��Ŀ����
þ���Ͻ�5.1g����300mL 2mol?L-1�����У���״���·ų��������Ϊ5.6L����Ӧ�����Һ�м���500mL NaOH��Һ��ַ�Ӧ�����ղ�����ɫ�����������й�˵������ȷ���ǣ�������
| A��5.1 gþ���Ͻ��к���þ�����������ֱ�Ϊ2.4 g��2.7 g |
| B���ܽ�5.1 gþ���Ͻ�ʱ��ʣ����������ʵ���Ϊ0.1 mol |
| C�����ɰ�ɫ�������������Ϊ13.6 g |
| D������ɫ����ֻ��Mg��OH��2����NaOH��Һ�����ʵ���Ũ������Ϊ1.2 mol?L-1 |
���㣺þ��������Ҫ������,��ѧ����ʽ���йؼ���
ר�⣺������Ҫ�Ľ������仯����
������������ӦMg+2HCl=MgCl2+H2����2Al+6HCl=2AlCl3+3H2����5.6L���������������������ʵ���Ϊ
=0.25mol���ʲμӷ�Ӧ��HCl�����ʵ���Ϊ0.25mol��2=0.5mol����Һ��n��HCl��=0.3L��2mol/L=0.6mol�����ڲμӷ�Ӧ��HCl�����ʵ������ʽ�����ȫ��Ӧ����Ӧ����Һ�м���һ�����ռ���Һ�����ɳ��������������þ���ӡ�������ǡ����ȫת��ΪMg��OH��2��Al��OH��3����m��������=m��������+m��OH-���������ɵ����������ʵ��������ݵ���ע���غ����n��OH-�����ٸ���m=nM����m��OH-�����ɴ˷������
| 5.6L |
| 22.4L/mol |
���
�⣺������ӦMg+2HCl=MgCl2+H2����2Al+6HCl=2AlCl3+3H2����5.6L���������������������ʵ���Ϊ
=0.25mol���ʲμӷ�Ӧ��HCl�����ʵ���Ϊ0.25mol��2=0.5mol����Һ��n��HCl��=0.3L��2mol/L=0.6mol�����ڲμӷ�Ӧ��HCl�����ʵ������ʽ�����ȫ��Ӧ��
��Ӧ����Һ�м���һ�����ռ���Һ�����ɳ��������������þ���ӡ�������ǡ����ȫת��ΪMg��OH��2��Al��OH��3����m��������=m��������+m��OH-����
A����þ�������ʵ����ֱ�Ϊxmol��ymol����
����ã�
������5.1gþ���Ͻ��к���þ�����������ֱ�Ϊ2.4g��2.7g����A��ȷ��
B�����������ʵ���Ϊ0.25mol���ʲμӷ�Ӧ��HCl�����ʵ���Ϊ0.25mol��2=0.5mol����Һ��n��HCl��=0.3L��2mol/L=0.6mol��ʣ����������ʵ���Ϊ0.1mol����B��ȷ��
C�����ݵ���ע���غ��֪n��OH-��=2n��H2��=0.25mol��2=0.5mol���ʳ������������Ϊ5.1g+0.5mol��17g/mol=13.6g����C��ȷ��
D������ɫ�������ʱ����ʱ����Ϊ��һ���Ȼ��ƣ��������غ㣬�����������Ƶ����ʵ���Ϊ0.6mol����NaOH��Һ�����ʵ���Ũ��Ϊ
=1.2mol?L-1��NaOH��Һ�����ʵ���Ũ�ȴ���1.2mol?L-1����D����
��ѡD��
| 5.6L |
| 22.4L/mol |
��Ӧ����Һ�м���һ�����ռ���Һ�����ɳ��������������þ���ӡ�������ǡ����ȫת��ΪMg��OH��2��Al��OH��3����m��������=m��������+m��OH-����
A����þ�������ʵ����ֱ�Ϊxmol��ymol����
|
|
B�����������ʵ���Ϊ0.25mol���ʲμӷ�Ӧ��HCl�����ʵ���Ϊ0.25mol��2=0.5mol����Һ��n��HCl��=0.3L��2mol/L=0.6mol��ʣ����������ʵ���Ϊ0.1mol����B��ȷ��
C�����ݵ���ע���غ��֪n��OH-��=2n��H2��=0.25mol��2=0.5mol���ʳ������������Ϊ5.1g+0.5mol��17g/mol=13.6g����C��ȷ��
D������ɫ�������ʱ����ʱ����Ϊ��һ���Ȼ��ƣ��������غ㣬�����������Ƶ����ʵ���Ϊ0.6mol����NaOH��Һ�����ʵ���Ũ��Ϊ
| 0.6mol |
| 0.5L |
��ѡD��
���������⿼��������йؼ��㣬�Ѷ��еȣ�ע��ӵ���ת���غ����n��OH-���ǹؼ���

��ϰ��ϵ�д�
�����Ŀ
��ϡ�����м���ͭ�ۣ�ͭ�۲��ܽ⣬�ټ���ijX���ʣ�����ͭ�����ܽ⣬�����ص�X�������ǣ�������
| A��FeCl3 |
| B��HNO3 |
| C��HCl |
| D��KNO3 |
��֪Ԫ��R��Q�ļ۵����Ų����£������γ� RQ2�ͻ�������ǣ�������
| A��2s22p2 ��2s22p4 |
| B��3s23p4 ��2s22p4 |
| C��3s2 ��2s22p5 |
| D��3s1 ��3s23p4 |
������ѧ�л���ѧ֪ʶ�ж����б�����������ȷ���ǣ�������
| A���ô�������ľ���һ���������ܷⱣ�棬ʱ��Խ��Խ�㴼 |
| B�����������ʴ���Һ���������ֲ��ı��������ʣ��ɼ�CuSO4��Һ |
| C����ϩʹ��ˮ��ɫ�뱽ʹ��ˮ��ɫ��ԭ����ͬ |
| D����ά�ء����ǡ������Ǻ�֬����һ�������¶��ɷ���ˮ�ⷴӦ���� |
��ͼ��ʾΪ��ˮ�ۺ����ò������̣��й�˵��������ǣ���������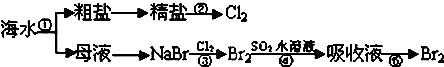
| A��ʵ���ҽ��ТٵIJ������õ����������������ƾ��� |
| B������һ��������ת��Ϊ��ѧ�ܵĹ��� |
| C���ۢܢ��漰�ķ�Ӧ��Ϊ������ԭ��Ӧ |
| D��NaCl��Һ�����ᾧʱ�����������д�����������ʱֹͣ���� |
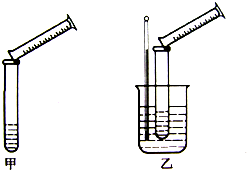 �����������ϡ����ķ�ӦΪ��Na2S2O3+H2SO4=Na2SO4+S��+SO2��+H2O��ijʵ����ȤС��������ͼ��ʾ��ʵ��װ��̽����������Ի�ѧ��Ӧ���ʵ�Ӱ�죨���������ͼг���������ʡ�ԣ�
�����������ϡ����ķ�ӦΪ��Na2S2O3+H2SO4=Na2SO4+S��+SO2��+H2O��ijʵ����ȤС��������ͼ��ʾ��ʵ��װ��̽����������Ի�ѧ��Ӧ���ʵ�Ӱ�죨���������ͼг���������ʡ�ԣ