��Ŀ����
1��ijͬѧ��ʵ�����о�Na2CO3��NaHCO3�����ʼ��ת������1���ֱ���Ũ�Ⱦ�Ϊ0.1mol/LNa2CO3��NaHCO3��Һ�еμӼ��η�̪�Լ���
��Na2CO3��Һ����ɫ��Ϊ��ɫ����ԭ����CO32-+H2O?HCO3-+OH-��
��NaHCO3��ҺҲ����ɫ��Ϊ��ɫ����ԭ����HCO3-+H2O?H2CO3+OH-��HCO3-?H++CO32-��ˮ��̶ȴ��ڵ���̶ȣ�
�۱Ƚ�������Һ����ɫ����ɫ��dz����NaHCO3���ѧʽ����
��2���о�NaHCO3��CaCl2�ķ�Ӧ��
| ʵ����� | ʵ����� | ʵ������ |
| ʵ��1 | ��2mL 0.001mol/L NaHCO3��Һ�м���1mL 0.1mol/LCaCl2��Һ | �����Ա仯���ü�������䣬û�г��ֹ�����ͨ·�� |
| ʵ��2 | ��2mL 0.1mol/L NaHCO3��Һ�м���1mL 0.1mol/LCaCl2��Һ | ����ɫ���ȵĻ��ǣ��ü�������䣬���ֹ�����ͨ·�� |
��ʵ��2���γɵķ�ɢϵ���ڽ��壮
�������ӷ���ʽ��ʾʵ��2�з����ķ�ӦCa2++2HCO3-=CaCO3��+CO2+H2O��
��3���ڲ������������ʵ�ǰ���£�ʵ��Na2CO3��NaHCO3֮���ת��������ͼ�б�����õ��Լ������������ʵ�Ũ�ȣ���Ӧǰ����Һ����ı仯�ɺ��ԣ���
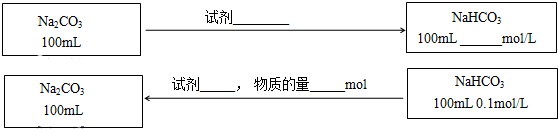
���� ��1��Ũ�Ⱦ�Ϊ0.1mol/LNa2CO3��NaHCO3��Һ��̼�������ˮ����Һ�ʼ��ԣ�̼���������ˮ��̶ȴ��ڵ���̶ȣ�������ҺҲ�ʼ��ԣ����Ũ��ʱ��Na2CO3ˮ���������������Һ����NaHCO3ˮ�������������������Na2CO3��Һ�ļ���ǿ��NaHCO3����Һ��
��2���������е�ʵ�������֪��0.001mol/L NaHCO3��Һ�е������CO32-Ũ�ȸ��ͣ�����c��Ca2+��•c��CO32-����Ksp��CaCO3�������Һ�в����ֳ�����0.1mol/L NaHCO3��Һ�е������̼�������Ũ�ȸ�һЩ����������γ���̼��Ƴ��������壻
��3����̼������Һ��ͨ�������̼���Խ�̼����ת����̼�����ƣ���̼��������Һ�м����������ƿ��Խ�̼������ת��Ϊ̼���ƣ����ݸ����ʵ����Ĺ�ϵ��ȷ���й����ʵ����ʵ�����Ũ�ȣ�
��� �⣺��1��Ũ�Ⱦ�Ϊ0.1mol/LNa2CO3��NaHCO3��Һ��̼�������ˮ����Һ�ʼ��ԣ�̼���������ˮ��̶ȴ��ڵ���̶ȣ�������ҺҲ�ʼ��ԣ����Ũ��ʱ��Na2CO3ˮ���������������Һ����NaHCO3ˮ�������������������Na2CO3��Һ�ļ���ǿ��NaHCO3����Һ�������Т�Na2CO3��Һ����ɫ��Ϊ��ɫ����ԭ���� CO32-+H2OHCO3-+OH-����NaHCO3��ҺҲ����ɫ��Ϊ��ɫ����ԭ���� HCO3-+H2O?H2CO3+OH-��HCO3-?H++CO32-��ˮ��̶ȴ��ڵ���̶ȣ��۱Ƚ�������Һ����ɫ����ɫ��dz����NaHCO3��
�ʴ�Ϊ����CO32-+H2O?HCO3-+OH-����HCO3-+H2O?H2CO3+OH-��HCO3-?H++CO32-��ˮ��̶ȴ��ڵ���̶ȣ���NaHCO3��
��2���������е�ʵ�������֪��0.001mol/L NaHCO3��Һ�е������CO32-Ũ�ȸ��ͣ�����c��Ca2+��•c��CO32-����Ksp��CaCO3�������Һ�в����ֳ�����0.1mol/L NaHCO3��Һ�е������̼�������Ũ�ȸ�һЩ����������γ���̼��Ƴ��������壬������
��ʵ��1��û�г��ְ�ɫ���ǵ�ԭ���� HCO3-Ũ�ȵͣ��������CO32-Ũ�ȸ��ͣ�����c��Ca2+��•c��CO32-����Ksp��CaCO3����
�ʴ�Ϊ��HCO3-Ũ�ȵͣ��������CO32-Ũ�ȸ��ͣ�����c��Ca2+��•c��CO32-����Ksp��CaCO3����
���ü�������䣬���ֹ�����ͨ·Ϊ��������ʣ�����ʵ��2���γɵķ�ɢϵ���ڽ��壬
�ʴ�Ϊ�����壻
