��Ŀ����
19��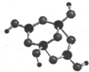 ��֪R��W��X��Y��Z�����ڱ���ǰ������Ԫ�أ����ǵ�ԭ���������ε�����R�Ļ�̬ԭ����ռ��������ԭ�ӹ���ĵ�����Ϊ1�� W���⻯��ķе��ͬ������Ԫ���⻯��ķе�ߣ�X2+��W2-������ͬ�ĵ��Ӳ�ṹ��YԪ�� ԭ�ӵ�3P�ܼ����ڰ����״̬��Z+�ĵ��Ӳ㶼�������ӣ���ش��������⣺
��֪R��W��X��Y��Z�����ڱ���ǰ������Ԫ�أ����ǵ�ԭ���������ε�����R�Ļ�̬ԭ����ռ��������ԭ�ӹ���ĵ�����Ϊ1�� W���⻯��ķе��ͬ������Ԫ���⻯��ķе�ߣ�X2+��W2-������ͬ�ĵ��Ӳ�ṹ��YԪ�� ԭ�ӵ�3P�ܼ����ڰ����״̬��Z+�ĵ��Ӳ㶼�������ӣ���ش��������⣺��1��д��Z�Ļ�̬ԭ�ӵ���Χ�����Ų�ʽ3d104s1��
��2��R��ij�����ξ��壬��������Am-����R��W��������Ԫ�أ������ģ������ͼ��ʾ����Am-�У�Rԭ�ӹ���ӻ�������SP2��SP3��m=2���������֣�
��3����X����̽����X��W�γɻ�����ľ���ṹ��NaCl�ľ���ṹ���ƣ�X2+����λԭ�� �����ɵ����弸�ι���Ϊ�������壮
��4����Z����������Һ�м��������ˮ��������[Z��NH3��4]S04������˵����ȷ����AD
A��[Z��NH3��4]SO4�������Ļ�ѧ�������Ӽ������Լ�����λ��
B����[Z��NH3��4]2+��Z2+�����¶Ե��ӣ�NH3�ṩ�չ��
C��[Z��NH3��4]SO4�����Ԫ���е�һ��������������Ԫ��
D��SO42-��PO43-��Ϊ�ȵ����壬�ռ乹�;�Ϊ������
��5������YCl5�Ľṹʵ������YCl4+��YCl6- ���ɵ����Ӿ��壬�侧��ṹ��CsCl���ƣ��������߳�Ϊa pm�������ܶ�Ϊ$\frac{417��1{0}^{30}}{{a}^{3}{N}_{A}}$g•cm-3��
���� R��W��X��Y��Z�����ڱ���ǰ������Ԫ�أ����ǵ�ԭ���������ε�����R�Ļ�̬ԭ����ռ��������ԭ�ӹ���ĵ�����Ϊ1����p�ܼ�ֻ��1�����ӣ�Rԭ�Ӻ�������Ų�Ϊ1s22s22p1����RΪ��Ԫ�أ�YԪ��ԭ�ӵ�3p�ܼ����ڰ����״̬��Yԭ�ӵĺ�������Ų�Ϊ1s22s22p63s23p3����YΪ��Ԫ�أ�W���⻯��ķе��ͬ������Ԫ���⻯��ķе�ߣ���������Ĵ��ڣ���X2+��W2-������ͬ�ĵ��Ӳ�ṹ����X���ڵ������ڡ�W���ڵڶ����ڣ���XΪMgԪ�ء�WΪ��Ԫ�أ�Z+�ĵ��Ӳ㶼�������ӣ����������Ϊ2��8��18����Zԭ�Ӻ��������Ϊ2+8+18+1=29����ZΪCuԪ�أ��Դ˽����⣮
��� �⣺R��W��X��Y��Z�����ڱ���ǰ������Ԫ�أ����ǵ�ԭ���������ε�����R�Ļ�̬ԭ����ռ��������ԭ�ӹ���ĵ�����Ϊ1����p�ܼ�ֻ��1�����ӣ�Rԭ�Ӻ�������Ų�Ϊ1s22s22p1����RΪ��Ԫ�أ�YԪ��ԭ�ӵ�3p�ܼ����ڰ����״̬��Yԭ�ӵĺ�������Ų�Ϊ1s22s22p63s23p3����YΪ��Ԫ�أ�W���⻯��ķе��ͬ������Ԫ���⻯��ķе�ߣ���������Ĵ��ڣ���X2+��W2-������ͬ�ĵ��Ӳ�ṹ����X���ڵ������ڡ�W���ڵڶ����ڣ���XΪMgԪ�ء�WΪ��Ԫ�أ�Z+�ĵ��Ӳ㶼�������ӣ����������Ϊ2��8��18����Zԭ�Ӻ��������Ϊ2+8+18+1=29����ZΪCuԪ�أ�
��1��ZΪCuԪ�أ�ԭ�Ӻ��������Ϊ29��ԭ�Ӻ�������Ų�ʽΪ1s22s22p63s23p63d104s1��Cu�Ļ�̬ԭ�ӵ���Χ�����Ų�ʽΪ3d104s1��
�ʴ�Ϊ��3d104s1��
��2��Hԭ�ӳ�1������Oԭ�ӳ�2������Bԭ��һ�����γ�3������B�γ�4����������1�����ܿ��ܾ�����λ�������3������Bԭ��ΪSP2�ӻ�����4������BΪSP3�ӻ���
�۲�ģ�ͣ���֪Am-�ǣ�H4B4O9��m-�����ݻ��ϼ�HΪ+1��BΪ+3��OΪ-2���ɵ�m=2��
�ʴ�Ϊ��SP2��SP3��2��
