��Ŀ����
8����ⷨ�ٽ����ʯ����Ҫ�ɷ���Mg2SiO4���̶�CO2�IJ��ֹ���������ͼ1����֪��Mg2SiO4��s��+4HCl��aq���T2MgCl2��aq��+SiO2 ��s��+2H2O��l����H=-49.04kJ•mol-1
ע����ʽ̼��þ3MgCO3•Mg��OH��2•3H2O
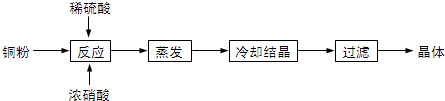
��1����̼ʱ��Ҫ��Ӧ�ķ���ʽΪNaOH��aq��+CO2 ��g��=NaHCO3 ��aq�����÷�Ӧ���÷�Ӧ����Ҫ�ǹ�ҵ��ͨ����ⷨ�õ�������ͼ1����ڲ���һ����ҵ��������
 ��
����2������ͼ����������Ҫ�ɷ���SiO2��
��3��д����Ӧ�����ӷ���ʽ3HCO3-+4Mg2++5H2O+5NH3�TMg2��OH��2CO3��+5 NH4+��
��4������������Ҳ����������̼������c��������ĸ��
a��CaCl2 b��CH3COONa c����NH4��2CO3
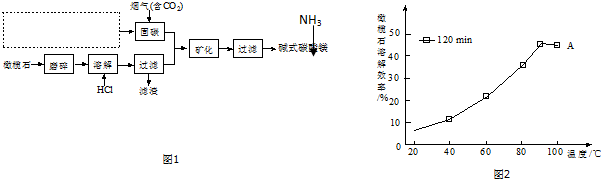
��5����ͼ2��֪��90�������A�ܽ�Ч���½���������ԭ��120min���ܽ�ﵽƽ�⣬����Ӧ���ȣ�����ƽ�������ƶ����ܽ�Ч�ʽ��ͣ�
��6�������������ü�ʽ̼��þ��Ʒ�к�������NaCl��Fe2O3��Ϊ�ᴿ���ɲ�ȡ�Ĵ�ʩ����Ϊ�����ܽ��������Һ���г����������Բ�Ʒ����ϴ�Ӵ������жϲ�Ʒϴ���IJ�����ȡ�������һ�ε�ϴ��Һ���������ữ����������Һ������������������ϴ����
���� ������ͼ����̼ʱ��Ҫ��Ӧ�ķ���ʽΪNaOH��aq��+CO2 ��g��=NaHCO3 ��aq������ҵ�����ȵ�ⱥ��ʳ��ˮ�����ռȱ���ռ����ȡ���̣��Ȼ�����Һ���õ�����������������������Һ����ѧ��Ӧ����ʽΪ��2NaCl+2H2O$\frac{\underline{\;���\;}}{\;}$2NaOH+H2��+Cl2������������������Һ��̼������̼�����Σ����ʯ����Ҫ�ɷ�ΪMg2SiO4�����Կ���MgO��SiO2��ɣ������ʯĥ����ٽ����ܽ��Ŀ��������Ӵ�������ӿ��ܽ����ʣ�ĥ����������ܽ⣬Mg2SiO4+4HCl�T2MgCl2+SiO2+2H2O�����˵õ���Һ�Ȼ�þ�������������裬��Ͽ�3HCO3-+4Mg2++5H2O+5NH3�TMg2��OH��2CO3��+5 NH4+���õ���ʽ̼��þ��
��1��������ͼ����̼ʱ���������ƣ���ҵ�����ȵ�ⱥ��ʳ��ˮ����ռ
��2�����ʯ����Ҫ�ɷ�ΪMg2SiO4�����Կ���MgO��SiO2��ɣ����������������Ӧ��
��3����ӦΪþ���Ӻ�̼��������ӷ�Ӧ���ɼ�ʽ̼��þ��
��4�������ܺͶ�����̼֮�䷴Ӧ�����������̶�������̼���ش�
��5�������¶ȶԻ�ѧ��Ӧƽ���ƶ���Ӱ��֪ʶ���ش�
��6�������ӵļ����������ữ����������
��� �⣺��1����������ͼ����̼ʱ��Ҫ��Ӧ�ķ���ʽΪNaOH��aq��+CO2 ��g��=NaHCO3 ��aq����ȱ���ռ����ȡ���̣���ҵ�����ȵ�ⱥ��ʳ��ˮ�����ռ��ѧ��Ӧ����ʽΪ��2NaCl+2H2O$\frac{\underline{\;���\;}}{\;}$2NaOH+H2��+Cl2��������ͼΪ ��
��
�ʴ�Ϊ�� ��
��
��2�����ʯ����Ҫ�ɷ�ΪMg2SiO4�����Կ���MgO��SiO2��ɣ������ʯĥ����ٽ����ܽ��Ŀ��������Ӵ�������ӿ��ܽ����ʣ�ĥ����������ܽ⣬Mg2SiO4+4HCl�T2MgCl2+SiO2+2H2O�����˵õ���Һ�Ȼ�þ�������������裬
�ʴ�Ϊ��SiO2��
��3�����ʯ�������ܽ⣬���˵õ���Һ�Ȼ�þ�����̼��IJ����Ͽ�������ӦΪ��3HCO3-+4Mg2++5H2O+5NH3�TMg2��OH��2CO3��+5 NH4+���õ���ʽ̼��þ��
�ʴ�Ϊ��3HCO3-+4Mg2++5H2O+5NH3�TMg2��OH��2CO3��+5 NH4+��
��4�������������У�ֻ�У�NH4��2CO3���ԺͶ�����̼֮�䷴Ӧ����̼����泥�����������̼�����Լ���
�ʴ�Ϊ��c��
��5��1 ͼ����ʾ�����Լ����߱仯֪����20min���ܽ�ﵽƽ�⣬���÷�Ӧ�Ƿ��ȣ����£�ƽ�������ƶ������ܽ�Ч�ʽ��ͣ�
�ʴ�Ϊ��120min���ܽ�ﵽƽ�⣬����Ӧ���ȣ�����ƽ�������ƶ����ܽ�Ч�ʽ��ͣ�
��6���жϲ�Ʒϴ��ֻ��Ҫ����ϴ��Һ�в����������Ӽ��ɣ������ӵļ����������ữ���������������ǣ�ȡ�������һ�ε�ϴ��Һ���������ữ����������Һ������������������ϴ����
�ʴ�Ϊ��ȡ�������һ�ε�ϴ��Һ���������ữ����������Һ������������������ϴ����
���� ������һ����ѧ��������ϵĹ��������⣬�����ڿ��Ե��ȵ㣬ע��֪ʶ��Ǩ�ƺ����Ӧ���ǽ���Ĺؼ�����Ŀ�Ѷ��еȣ�

| A�� | Zn��Fe | B�� | Zn��Cu | C�� | Fe��Cu | D�� | Zn��Fe��Cu |
| A�� | b��a��c | B�� | a��c��b | C�� | c��a��b | D�� | c��b��a |
��֪����NaClO2���ܽ�����¶����߶������ʵ������¿ɽᾧ����NaClO2•3H2O��
��Ksp��FeS��=6.3��10-18�� Ksp��CuS��=6.3��10-36��Ksp��PbS��=2.4��10-28
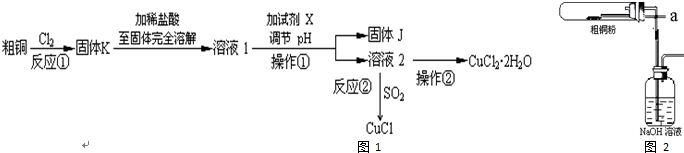
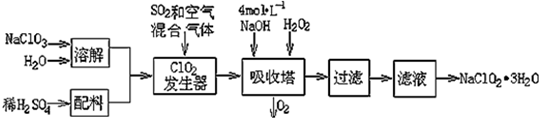
��1���������ڷ�����Ӧ�����ӷ���ʽΪ2NaOH+2ClO2+H2O2=2NaClO2+2H2O+O2���ù��������е�NaClO3��ClO2��NaClO2����ǿ�����������Ƕ��ܺ�Ũ���ᷴӦ��ȡCl2�����ö������Ⱥ�Ũ������ȡCl2��������5mol Cl2ʱ��ͨ����ԭ��Ӧ�Ƶ�����������Ϊ71g��
