ʴ�Ϊ��4�����ӣ�
��5���þ����У�ͭԭ�Ӹ���=8��
+4��
+1=4����ԭ�Ӹ���=6��
+4��
=4����ԭ�Ӹ���=8�������仯ѧʽΪCuFeS2����=
=
=
g/cm3 ��
�ʴ�Ϊ��CuFeS2��
��
���ʴ�Ϊ��O��N��C��N��O��C��
��2��ͭ��29��Ԫ�أ��������29�����ӣ����ݹ���ԭ��֪���������Ų�ʽΪ��1s22s22p63s23p63d104s1��[Ar]3d104s1��
�ʴ�Ϊ��1s22s22p63s23p63d104s1��[Ar]3d104s1��
��3�����Ӿ�����۵��뾧���ܳ����ȣ�FeS��NaCl���۵�ֱ�Ϊ985��C��801��C����ԭ����FeS����ľ����ܱ�NaCl���徧���ܴʴ�Ϊ��FeS����ľ����ܱ�NaCl���徧���ܴ�
��4������ͼƬ֪����λ����4��������������ԭ���жϣ���̬���ʻ��������ڷ��Ӿ��壬�ʴ�Ϊ��4�����ӣ�
��5���þ����У�ͭԭ�Ӹ���=8��
| 1 |
| 8 |
| 1 |
| 2 |
| 1 |
| 2 |
| 1 |
| 4 |
| m |
| V |
| ||
| a2bcm3 |
| 4M |
| a2bNA |
�ʴ�Ϊ��CuFeS2��
| 4M |
| a2bNA |
���������⿼�������ʽṹ�����ʣ��漰֪ʶ��϶࣬��Щ����ѧϰ�ص㣬�ѵ����ܶȵļ��㣬�����������ʽ��������ѶȽϴ�

��ϰ��ϵ�д�
 ���Ǽ���С����ϵ�д�
���Ǽ���С����ϵ�д�
�����Ŀ
 ����˹ƥ�ַ���������NaOH��Һ����У��ܷ�����Ӧ�Ļ�ѧ���ǣ�������
����˹ƥ�ַ���������NaOH��Һ����У��ܷ�����Ӧ�Ļ�ѧ���ǣ�������| A��1��2 | B��2��3 |
| C��3��4 | D��1��4 |
�����������ڵ���ʵ��ǣ�������
| A������ͭ | B��ʳ��ˮ |
| C���ƾ� | D���Ȼ��ƾ��� |
���з�Ӧ�У�����������ԭ��Ӧ����ˮ�Ȳ������������ֲ��ǻ�ԭ�����ǣ�������
| A��SO3+H2O=H2SO4 |
| B��2Na2O2+2H2O=4NaOH+O2�� |
| C��2F2+2H2O=4HF+O2 |
| D��Mg+2H2O=Mg��OH��2+H2�� |
���и�������������Һ�д���������ǣ�������
| A��H+��Ba2+��NO3-��OH- |
| B��Mg2+��Na+��Cl-��OH- |
| C��Na+��H+��NO3-��CO32- |
| D��Cu2+��Fe3+��Cl-��SO42- |
���й��ڷ�ӦZn+2HNO3+NH4NO3�TN2��+3H2O+Zn��NO3��2��˵������ȷ���ǣ�������
| A��N2ֻ�ǻ�ԭ���Zn��NO3��2ֻ���������� |
| B��NH4NO3ֻ����������Zn�ǻ�ԭ�� |
| C����Ӧ������1 mol N2ʱ����5 mol���ӷ���ת�� |
| D��HNO3�ڷ�Ӧ��û�б��ֳ����� |
���з�Ӧ�����ӷ���ʽ��ȷ���ǣ�������
| A��CuSO4��Һ����H2S���壺Cu2++H2S��CuS��+2H+ | ||
B��Ca��HCO3��2��Һ��������NaOH��Һ��ϣ�Ca2++2HC
| ||
| C������������Һ�м����������ữ�Ĺ���������Һ��Fe2++2H++H2O2��Fe2++2H2O | ||
| D�����۵⻯����Һ�ڿ����б�����4I-+O2+2H2O��2I2+4OH- |