��3����X����̽����Mg��O�γɻ�����ľ���ṹ��NaCl�ľ���ṹ���ƣ�Mg2+����Χ��6��Oԭ�ӣ�Ϊ��ͼ�ṹ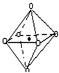 ������λԭ�������ɵ����弸�ι���Ϊ�������壬
������λԭ�������ɵ����弸�ι���Ϊ�������壬
�ʴ�Ϊ���������壻
��4��A��[Z��NH3��4]SO4�У��ڽ�����[Cu��NH3��4]2+���������SO42�γ����Ӽ���Cu2+��NH3�γ���λ����NH3��Nԭ����Hԭ��֮���γɼ��Լ�����A��ȷ��
B����[Z��NH3��4]SO4��Z2+�ṩ�չ����NH3�����¶Ե��ӣ���B����
C��[Z��NH3��4]SO4���Ԫ���е�һ������������NԪ�أ���C����
D��SO42-��PO43-��Ϊ�ȵ����壬�۲����������ͬ���ռ�ṹ��ͬ��SO42-��Sԭ�ӳ�4���Ҽ����¶Ե��Ӷ���Ϊ$\frac{6+2-4��2}{2}$=0���۲���Ӷ�Ϊ4��Ϊ�������壬��D��ȷ��
�ʴ�Ϊ��AD��
��5������PCl5�Ľṹʵ������PCl4+��PCl6-���ɵ����Ӿ��壬�侧��ṹ��CsCl���ƣ��������ΪP2Cl10���������൱�ں���2��PCl5���������߳�Ϊapm�������ܶ�Ϊ$\frac{\frac{208.5g/mol}{{N}_{A}mo{l}^{-1}}��2}{��a��1{0}^{-10}cm��^{3}}$=$\frac{417��1{0}^{30}}{{a}^{3}{N}_{A}}$g•cm-3��
�ʴ�Ϊ��$\frac{417��1{0}^{30}}{{a}^{3}{N}_{A}}$��
���� ���⿼���Ϊ�ۺϣ��漰��������Ų����ɡ������ӻ�����������ṹ�����ȣ�Ϊ�߿��������ͣ�������ѧ���ķ��������Ŀ��飬�ѶȽϴ�3����5��Ϊ�״��㡢�ѵ㣬��Ҫѧ��ʶ�dz�������ľ����ṹ��

 ��ʦָ����ĩ��̾�ϵ�д�
��ʦָ����ĩ��̾�ϵ�д� �����ܿ����ϵ�д�
�����ܿ����ϵ�д�| A�� | 9.0 | B�� | 0.9 | C�� | 0.45 | D�� | 4.5 |
| X | Y | ||
| W | Z | ||
| T |
| A�� | X��Y��Z����Ԫ����ͼ��⻯��ķе��������� | |
| B�� | ��X��Y��������Ԫ���γɵĻ�������ֻ�й��ۼ� | |
| C�� | ����WY2��WZ4�����۵�ߡ�Ӳ�ȴ������ | |
| D�� | TԪ�صĵ��ʿ��ܾ��а뵼������� |
| A�� | ��H2���ϵ�������At2��I2 | B�� | ���ڳ�����Ϊ��ɫ���� | ||
| C�� | AgAt������ˮ | D�� | ��������ˮ�����������Ȼ�̼ |
| A�� | ú�ĸ����ʯ�͵ķ��������ѧ�仯 | |
| B�� | Һ��ʯ��������Ȼ������Ҫ�ɷֶ��Ǽ��� | |
| C�� | ʯ���ѽ⡢�ѻ������������л��߷�������С���ӵĹ��� | |
| D�� | ú�Ϳ���ʯ�ͷ����ã�������ȼ�Ϻͱ������������� |
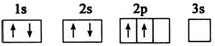
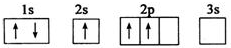
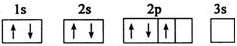
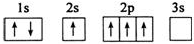
 ����A��B��C��D��E����Ԫ�أ�ԭ���������������Ҳ�����36��AԪ�صĻ�̬ԭ�������������Ǵ�����������BԪ�صĻ�̬ԭ�Ӻ�����13�ֲ�ͬ�˶�״̬�ĵ��ӣ�C��Bͬһ���ڣ�ԭ����δ�ɶԵ�������ͬ���������ģ�D2-�ĺ�������Ų����ԭ����ͬ��EԪ�صĻ�̬ԭ�Ӽ۵����Ų�ʽΪ3d104s1������������Ϣ���ش��������⣺
����A��B��C��D��E����Ԫ�أ�ԭ���������������Ҳ�����36��AԪ�صĻ�̬ԭ�������������Ǵ�����������BԪ�صĻ�̬ԭ�Ӻ�����13�ֲ�ͬ�˶�״̬�ĵ��ӣ�C��Bͬһ���ڣ�ԭ����δ�ɶԵ�������ͬ���������ģ�D2-�ĺ�������Ų����ԭ����ͬ��EԪ�صĻ�̬ԭ�Ӽ۵����Ų�ʽΪ3d104s1������������Ϣ���ش��������⣺