��Ŀ����
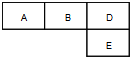 ��M��A��B��D��N��E���ֶ�����Ԫ�أ�ԭ��������������MԪ�صĵ�������Ȼ����������壬NԪ�ص�ԭ�Ӱ뾶����������ԭ�Ӱ뾶���ģ�A��B��D��E�ֱ����ұ������ڱ���һ���֣�ռ����Ӧ��λ�ã����ǵ�ԭ������֮��Ϊ37���Իش�
��M��A��B��D��N��E���ֶ�����Ԫ�أ�ԭ��������������MԪ�صĵ�������Ȼ����������壬NԪ�ص�ԭ�Ӱ뾶����������ԭ�Ӱ뾶���ģ�A��B��D��E�ֱ����ұ������ڱ���һ���֣�ռ����Ӧ��λ�ã����ǵ�ԭ������֮��Ϊ37���Իش���1����A��B��D��E����Ԫ�ص�����⻯���У��е���ߵ���
��2��A��B��D��M����ɶ���18���ӷ��ӣ���д��2�־���18���ӵĻ�����Ļ�ѧʽ����Ԫ�ط��ű�ʾ��
��3����A��D��N����Ԫ����ɵ������Է�������106������ˮ��Һ�ʼ��ԣ������ӷ���ʽ��ʾ��ԭ��
��4����D��N�γɵ�ԭ�Ӹ���1��1�Ļ�����ĵ���ʽΪ
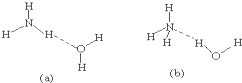
��5��NH3?H2O�ĵ��뷽��ʽΪNH3?H2O?NH4++OH-�����ж�NH3����ˮ���γɵ�NH3?H2O�ĺ����ṹ�ǣ�
��6��A��E������������Ӧ��ˮ���������Խ�ǿ����
д��������֤�ý��۵�һ�����ӷ�Ӧ����ʽ
���㣺Ԫ�������ɺ�Ԫ�����ڱ����ۺ�Ӧ��
ר�⣺Ԫ����������Ԫ�����ڱ�ר��
������MԪ�صĵ�������Ȼ����������壬MΪHԪ�أ�A��B��D��E��ԭ������֮��Ϊ37��������ǵ�λ�ù�ϵ��֪��Ϊ����������Ԫ�أ���A��ԭ������Ϊx����BΪx+1��DΪx+2��EΪx+10������x+x+1+x+2+x+10=37�����x=6����AΪC��BΪN��DΪO��EΪS��NԪ�ص�ԭ�Ӱ뾶����������ԭ�Ӱ뾶���ģ����ڢ�A�壬���M��A��B��D��N��Eԭ���������������֪����NӦΪ�������ڵڢ�A���Na��
��1�����ݳ������⻯���״̬�����жϷе㣻���Ӳ�ṹ��ͬ���˵����Խ�����Ӱ뾶ԽС�����Ӳ�Խ�����Ӱ뾶Խ��
��2��C��N��O��HԪ�ؿ���ɶ���18���ӷ��ӣ���C2H6��N2H4��H2O2��CH3OH��
��3����C��O��Na����Ԫ����ɵ������Է�������106����ΪNa2CO3����Һ��CO32-ˮ�⣬��Һ�ʼ��ԣ�
��4����O��Na�γɵ�ԭ�Ӹ���1��1�Ļ�����ΪNa2O2��������������������ӹ��ɣ���������������ԭ��֮���γɹ��ۼ���
��5����ˮ�ĵ�������NH4+��OH-��˵��NH3?H2O ��O-H���������ѣ���ȷ��NH3?H2O����������
��6���ǽ�����Խǿ����ۺ����������Խǿ��������ۺ������ǿ���Ʊ����ᷴӦ��֤��
��1�����ݳ������⻯���״̬�����жϷе㣻���Ӳ�ṹ��ͬ���˵����Խ�����Ӱ뾶ԽС�����Ӳ�Խ�����Ӱ뾶Խ��
��2��C��N��O��HԪ�ؿ���ɶ���18���ӷ��ӣ���C2H6��N2H4��H2O2��CH3OH��
��3����C��O��Na����Ԫ����ɵ������Է�������106����ΪNa2CO3����Һ��CO32-ˮ�⣬��Һ�ʼ��ԣ�
��4����O��Na�γɵ�ԭ�Ӹ���1��1�Ļ�����ΪNa2O2��������������������ӹ��ɣ���������������ԭ��֮���γɹ��ۼ���
��5����ˮ�ĵ�������NH4+��OH-��˵��NH3?H2O ��O-H���������ѣ���ȷ��NH3?H2O����������
��6���ǽ�����Խǿ����ۺ����������Խǿ��������ۺ������ǿ���Ʊ����ᷴӦ��֤��
���
�⣺MԪ�صĵ�������Ȼ����������壬MΪHԪ�أ�A��B��D��E��ԭ������֮��Ϊ37��������ǵ�λ�ù�ϵ��֪��Ϊ����������Ԫ�أ���A��ԭ������Ϊx����BΪx+1��DΪx+2��EΪx+10������x+x+1+x+2+x+10=37�����x=6����AΪC��BΪN��DΪO��EΪS��NԪ�ص�ԭ�Ӱ뾶����������ԭ�Ӱ뾶���ģ����ڢ�A�壬���ԭ��������֪����NӦΪ�������ڵڢ�A���Na��
