��Ŀ����
6��Ϊ��֤Cl2��Br2��������ǿ�������������ʵ�飺���ɹ�ѡ����Լ��У���ˮ����ˮ��NaCl��Һ��NaBr��Һ���ƾ������Ȼ�̼��ʵ�鲽����ͼ����дͼ�еĿհף�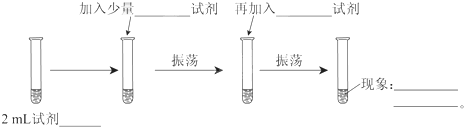
��1��2mL �Լ�NaBr��Һ������������ˮ�Լ����ټ������Ȼ�̼
�Լ��� ������Һ�ֲ㣬�²�Ⱥ�ɫ���ϲ���ɫ��
��2������֤��Cl2��������ǿ�ڣ��ǿ�ڡ������ڡ���Br2�������ԣ���Ԫ�صķǽ�����ǿ�ڣ��ǿ�ڡ������ڡ�����Ԫ�صķǽ����ԣ�
�� 3 ���˷�Ӧ�����ӷ���ʽΪ��Cl2+2Br-=Br2+2Cl-��
��4��ij��Һ��Cl-��Br-��I-�����ʵ���֮��Ϊ4��1��4��Ҫʹ��Һ�е�Cl-��Br-��I-�����ʵ���֮�ȱ�Ϊ7��1��1����ͨ��Cl2�����ʵ�����ԭ��Һ��I-�����ʵ���֮��Ϊ3��8��
���� ����ͬһ������ԭ��Ӧ���������������Դ�����������������������ʵ�飬Ҫ��Ƚ������������Դ�����������ԣ�ֻҪʹ�������������Һ��Ϲ۲��Ƿ����嵥�����ɣ�������嵥�����ɾ�˵�������������Դ����嵥�ʣ�������֤������ԭ�Դ�СΪI-��Br-��Cl-��ͨ��һ������Cl2ʹ��ֵ��Ϊ1��1��1��˵����Һ����ʣ���I-����Ӧ��ֻ����I-����Һ��Br-���䣬�迪ʼCl-��Br-��I-�������ʵ����ֱ���4mol��1mol��4mol�����㷴Ӧ������ӵ����ʵ���������Cl-�����ʵ����仯����μӷ�Ӧ��������
��� �⣺��1��Ҫ��Ƚ��������嵥�ʵ������Դ�С��������ʵ�飺
�����廯�Ʒ�Ӧ����������ˮ������Һ�ɻ���ɫ��ɳ�ɫ�����������Ȼ�̼������������Һ�ֲ㣬�²�Ⱥ�ɫ���ϲ���ɫ����˵�������������ӷ�Ӧ����֤�������������Դ����壬�嵥�������Ȼ�̼�е��ܽ�ȴ�����ˮ�е��ܽ�ȣ�ˮ�����Ȼ�̼�ǻ������ܵģ����Իῴ������Һ�ֲ㣬�²�Ⱥ�ɫ���ϲ���ɫ��
�ʴ�Ϊ��NaBr��Һ����ˮ�����Ȼ�̼����Һ�ֲ㣬�²�Ⱥ�ɫ���ϲ���ɫ��
��2��ͨ������ʵ��֪�������ܽ��嵥���û������������������Դ����壬���ӷ���ʽΪ��Cl2+2Br-=Br2+2Cl-������������Խǿ��Ӧ�ǽ�����Խǿ������Ԫ�صķǽ�����ǿ����Ԫ�صķǽ����ԣ��ʴ�Ϊ��ǿ�ڣ�
��3�������ܽ��嵥���û������������������Դ����壬���ӷ���ʽΪ��Cl2+2Br-=Br2+2Cl-��
�ʴ�Ϊ��Cl2+2Br-=Br2+2Cl-��
��4����ԭ�ԣ�I-��Br-��Cl-��ͨ��һ������Cl2��Cl-��Br-��I-�������ʵ���֮����4��1��4��Ϊ7��1��1��˵����Һ����ʣ���I-������Һ�е�Br-����Ӧ��
�迪ʼCl-��Br-��I-�������ʵ����ֱ���4mol��1mol��4mol����Ӧ��ֱ�Ϊ7mol��1mol��1mol����Cl-����7mol-4mol=3mol��
�ɷ���ʽ2I-+Cl2=I2+2Cl-��֪�μӷ�Ӧ��Cl2Ϊ��3mol��$\frac{1}{2}$=1.5mol��
����ͨ��Cl2�����ʵ�����ԭ��Һ��I-���ʵ���֮��Ϊ��$\frac{1.5mol}{4mol}$=$\frac{3}{8}$��
�ʴ�Ϊ��$\frac{3}{8}$��
���� ���⿼����̽��±�ص��ʼ���û���Ӧ����ȷʵ��Ŀ�ġ���ȷѡȡʵ��ҩƷ���ܸ���ʵ������ó������ǽⱾ��Ĺؼ�����ѧ����˵�Ѷ��еȣ�

 ��ĩ���䵥Ԫ�����ิϰ��ϵ�д�
��ĩ���䵥Ԫ�����ิϰ��ϵ�д�| ԭ�� | �˵���� | ������ | ������ | ������ | ������ | ��������� |
| A | 4 | 8 | ||||
| B | 6 | 8 | ||||
| C | 12 | 2��8��1 | ||||
| D | 16 | 16 | ||||
| E | 16 | 8 | ||||
| F | 27 | 2��8��3 |
��2��д��Dԭ�ӵĽṹʾ��ͼ

��3��д��E���ӵĵ���ʽ
 ��
�� 
| A�� | aΪþ�� | |
| B�� | bΪ�����������۵Ļ���� | |
| C�� | 2 mol���������뷴Ӧ�����ת��6 NA�� | |
| D�� | ��ԭ����������������ʵ���֮��Ϊ1��2 |
| A�� | �٢ڢۢ� | B�� | �ڢۢݢ� | C�� | �٢ܢݢ� | D�� | �ۢܢݢ� |
| �� ���� | IA | IIA | IIIA | IVA | VA | VIA | VIIA | 0 |
| 2 | �� | �� | ||||||
| 3 | �� | �� | �� | �� | �� | |||
| 4 | �� | �� | �� |
��1���ڢۡ���Ԫ���У�ԭ�Ӱ뾶������Ca����Ԫ�ط��ţ���
��2���١�����Ԫ������������Ӧ��ˮ������������ǿ����HClO4�������ʻ�ѧʽ����
д���ٺ͢ݵ�����������ˮ���ﷴӦ�����ӷ���ʽOH-+Al��OH��3=AlO2-+2H2O��
��3����Ԫ�ص�����������Ӧˮ���������⻯����������M��M�к��еĻ�ѧ�����������Ӽ��������ԣ����ۼ���
��4��Ԫ�آ�����γɻ�����ĵ���ʽΪ
 ��
����5��д����ҵұ���ݵĻ�ѧ����ʽ2Al2O3$\frac{\underline{\;\;\;���\;\;\;}}{����ʯ}$4Al+3O2����
| A�� | �� | B�� | ������ | C�� | �ռ� | D�� | ������ |
| A�� | 18 g H2O�к��е�������Ϊ8NA | |
| B�� | 12 g���ʯ�к��е�̼ԭ����Ϊ4NA | |
| C�� | 46 g NO2��N2O4��������к���ԭ������Ϊ3NA | |
| D�� | 1 mol Na������O2��Ӧ������Na2O��Na2O2�Ļ�����ʧȥ2NA������ |