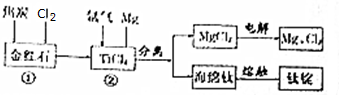
��Ŀ����
1��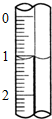 ��ʹ������к͵ζ����ⶨ���۰״�Ũ�ȣ�
��ʹ������к͵ζ����ⶨ���۰״�Ũ�ȣ���ʵ�鲽�裺
��1��ȡ10.00mL���۰״����ձ������������ƣ���ͬ���У���ˮϡ�ͺ�ת�Ƶ�100mL����ƿ�У�ϴ�ӡ�ת�ơ����ݡ�ҡ�ȼ��ô���״���Һ��
��2��ȡ����״���Һ20.00mL����ƿ�У������еμ�2�η�̪��ָʾ����
��3����ȡʢװ0.1000mol/L NaOH ��Һ�ĵζ��ܵij�ʼ���������Һ��λ������ͼ��ʾ�����ʱ�Ķ���Ϊ1.10mL��
��4���ζ�ʱһ�ֿ��ƻ�����һ����ס��ƿ������ҡ����˫��ע����ƿ����Һ����ɫ�仯������Һ����ɫǡ�ñ�Ϊdz��ɫ�����ڰ�����ڲ���ɫʱ��
ֹͣ�ζ�������¼NaOH��Һ���ն������ظ��ζ�3�Σ�
��ʵ���¼��
���� ���ݣ�mL�� | 1 | 2 | 3 |
| V����Ʒ�� | 20.00 | 20.00 | 20.00 |
| V��NaOH�������ģ� | 14.95 | 16.05 | 15.95 |
��5����ͬѧ�ڴ�������ʱ����ã�
ƽ�����ĵ�NaOH��Һ�����V=$\frac{��14.95+16.05+15.95��}{3}$=15.65��mL��
��ͬѧ��Ϊ��������ʱ��һ��ʵ�������Ӧ����ȥ��ԭ���ǵ�1������������Թ������쳣ֵ��Ӧ��ȥ��
����ȷ���ݴ������ɵ�c�����۰״ף�=0.80mol/L��
��6���ڱ�ʵ��ĵζ������У����в�����ʹʵ����ƫ�����ab����д��ţ���
a���ζ����ڵζ�ʱδ�ñ�NaOH��Һ��ϴ
b���ζ��ܵļ����ڵζ�ǰ�����ݣ��ζ���������ʧ
c����ƿ�м������״���Һ���ټ�����ˮ
d����ƿ�ڵζ�ʱ����ҡ����������Һ�彦����
���� ��1����Һϡ�����ձ��н��У���Һ�Ķ����ڶ�Ӧ���������ƿ�н��У�
��2��ǿ��ζ�����ʱӦѡ����Ա�ɫ��Χ�ڵ�ָʾ����̪��
��3�����ݵζ��ܵĽṹ�뾫ȷ����������
��4���ζ�ʱ��һ�ֿ��ƻ�����һ����ס��ƿ������ҡ����˫��ע����ƿ����Һ����ɫ�仯���ζ��յ�ʱ��Һ����ɫǡ�ñ�Ϊdz��ɫ�����ڰ�����ڲ���ɫ��
��5���������ݵĺ����������������ƽ�����ĵ�NaOH��Һ�������Ȼ����ݰ״���NaOH ��Һ��Ӧ�Ĺ�ϵʽ�����
��6������c�����⣩=$\frac{c��������V������}{V�����⣩}$������
��� �⣺��1���õζ��ܣ���10mL��Һ�ܣ�����ȡ10.00mLʳ�ð״ף����ձ�����ˮϡ�ͺ�ת�Ƶ�100mL ����ƿ�ж��ݣ�ҡ�ȼ��ô���״���Һ��
�ʴ�Ϊ���ձ�������ƿ��
��2��ʳ����NaOH��Ӧ������ǿ�������Σ���Һ�ʼ��ԣ�Ӧѡ����Ա�ɫ��Χ�ڵ�ָʾ����̪��
�ʴ�Ϊ����̪��
��3���ζ���Һ��Ķ���1.10mL��
�ʴ�Ϊ��1.10��
��4���ζ�ʱ��һ�ֿ��ƻ�����һ����ס��ƿ������ҡ����˫��ע����ƿ����Һ����ɫ�仯��NaOH�ζ�ʳ���յ�Ϊ����Һ����ɫǡ�ñ�Ϊdz��ɫ�����ڰ�����ڲ���ɫ��
�ʴ�Ϊ�����ƻ�������ס��ƿ������ҡ������ƿ����Һ����ɫ�仯����Һ����ɫǡ�ñ�Ϊ��ɫ�����ڰ�����ڲ���ɫ��
��5����1�εζ�������Դ����쳣ֵ��Ӧ��ȥ�����������ĵ�NaOH��Һ�����Ϊ��16.05mL��15.95mL����NaOH��Һ�������ƽ��ֵΪ16.00mL��
��20mL���۰״���Ʒ���� CH3COOH xmol����
CH3COOH��NaOH
1 1
x 0.1000mol/L��0.016L
x=0.0016mol
��Ʒ��Ũ��Ϊ$\frac{0.0016mol}{0.02L}$��10=0.80mol/L��
�ʴ�Ϊ����1�εζ�������Դ����쳣ֵ��Ӧ��ȥ��0.80��
��6��a����ʽ�ζ����ڵζ�ʱδ�ñ�NaOH��Һ��ϴ����ҺŨ�Ƚ��ͣ����V������ƫ����c�����⣩=$\frac{c��������V������}{V�����⣩}$������֪c�����⣩ƫ��a��ȷ��
b����ʽ�ζ��ܵļ����ڵζ�ǰ�����ݣ��ζ���������ʧ�����V������ƫ����c�����⣩=$\frac{c��������V������}{V�����⣩}$������֪c�����⣩ƫ��b��ȷ��
c����ƿ�м������״���Һ���ټ�����ˮ����V��������Ӱ�죬����c�����⣩=$\frac{c��������V������}{V�����⣩}$������֪c�����⣩���䣬��c����
d����ƿ�ڵζ�ʱ����ҡ����������Һ�彦��������Һ���ʵ���ƫС�����V������ƫС������c�����⣩=$\frac{c��������V������}{V�����⣩}$������֪c�����⣩ƫС����d����
�ʴ�Ϊ��ab��
���� ������Ҫ�����˺����IJⶨ���漰�к͵ζ�������ʵ��������ѡ�������㡢�������ȣ��Ѷ��еȣ�ע������к͵ζ�ʵ���ԭ����ע�����Ũ�ȹ�ʽ��������������ڣ�5�����״���

| A�� | 1�� | B�� | 2�� | C�� | 3�� | D�� | 4�� |
| A�� | NH4++2H2O?NH3•H2O+H3O+ | B�� | AlO2-+HCO3-+H2O�TAl��OH��3��+CO32- | ||
| C�� | HS-+H+?H2S | D�� | Cl2+H2O?H++Cl-+HClO |
| A�� | 6.5��þ��0.1molп | B�� | 1��þ��1��п | ||
| C�� | 0.1��þ��0.1molп | D�� | 10��þ��13��п |
| A�� | ���� | B�� | ̼ˮ������ | C�� | ̼�⻯���� | D�� | ���� |
| A�� | ŨH2SO4��Һ | B�� | FeCl3��Һ | C�� | KSCN��Һ | D�� | AgNO3��Һ |
| A�� |  | B�� |  | C�� |  | D�� |  |