��2������Һ�еõ�NaClO2•3H2O������������������dc ����д��ţ���
a������ b������ c������ d����ȴ�ᾧ e������
��3��ӡȾ��ҵ�����������ƣ�NaClO2��Ư��֯�Ư��֯��ʱ���������õ���HClO2��
���� 25��ʱHClO2�����ֳ�������ĵ���ƽ�ⳣ����
| ���� | HClO2 | HF | HCN | H2S |
| Ka | 1��10-2 | 6.3��10-4 | 4.9��10-10 | K1=9.1��10-8 K2=1.1��10-12 |
��Na2S�dz��õij�������ij��ҵ��ˮ�к��е�Ũ�ȵ�Cu2+��Fe2+��Pb2+���ӣ��μ�Na2S��Һ�����������ij�����CuS�������һ�����ӳ�����ȫʱ��������Ũ��Ϊ10-5mol•L-1������ʱ��ϵ�е�S2-��Ũ��Ϊ6.3��10-13mol/L��
��֪���ٻ���������Cl2��Ӧ����PCl3�������Cl2��Ӧ����PCl5��
��PCl3��ˮ��ǿ��ˮ������ H3PO3��HC1��
��PCl3��O2������P0Cl3��P0Cl3����PCl3��
��PCl3��POCl3���۷е���±���
| ���� | �۵�/�� | �е�/�� |
| PCl3 | -112 | 75.5 |
| POCl3 | 2 | 105.3 |
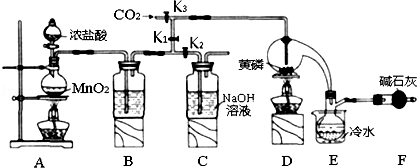
��1��Aװ���������������ӷ���ʽΪMnO2+4H++2Cl-$\frac{\underline{\;\;��\;\;}}{\;}$Mn2++Cl2��+2H2O��
��2��װ��F�������Ǹ���ܣ�����װ�ļ�ʯ�ҵ����������ն������������ֹ�����е�H2O������ƿ��PCl3 ��Ӧ��
��3��ʵ��ʱ�����װ�������Ժ��ȴ�K3ͨ������CO2����Ѹ�ټ�����ף�ͨ����CO2���������ž�װ���еĿ�������ֹ������ȼ��
��4���ֲ�Ʒ�г�����POCl3��PCl5�ȣ���������ȳ�ȥPCl5��ͨ��������ʵ��������ƣ������ɵõ��ϴ�����PCl3��
��5��ʵ�����ʱ����������C�е��Լ����ն����������C�з�Ӧ�����ӷ���ʽΪCl2+2OH-=Cl-+ClO-+2H2O��
��6��ͨ�����淽���ɲⶨ��Ʒ��PCl3����������
��Ѹ�ٳ�ȡ1.00g��Ʒ����ˮ��Ӧ�����250mL��Һ��
��ȡ������Һ25.00mL�������м���10.00mL 0.1000mol•L-1��ˮ����ַ�Ӧ��
�����������Һ�м��뼸�ε�����Һ����0.1000mol•L-1��Na2S2O3����Һ�ζ���
���ظ��ڡ��۲�����ƽ������Na2S2O3��Һ8.40ml��
��֪��H3PO3+H2O+I2�TH3PO4+2HI��I2+2Na2S2O3�T2NaI+Na2S4O6������ⶨ������û��������Ӧ�������������ݣ��ò�Ʒ��PC13����������Ϊ79.75%��
 ����ȩ��
����ȩ��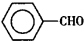 ���������ᣨ
���������ᣨ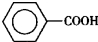 ��������Ҫ�Ļ���ԭ�ϣ������üױ���
��������Ҫ�Ļ���ԭ�ϣ������üױ��� ��Ϊԭ���������±��г����й����ʵIJ����������ʣ���ش�
��Ϊԭ���������±��г����й����ʵIJ����������ʣ���ش�| ���� | ��״ | �۵㣨�棩 | �е㣨�棩 | ����ܶ� ��ˮ=1g/cm3 | �ܽ��� | |
| ˮ | �Ҵ� | |||||
| �ױ� | ��ɫҺ����ȼ�ӷ� | -95 | 110.6 | 0.8660 | ���� | ���� |
| ����ȩ | ��ɫҺ�� | -26 | 179 | 1.0440 | �� | ���� |
| ������ | ��ɫƬ״����״���� | 122.1 | 249 | 1.2659 | �� | ���� |
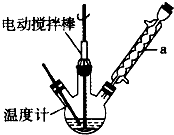
ʵ���ҿ�����ͼװ��ģ���Ʊ�����ȩ��ʵ��ʱ��������ƿ�м���0.5g��̬�����Դ������ټ���15mL�������
2mL�ױ�������������70�棬ͬʱ��������12mL�������⣬�ڴ��¶��½��跴Ӧ3Сʱ��
��1������a����Ҫ������������������ֹ�ױ��ӷ����²��ʽ��ͣ�����ƿ�з�����Ӧ�Ļ�ѧ����ʽΪ
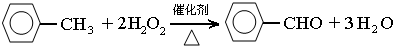 ��
����2�����ⶨ����Ӧ�¶�����ʱ���ױ���ת�����������¶ȹ���ʱ������ȩ�IJ���ȴ�������٣����ܵ�ԭ�����¶ȹ���ʱ��������ֽ��ٶȼӿ죬ʵ�ʲμӷ�Ӧ�Ĺ�������������С��Ӱ�������
��3����Ӧ��Ϻ�Ӧ���Һ������Ȼ��ȴ������ʱ����Ӧ�������ˡ�������������ƣ��Ȳ��������ܵõ�����ȩ�ֲ�Ʒ��
��4��ʵ���м�������Ĺ������Ⲣ�ӳ���Ӧʱ��ʱ����ʹ����ȩ��Ʒ�в����϶�ı����ᣮ
������ӻ��б�����ı���ȩ�з���������ᣬ��ȷ�IJ���������dacb��������˳������ĸ����
a���Ի��Һ���з�Һ b�����ˡ�ϴ�ӡ�����
c��ˮ���м����������pH=2 d��������̼��������Һ�����
������ʵ����л�õı������Ʒ���д��Ȳⶨ���ɳ�ȡ2.500g��Ʒ������100mL�Ҵ������Һ����ȡ���õ��Ҵ���Һ10.00mL����ƿ���μ�2��3�η�ָ̪ʾ����Ȼ����Ԥ����õ�0.1000mol/LKOH��Һ�ζ�������ζ��յ�ʱ����KOH��Һ20.00mL����Ʒ�б��������������Ϊ97.60%�����������ʹ�ⶨ���ƫ�͵���ad������ĸ����
a���ζ�ʱ���Ӷ�ȡ�ļ��� b����ʽ�ζ���������ˮϴ����ʢװKOH��Һ
c������KOH��Һʱ���Ӷ��� d������ָ̪ʾ����Ϊ������Һ��