��ʵ��2�з����ķ�Ӧ�����ӷ���ʽΪCa2++2HCO3-=CaCO3��+CO2+H2O��
�ʴ�Ϊ��Ca2++2HCO3-=CaCO3��+CO2+H2O��
��3��100mL0.1mol/LNa2CO3����ͨ��CO2����̼�����ƣ�������Ԫ���غ��֪���õ���̼��������Һ��Ũ��Ϊ0.2mol/L����100mL0.1mol/LNaHCO3��Һת��Ϊ̼������Һ�������0.01mol���������ƹ��弴�ɣ�
�ʴ�Ϊ��CO2��0.2��NaOH��0.01��
���� ������Ҫ��������ˮ���������ʵĵ���ȣ��Ѷ��еȣ�ע��̼������̼�����Ƶ����ʵ�������ã�

| A�� | Na2O2+2H2O�T2Na++2OH-+H2�� | B�� | 2O22-+4H+�T4OH-+O2�� | ||
| C�� | Na2O2+2H2O�T2Na++2OH-+O2�� | D�� | 2Na2O2+2H2O�T4Na++4OH-+O2�� |
| A�� | ��ϩ����Ȳ | B�� | 1-��ϩ���ױ� | C�� | ���������� | D�� | ���� ��Ȳ |
| A�� | С�մ�NaHCO3 | B�� | �ռ�NaOH | C�� | �̷�FeSO4•7H2O | D�� | ����BaCO3 |
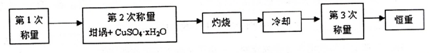
��1�����������ͭ����Ļ�ѧʽΪCuSO4•xH2O���������ͭ��������ʧȥȫ���ᾧˮ�Ļ�ѧ����ʽΪCuSO4•xH2O$\frac{\underline{\;\;��\;\;}}{\;}$CuSO4+xH2O��

a���в� b������ c������ d�����Ǽ�
��2��������ʵ�����õ��ļ����������������Ӧ������ȷ����ac��������ţ�
��3������ȴ������Ӧ�ڸ����������������ƣ��н��У������ա�ʱ��Դѡ�õ��Ǿƾ��ƶ����Ǿƾ���ƣ������Ǿƾ�����¶�̫�ߣ�����ʹCuSO4���ȷֽ⣮
��4�����ز�����Ŀ����ȷ������ͭ����ʧˮ��ȫ��
��5���ⶨ����ͭ����ᾧˮ����ʱ����ʵ����ƫ�ͣ�ԭ�������d��������ţ�
a������δ��ȫ����b�����ȹ����о�����������ʧ
c��������մ�����Ȳ��ֽ������d�����Ⱥ�δ�������������ȴ
��6��������ijѧ��ʵ���һ�����ݣ�����ɼ���
| ����ǰ���� | ���Ⱥ����� | ||
| m1 | m2 | m3 | m4 |
| 11.721g | 22.692g | 18.631g | a |
��2�������Ⱥ����������������Ϊ18.631g��ʵ��ֵx=5.22��������λС����
��3��������ֵx=5�����ʵ�����������4.4%��
| A�� | ��AlCl3��Һ�еμӰ�ˮ���������ɫ�������ټ���NaHSO4����������ʧ | |
| B�� | Ũ��NaOH��Һ��������Ƥ���ϣ������ô���ˮ��ϴ����Ϳ�������ϡ������Һ | |
| C�� | AlCl3��Һ�м������NaF���壬�ٵμӰ�ˮ����������˵��[AlF6]3-���ѵ��� | |
| D�� | ����Cl-ʱ��Ϊ���ų�SO42-�ĸ��ţ����ȵμӹ�����Ba��NO3��2��Һ��ȡ�ϲ���Һ���ٵμ�AgNO3��Һ�����а�ɫ�����������ԭ��Һ����Cl-���� |
| A�� | ��ij��ɫ��Һ�м��������ܲ�������H2�������Һ�д��ڴ���Na+��Ca2+��C1-��NO3- | |
| B�� | ˮ�������c��H+��=1��10-3mol•L-1��ˮ��Һ��Na+��CO32-��Cl-��K+ | |
| C�� | pH=1��ˮ��Һ��NH4+��Cl-��Mg2+��SO42- | |
| D�� | Al3+��HCO3-��I-��Ca2+ |
 CO2����Ȼ����̼ѭ������Ҫ���壬ά�ִ�����CO2��ƽ�����̬��������Ҫ���壮
CO2����Ȼ����̼ѭ������Ҫ���壬ά�ִ�����CO2��ƽ�����̬��������Ҫ���壮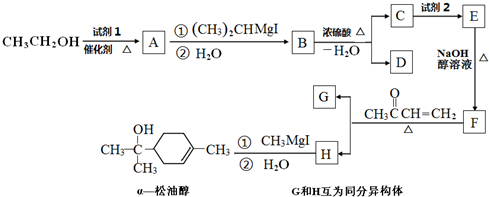
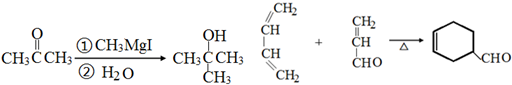
 ��
��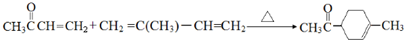 ��
��