��Ŀ����
��2��ȼ�ϵ�ؾ��й����ķ�չǰ;����ѧ�ҽ������Ƴ�һ���͵�ȼ�ϵ�أ����ü״�ȡ��������ȼ�Ͽ��Լ����ƣ��õ������ȡ����ͳ��أ�ijѧ����ʵ�������ü��Լ״�ȼ�ϵ�ص��Na2SO4��Һ�������ͼʾ�ش��������⣺
��ͼ��b�缫�� ������������������������������������������õ缫�Ϸ����ĵ缫��ӦʽΪ ��
�ڼ��������£�ͨ��״���һ�������ĵ缫��ӦʽΪ ��
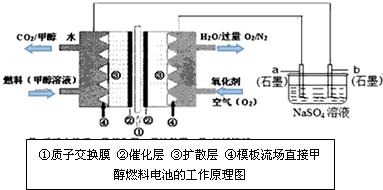
��ͼ��b�缫��
�ڼ��������£�ͨ��״���һ�������ĵ缫��ӦʽΪ

���㣺��ѧ��Դ���͵��
ר�⣺�绯ѧר��
��������1��ȼ�ϵ���У�ͨ��ȼ�ϵĵ缫�Ǹ�����ͨ�������ĵ缫�������������У��͵�Դ���������ļ����������͵�Դ������������������
��2��ȼ�ϵ�صĸ�������ȼ�Ϸ���ʧ���ӵ�������Ӧ�����ݵ���ʻ������ش�
��2��ȼ�ϵ�صĸ�������ȼ�Ϸ���ʧ���ӵ�������Ӧ�����ݵ���ʻ������ش�
���
�⣺��1��ȼ�ϵ���У�ͨ��ȼ�ϼ״��ĵ缫�Ǹ�����ͨ�������ĵ缫�������������У��͵�Դ���������ļ�b�������������������Һ�����������������ӷ����õ��ӵĻ�ԭ��Ӧ���õ缫�Ϸ����ĵ缫��ӦʽΪ��2H++2e-=H2�����ʴ�Ϊ��������2H++2e-=H2����
��2��ȼ�ϵ�صĸ�������ȼ�ϼ״�����ʧ���ӵ�������Ӧ������ʻ����Ǽ��ԣ���缫��ӦΪ��CH3OH+8OH--6e-=CO32-+6H2O��
�ʴ�Ϊ��CH3OH+8OH--6e-=CO32-+6H2O��
��2��ȼ�ϵ�صĸ�������ȼ�ϼ״�����ʧ���ӵ�������Ӧ������ʻ����Ǽ��ԣ���缫��ӦΪ��CH3OH+8OH--6e-=CO32-+6H2O��
�ʴ�Ϊ��CH3OH+8OH--6e-=CO32-+6H2O��
������������һ���Ȼ�ѧ�͵绯ѧ֪ʶ���ۺϿ����⣬ע��ԭ��غ͵��صĹ���ԭ���ǽ���ؼ����ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
��NA��ʾ�����ӵ�������ֵ������˵����ȷ���ǣ�������
| A����״���£�1 mol CO2��CO�Ļ�����к��е���ԭ����Ϊ1.5NA |
| B�����³�ѹ�£�33.6 L �Ȼ��������к��еķ�����Ϊ1.5NA |
| C��12g Mg������������̼��Ӧת�Ƶĵ�����ΪNA |
| D��1 mol?L-1 CuCl2��Һ�к��е���������Ϊ2NA |
���й���û�з�����ѧ��Ӧ���ǣ�������
| A���û���̿ȥ�������е���ζ |
| B�����ȼ�ˮ��������ϲ��������� |
| C���ý��ݹ����������Һ��שͷ����ˮ�� |
| D���ú��轺�����۵���С����ʳƷһ���ܷ��װ |
������Һ���й��������ʵ���Ũ�ȹ�ϵ��ȷ���ǣ�������
| A�������ʵ���Ũ�ȵ������NH4HSO4��NaOH��Һ��ϣ�c��Na+��=c��SO42-����c��NH4+����c��OH-����c��H+�� |
| B�������£���CH3COONa��Һ��ϡ����������ҺpH=7��c��Na+������CH3COO-����c��Cl-����c��CH3COOH����c��H+��=c��OH-�� |
| C�������£�pH=6��NaHSO3��Һ�У�c��SO32-��-c��H2SO3��=9.9��10-7 mol?L-1 |
| D�����ʵ���Ũ��֮��Ϊ1��2��NaClO��NaHCO3�����Һ�У�c��HClO��+c��ClO-��=2c��HCO3-��+2c��H2CO3��+2c��CO32-�� |
ȡһ����FeO��Fe2O3�Ļ�����H2���м��ȳ�ַ�Ӧ����ȴ���Ƶ�ʣ������ԭ�������������1.200g������ͬ���ĸû���������ᷴӦ��ȫ��������1mol/L��������Ϊ��������
| A��37.5mL |
| B��75mL |
| C��150mL |
| D��300mL |
������������ȶ���ǰ�߱Ⱥ���ǿ���ǣ�������
| A��CO��CO2 |
| B��GeO��GeO2 |
| C��SnO��SnO 2 |
| D��PbO��PbO2 |
����º����ܱ������м���1molHI���������з�Ӧ2HI��g���TH2��g��+I2��g������H��0���ﵽƽ������������ټ���1molHI���й�˵����ȷ���ǣ�������
| A��ƽ�ⲻ�ƶ���HIƽ��ת���ʲ��� |
| B��ƽ�������ƶ���HIƽ��ת���ʲ��� |
| C��ƽ�������ƶ���HIƽ��ת���ʼ�С |
| D��ƽ�������ƶ���HIƽ��ת�������� |
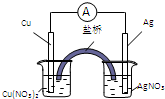 ��ͭƬ����Ƭ��Cu��NO3��2��Һ��AgNO3��Һ�����ߺ����ţ�ʢ��KNO3��֬��U�ܣ�����һ��ԭ��أ������йظ�ԭ��ص�������ȷ����
��ͭƬ����Ƭ��Cu��NO3��2��Һ��AgNO3��Һ�����ߺ����ţ�ʢ��KNO3��֬��U�ܣ�����һ��ԭ��أ������йظ�ԭ��ص�������ȷ����