��Ŀ����
12���л�������F�Ǻϳɵ��ӱ�Ĥ���ϸ߾���Z �����ܼ�P����Ҫԭ�ϣ���1��ijͬѧ���������ϩ�ϳɸ߾���Z��3��·�ߣ�I��II��III����ͼ��ʾ��
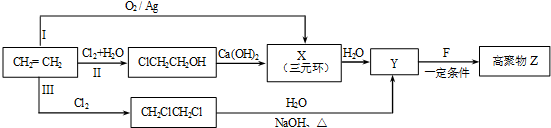
��3���ϳ�·���У�����Ϊ���ϡ�ԭ�Ӿ��á�Ҫ��ĺϳ�·���ǣ�����š�I������II����III����I��
��X�Ľṹ��ʽ��
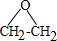 ��
����1mol F��O2�г��ȼ�գ�����7.5mol O2������8molCO2 ��3molH2O��1mol F������NaHCO3��Һ��Ӧ����2mol CO2��������ڵ���ԭ�Ӵ���3�ֲ�ͬ�Ļ�ѧ������
F�����к��еĺ��������ŵ��������Ȼ���
Y+F��Z��Ӧ�Ļ�ѧ����ʽ��
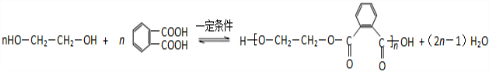 ��
����2����֪��
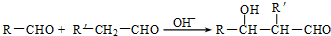 ��R��R'������������ԭ�ӣ����ϳ�P��·������ͼ��ʾ��D��������8��̼ԭ�ӣ�����������6��̼ԭ�ӣ��ҷ�����ֻ��������-CH3��
��R��R'������������ԭ�ӣ����ϳ�P��·������ͼ��ʾ��D��������8��̼ԭ�ӣ�����������6��̼ԭ�ӣ��ҷ�����ֻ��������-CH3��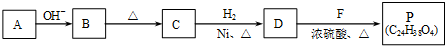
��A��B��Ӧ�Ļ�ѧ����ʽ��
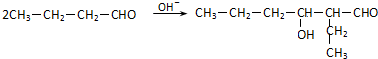 ��
����B��C�ķ�Ӧ�У�B�����ڼ�����������ȥһ��ˮ���ӣ�����C��C������ֻ��1��̼ԭ��������ԭ�ӣ�C�Ľṹ��ʽ��
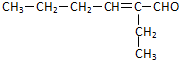 ��
����P�Ľṹ��ʽ��
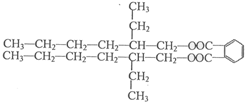 ��
���ܷ�������������B��ͬ���칹�干�У������֣�2�֣�
a��������������ˮ��ΪM��N b��һ��������M����ת��ΪN��
���� ��1����ϩ��Ag�����������±�������������X��X����Ԫ������ClCH2CH2OH���������Ʒ�ӦҲ����X����XΪ�������飬��ṹ��ʽΪ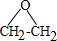 ��1��2-����������������Ƶ�ˮ��Һ��������Y����Y�Ľṹ��ʽΪHOCH2CH2OH��1mol F��O2�г��ȼ�գ�����7.5mol O2������8molCO2 ��3molH2O������ԭ���غ�֪һ��F�����к���8��Cԭ�ӡ�6��Hԭ�ӡ�4��Oԭ�ӣ�1mol F������NaHCO3��Һ��Ӧ����2mol CO2��˵��F�к���2���Ȼ���������ڵ���ԭ�Ӵ���3�ֲ�ͬ�Ļ�ѧ��������F�Ľṹ��ʽΪ
��1��2-����������������Ƶ�ˮ��Һ��������Y����Y�Ľṹ��ʽΪHOCH2CH2OH��1mol F��O2�г��ȼ�գ�����7.5mol O2������8molCO2 ��3molH2O������ԭ���غ�֪һ��F�����к���8��Cԭ�ӡ�6��Hԭ�ӡ�4��Oԭ�ӣ�1mol F������NaHCO3��Һ��Ӧ����2mol CO2��˵��F�к���2���Ȼ���������ڵ���ԭ�Ӵ���3�ֲ�ͬ�Ļ�ѧ��������F�Ľṹ��ʽΪ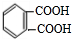 ��Y��F��Ũ���������������������·������۷�Ӧ����Z����Z�Ľṹ��ʽΪ
��Y��F��Ũ���������������������·������۷�Ӧ����Z����Z�Ľṹ��ʽΪ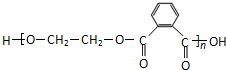 ��
��
��2��D��������8��̼ԭ�ӣ�����������6��̼ԭ�ӣ��ҷ�����ֻ��������-CH3����֪AΪCH3CH2CH2CHO��BΪ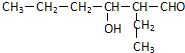 ��CΪ
��CΪ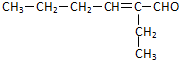 ��DΪ
��DΪ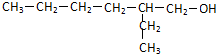 ��PΪ
��PΪ ��
��
��� �⣺��1����ϩ��Ag�����������±�������������X��X����Ԫ������ClCH2CH2OH���������Ʒ�ӦҲ����X����XΪ�������飬��ṹ��ʽΪ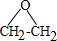 ��1��2-����������������Ƶ�ˮ��Һ��������Y����Y�Ľṹ��ʽΪHOCH2CH2OH��1mol F��O2�г��ȼ�գ�����7.5mol O2������8molCO2 ��3molH2O������ԭ���غ�֪һ��F�����к���8��Cԭ�ӡ�6��Hԭ�ӡ�4��Oԭ�ӣ�
��1��2-����������������Ƶ�ˮ��Һ��������Y����Y�Ľṹ��ʽΪHOCH2CH2OH��1mol F��O2�г��ȼ�գ�����7.5mol O2������8molCO2 ��3molH2O������ԭ���غ�֪һ��F�����к���8��Cԭ�ӡ�6��Hԭ�ӡ�4��Oԭ�ӣ�
1mol F������NaHCO3��Һ��Ӧ����2mol CO2��˵��F�к���2���Ȼ���������ڵ���ԭ�Ӵ���3�ֲ�ͬ�Ļ�ѧ��������F�Ľṹ��ʽΪ ��Y��F��Ũ���������������������·������۷�Ӧ����Z����Z�Ľṹ��ʽΪ
��Y��F��Ũ���������������������·������۷�Ӧ����Z����Z�Ľṹ��ʽΪ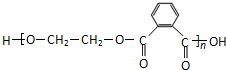 ��
��
������ϩ�ϳɸ߾���Z��3��·�ߢ���������ϡ�ԭ�Ӿ��á�Ҫ���и�����CaCl2��ˮ�������ϡ�ԭ�Ӿ��á�Ҫ��
�ʴ�Ϊ��I��
��ͨ�����Ϸ���֪��X�Ľṹ��ʽ��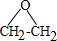 ���ʴ�Ϊ��
���ʴ�Ϊ��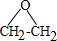 ��
��
��ͨ�����Ϸ���֪��F�Ľṹ��ʽΪ ����������������Ȼ���
����������������Ȼ���
Y��F��Ũ���������������������·������۷�Ӧ����Z���÷�Ӧ����ʽΪ ��
��
�ʴ�Ϊ���Ȼ��� ��
��
��2��D��������8��̼ԭ�ӣ�����������6��̼ԭ�ӣ��ҷ�����ֻ��������-CH3����֪AΪCH3CH2CH2CHO��BΪ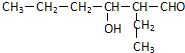 ��CΪ
��CΪ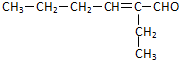 ��DΪ
��DΪ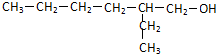 ��PΪ
��PΪ ��
��
��AΪCH3CH2CH2CHO��BΪ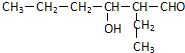 ��A��B�ķ�Ӧ�ķ���ʽΪ
��A��B�ķ�Ӧ�ķ���ʽΪ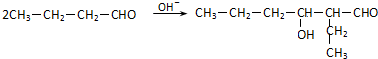 ��
��
�ʴ�Ϊ��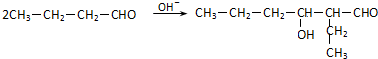 ��
��
��ͨ�����Ϸ���֪��C�Ľṹ��ʽΪ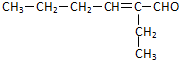 ��
��
�ʴ�Ϊ��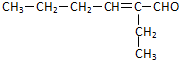 ��
��
��ͨ�����Ϸ���֪P�Ľṹ��ʽ�� ��
��
�ʴ�Ϊ�� ��
��
��BΪ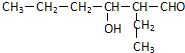 ����ͬ���칹����ϣ�a��������������ˮ��ΪM��N��˵������������b��һ��������M����ת��ΪN����M��N������̼ͬԭ����Ŀ��NΪ���ᣬMΪ���Һ���-CH2OH���ţ�����������B��ͬ���칹����CH3-CH2-CH2COO-CH2-CH2-CH2-CH3��CH3CH��CH3��COOCH2CH��CH3��CH3����2�֣�
����ͬ���칹����ϣ�a��������������ˮ��ΪM��N��˵������������b��һ��������M����ת��ΪN����M��N������̼ͬԭ����Ŀ��NΪ���ᣬMΪ���Һ���-CH2OH���ţ�����������B��ͬ���칹����CH3-CH2-CH2COO-CH2-CH2-CH2-CH3��CH3CH��CH3��COOCH2CH��CH3��CH3����2�֣�
�ʴ�Ϊ��2��
���� ���⿼���л����ƶ���ϳɣ�����ƴ������Ŀ�����ؿ���ѧ������������֪ʶǨ��Ӧ���������������ճ����л���Ĺ����ż������ʣ���Ŀ�Ѷ��еȣ�

 Сѧͬ�����������ܾ�ϵ�д�
Сѧͬ�����������ܾ�ϵ�д�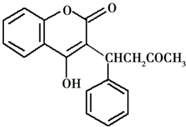
| A�� | ������ķ���ʽΪC19H17O4 | |
| B�� | �����������ˮ�����ӳɷ�Ӧ | |
| C�� | ���������е�̼ԭ�Ӷ�������ͬһ��ƽ���� | |
| D�� | 1 mol������������8 mol H2 ��Ӧ |
| Ԫ�ش��� | X | Y | Z | W |
| ԭ�Ӱ뾶/nm | 0.160 | 0.143 | 0.102 | 0.074 |
| ��Ҫ���ϼ� | +2 | +3 | +6��-2 | -2 |
| A�� | X��Y�ֱ���W�γɵĻ����ﶼ�������� | |
| B�� | Z��W��ͬһ���� | |
| C�� | ��̬�⻯����ȶ���ΪH2W��H2Z | |
| D�� | X��Y��Ӧ����������ļ���ǿ���Ƚϣ�X��OH��2��Y��OH��3 |
| A�� | pH=5��H2S��Һ�У�c��H+��=c��HS-��=1��10-5 mol•L-1 | |
| B�� | pH=a�İ�ˮ��Һ��ϡ��10������pH=b����a��b+1 | |
| C�� | pH=9�Ģ�CH3COONa����NaHCO3����NaClO������Һ��c��Na+�����٣��ۣ��� | |
| D�� | pH=2��H2C2O4��Һ��pH=12��NaOH��Һ���������ϣ�c��Na+��+c��H+��=c��OH-��+c��HC2O4-��+2c��C2O42-�� |
| A�� | pH HCl��CH3COOH | |
| B�� | c��Cl-��=c��CH3COO-�� | |
| C�� | �����������ʹ�����Һ�ֱ���������Zn��ȫ��Ӧ�����������H2�� | |
| D�� | ��ͬŨ�ȵ�NaOH��Һ�ֱ�������������ʹ�����Һǡ�÷�Ӧ��ȫ�����ĵ�NaOH��Һ���һ���� |
| A�� | NaCl��KNO3��AlCl3��Fe��NO3��3 | B�� | Fe��FeO��CuO��FeS��CuS��MnO2 | ||
| C�� | MgCl2��FeCl2��KCl��AlCl3��NH4Cl | D�� | K2CO3��NaI��Na2CO3��NaCl��Na2SO4 |
| A�� | ������60gSiO2���������Ӧ������ͷų�2NA��������� | |
| B�� | 1molC5H12�����й��ۼ�����Ϊ14NA | |
| C�� | 78gNa2O2������������H2O��Ӧת�Ƶĵ�����ΪNA | |
| D�� | ��״���£�2.24LNH3��CH4�Ļ�����壬������������Ϊ2NA |
| A�� | ����������ȼ�� | B�� | ����������ȼ�� | ||
| C�� | ������������ȼ�� | D�� | ����������ȼ�� |