��1���⻯���У������£�ֻ��H2OҺ�壬����Ϊ���壬��H2O�ķе���ߣ����Ӳ�ṹ��ͬ���˵����Խ�����Ӱ뾶ԽС�����Ӳ�Խ�����Ӱ뾶Խ�����Ӱ뾶��S2-��O2��Na+���ʴ�Ϊ��H2O��S2-��O2��Na+��
��2��C��N��O��HԪ�ؿ���ɶ���18���ӷ��ӣ���C2H6��N2H4��H2O2��CH3OH���ʴ�Ϊ��C2H6��H2O2��
��3����C��O��Na����Ԫ����ɵ������Է�������106����ΪNa2CO3����Һ��CO32-ˮ��CO32-+H2O?HCO3-+OH-���ƻ�ˮ�ĵ���ƽ�⣬��Һ�ʼ��ԣ�
�ʴ�Ϊ��CO32-+H2O?HCO3-+OH-��
��4����O��Na�γɵ�ԭ�Ӹ���1��1�Ļ�����ΪNa2O2��������������������ӹ��ɣ�����ʽΪ��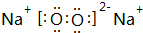 ����������������ԭ��֮���γɹ��ۼ����������Ӽ������ۼ���
����������������ԭ��֮���γɹ��ۼ����������Ӽ������ۼ���
�ʴ�Ϊ�� �����Ӽ������ۼ���
�����Ӽ������ۼ���
��5��NH3����ˮ���γɵ�NH3?H2O�У�NH3?H2O�ĵ��뷽��ʽΪNH3?H2O?NH4++OH-����֪�ṹ�к���笠����������Ļ����ṹ��Ӧ�ǰ�����Nԭ����ˮ��Hԭ��֮���γ������NH3?H2O�ĺ����ṹ��b���ʴ�Ϊ��b��
��6���ǽ�����S��C�ǽ�����Խǿ����ۺ����������Խǿ�������ԣ�H2SO4��H2CO3��������ۺ������ǿ���Ʊ����ᷴӦ��֤����Ӧ���ӷ���ʽΪ��CO32-+2H+=CO2��+H2O��
�ʴ�Ϊ��H2SO4��CO32-+2H+=CO2��+H2O��
��1���⻯���У������£�ֻ��H2OҺ�壬����Ϊ���壬��H2O�ķе���ߣ����Ӳ�ṹ��ͬ���˵����Խ�����Ӱ뾶ԽС�����Ӳ�Խ�����Ӱ뾶Խ�����Ӱ뾶��S2-��O2��Na+���ʴ�Ϊ��H2O��S2-��O2��Na+��
��2��C��N��O��HԪ�ؿ���ɶ���18���ӷ��ӣ���C2H6��N2H4��H2O2��CH3OH���ʴ�Ϊ��C2H6��H2O2��
��3����C��O��Na����Ԫ����ɵ������Է�������106����ΪNa2CO3����Һ��CO32-ˮ��CO32-+H2O?HCO3-+OH-���ƻ�ˮ�ĵ���ƽ�⣬��Һ�ʼ��ԣ�
�ʴ�Ϊ��CO32-+H2O?HCO3-+OH-��
��4����O��Na�γɵ�ԭ�Ӹ���1��1�Ļ�����ΪNa2O2��������������������ӹ��ɣ�����ʽΪ��
 ����������������ԭ��֮���γɹ��ۼ����������Ӽ������ۼ���
����������������ԭ��֮���γɹ��ۼ����������Ӽ������ۼ����ʴ�Ϊ��
 �����Ӽ������ۼ���
�����Ӽ������ۼ�����5��NH3����ˮ���γɵ�NH3?H2O�У�NH3?H2O�ĵ��뷽��ʽΪNH3?H2O?NH4++OH-����֪�ṹ�к���笠����������Ļ����ṹ��Ӧ�ǰ�����Nԭ����ˮ��Hԭ��֮���γ������NH3?H2O�ĺ����ṹ��b���ʴ�Ϊ��b��
��6���ǽ�����S��C�ǽ�����Խǿ����ۺ����������Խǿ�������ԣ�H2SO4��H2CO3��������ۺ������ǿ���Ʊ����ᷴӦ��֤����Ӧ���ӷ���ʽΪ��CO32-+2H+=CO2��+H2O��
�ʴ�Ϊ��H2SO4��CO32-+2H+=CO2��+H2O��
���������⿼��Ԫ�����ڱ���Ԫ���������ۺ�Ӧ�ã��Ѷ��еȣ����ضԻ���֪ʶ��Ӧ����ѧ�����������Ŀ��飮

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
����MgCl2��AlCl3��CuCl2��NaCl������Һ�����ֻ��һ���Լ��������������������Լ��ǣ�������
| A����ˮ |
| B��AgNO3 |
| C��NaOH |
| D��BaCl2 |