��Ŀ����
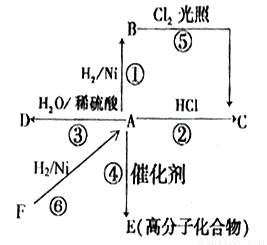
��֪Aʯ���ѽ����Ҫ�����Է�������Ϊ28��BΪ���壬�ڱ�״���µ��ܶ�Ϊ1.96g/L��D��F��Ϊͬ���칹�壬F�ǻ�״�����G�Ľṹ��ʽΪ��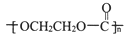 ����һ�������£���������ͼת����ϵ��
����һ�������£���������ͼת����ϵ��

��ش�
��1��E�й����ŵ�����______________��
��2��B��F��Ӧ����G�ķ�Ӧ����Ϊ_____________��
��3��A F�Ļ�ѧ��Ӧ����ʽ______________��
F�Ļ�ѧ��Ӧ����ʽ______________��
��4������˵������ȷ����______________
A.�����ý���Na����C���Ƿ�������ˮ
B.������С�մ���Һ����D��E��ˮ��Һ
C.��A����C��A����D������������Ӧ
D.G������ϡ�����NaOH��Һ�з���ˮ��
 �Ķ��쳵ϵ�д�
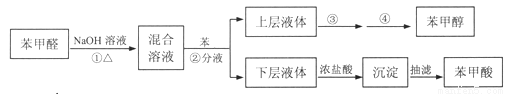
�Ķ��쳵ϵ�д��������⡿����ȩ������ˮ���������л��ܼ����ܶ�Լ����ˮ���ܶȣ��ڼ��������·����绯��Ӧ�����Ʊ�����ȩ����ˮ���ܽ�Ȳ����������л��ܼ����ܶ�Լ����ˮ���ܶȣ��������ᡣ��Ӧԭ�����£�
2C6H5CHO+NaOH C6H5CH2OH+C6H5COONa
C6H5CH2OH+C6H5COONa
C6H5COONa+HCl C6H5COOH+NaCl
C6H5COOH+NaCl
������������������±���
����ȩ | ���״� | ������ | �� | |
�е�/�� | 178 | 205 | 249 | 80 |
�۵�/�� | 26 | -15 | 122 | 5.5 |
��������ˮ�е��ܽ�� | ||
17�� | 25�� | 100�� |
0.21g | 0.34g | 5.9g |
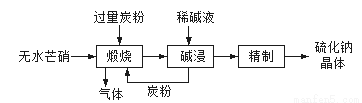
ʵ���������£�
��1���ڢ�����������1Сʱ����ͼ1�������м��Ȼ�Ϲ̶�װ��Ϊ������
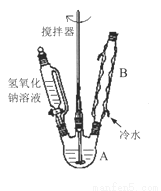
����A������Ϊ_______����������B��Ϊ����C��Ч������B��˵��ԭ��_______��
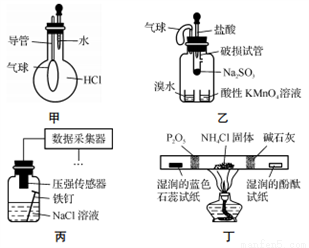
��2���������йط�Һ©����ʹ�ò���ȷ����_______
A.��Һ©����ʹ��֮ǰ��������Ƿ�©ˮ
B.��Һ©���ڵ�Һ�岻�ܹ��࣬����������
C.�����Һ©����������̨�Ͼ��ã��ֲ���������������з�Һ
D.��Һʱ���²�Һ�����������ر������������ձ��ٴ�����ʹ�ϲ�Һ������
��3�����������÷�ˮԡ���������ٽ��в����ܣ���ͼ2�����ռ�______�����֡�ͼ2����һ�����Դ�����ȷ��Ӧ��Ϊ_____________��
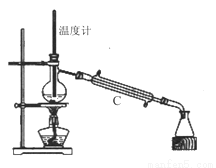
��4������ʱ����ͼ3���ձ��б����ᾧ��ת�벼��©��ʱ�������ϻ�ճ���������壬��_____��ϴ�����ϲ����ľ��塣������ɺ���������ˮ�Ծ������ϴ�ӣ�ϴ��Ӧ____________��
��5���õ�����ƽȷ��ȡ0.2440g����������ƿ�м�100mL����ˮ�ܽ⣨��Ҫʱ���Լ��ȣ�������0.1000mol/L�ı�����������Һ�ζ��������ı�����������Һ19.20mL��������Ĵ���Ϊ_____%��


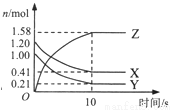
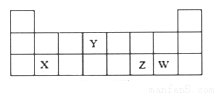
 7N2��12H2O�����й��ڸ÷�Ӧ˵����ȷ����
7N2��12H2O�����й��ڸ÷�Ӧ˵����ȷ����